Click the "draw structure" button to launch the drawing utility. Follow the curved arrows to draw a second resonance structure for the following species. CH, -C-CH' CH,
Click the "draw structure" button to launch the drawing utility. Follow the curved arrows to draw a second resonance structure for the following species. CH, -C-CH' CH,
Chapter6: The States Of Matter
Section: Chapter Questions
Problem 6.9E
Related questions
Question
First pictures was my answer but its wrong. The second picture is telling why my answer was wrong. Please help me why and correct it for me. Thanks
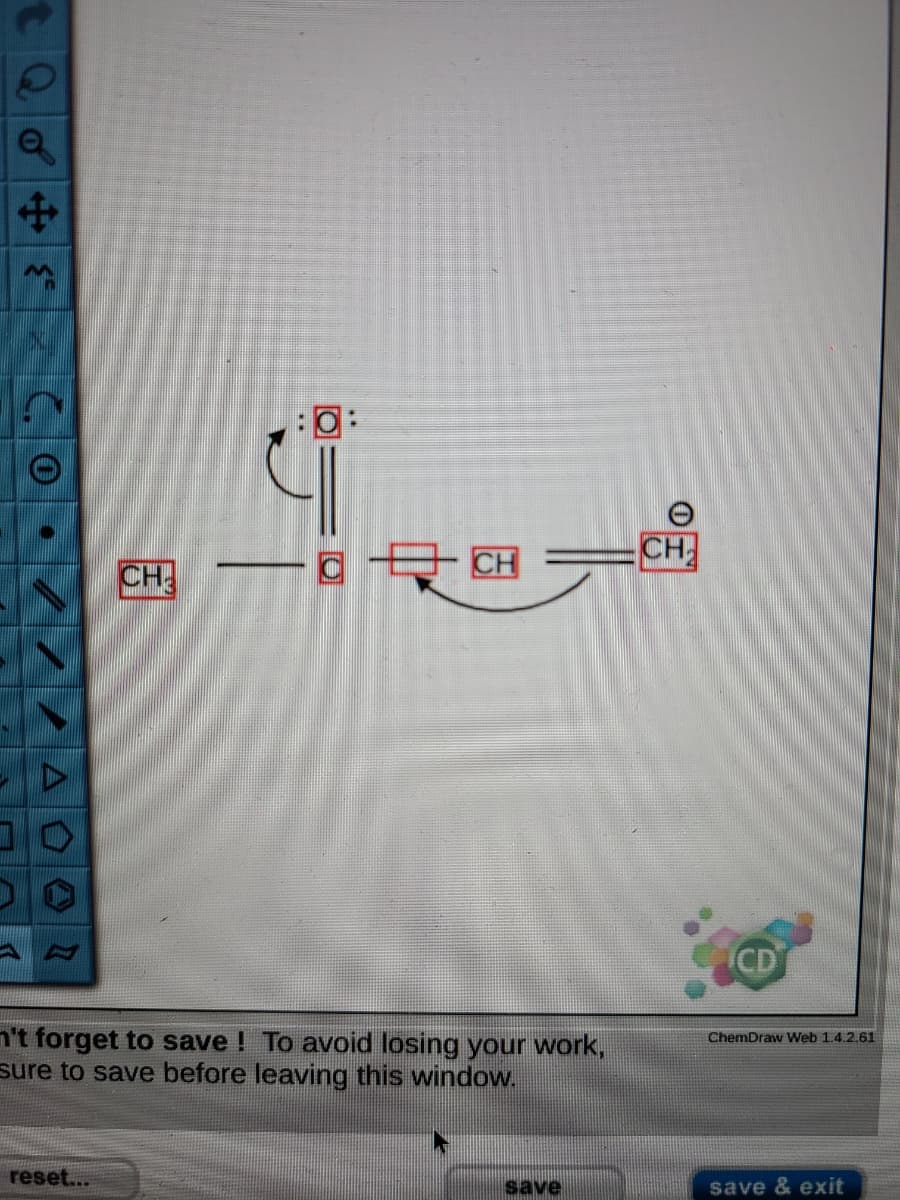
Transcribed Image Text:- CH
CH
CH
CD
n't forget to save ! To avoid losing your work,
sure to save before leaving this window.
ChemDraw Web 1.4.2.61
reset...
save
save & exit
o中
Transcribed Image Text:t 2- Ce X
O (18) Chapter 2 The Nature of Mi
nheducation.com/ext/map/index.html?_con3con&external_browser%-D08launchUrl=https%253A%252F%252Fblackboard.matc.edu%252Fwebapps%252
art 2
Saved
1 attempts left
Check my work
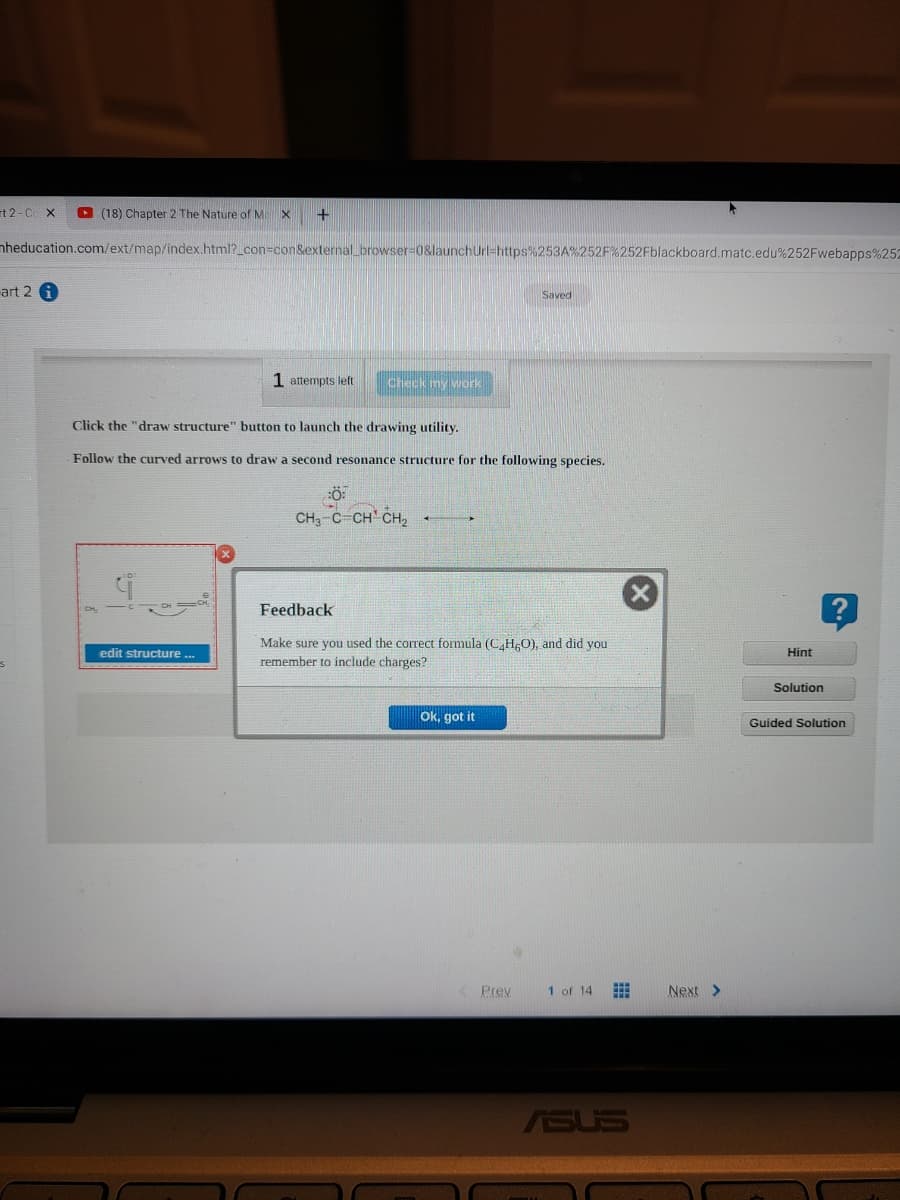
Click the "draw structure" button to launch the drawing utility.
Follow the curved arrows to draw a second resonance structure for the following species.
CH,-C-CH CH,
Feedback
Make sure you used the corTect formula (C H,O), and did you
remember to include charges?
edit structure .
Hint
Solution
Ok, got it
Guided Solution
Prey
1 of 14
Next >
ASUS
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

