Color of Solution Solution Solute A Na,CrO4 Yellow (NH4)2C2O4 Colorless B AgNO3 Colorless D CaCl2 Colorless Experiment Solutions Mixed Number Result A + B A + C No precipitate, yellow solution Red precipitate forms A + D Yellow precipitate forms B + C White precipitate forms B + D White precipitate forms C + D White precipitate forms 3.
Color of Solution Solution Solute A Na,CrO4 Yellow (NH4)2C2O4 Colorless B AgNO3 Colorless D CaCl2 Colorless Experiment Solutions Mixed Number Result A + B A + C No precipitate, yellow solution Red precipitate forms A + D Yellow precipitate forms B + C White precipitate forms B + D White precipitate forms C + D White precipitate forms 3.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter15: Complex Ion And Precipitation Equilibria
Section: Chapter Questions
Problem 64QAP
Related questions
Question
You choose to investigate some of the solubility guidelines
for two ions , the chromate ion(CrO4)-2 and the oxalate ion (C2O4) - 2. You are given 0.01 M
solutions (A, B, C, D) of four water-soluble salts:
When these solutions are mixed, the following observations
are made as given in table:
(a) Write a net ionic equation for the reaction that occurs in
each of the experiments. (b) Identify the precipitate formed,
if any, in each of the experiments.
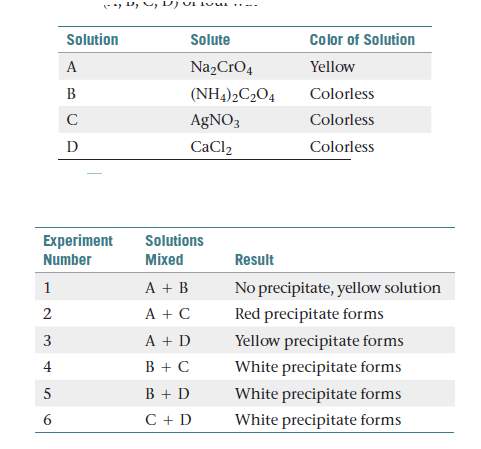
Transcribed Image Text:Color of Solution
Solution
Solute
A
Na,CrO4
Yellow
(NH4)2C2O4
Colorless
B
AgNO3
Colorless
D
CaCl2
Colorless
Experiment
Solutions
Mixed
Number
Result
A + B
A + C
No precipitate, yellow solution
Red precipitate forms
A + D
Yellow precipitate forms
B + C
White precipitate forms
B + D
White precipitate forms
C + D
White precipitate forms
3.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax