Combined Spectra 14.46 Identify the C3 H5Br isomers on the basis of the following information: (a) Isomer A has the 'H NMR spectrum shown in 4 Figure 14.49. (b) Isomer B has three peaks in its 13C NMR spectrum: 8 32.6 (CH2); 118.8 (CH2 ); and 134.2 (CH). (c) Isomer C has two peaks in its 13C NMR spectrum: ô 12.0 (CH2) and 16.8 (CH). The peak at lower field is only half as intense as the one at higher field. 10 9 8 7 4 3 2 1 Chemical shift (8, ppm)
Combined Spectra 14.46 Identify the C3 H5Br isomers on the basis of the following information: (a) Isomer A has the 'H NMR spectrum shown in 4 Figure 14.49. (b) Isomer B has three peaks in its 13C NMR spectrum: 8 32.6 (CH2); 118.8 (CH2 ); and 134.2 (CH). (c) Isomer C has two peaks in its 13C NMR spectrum: ô 12.0 (CH2) and 16.8 (CH). The peak at lower field is only half as intense as the one at higher field. 10 9 8 7 4 3 2 1 Chemical shift (8, ppm)
Chapter12: Structure Determination: Mass Spectrometry And Infrared Spectroscopy
Section12.SE: Something Extra
Problem 19AP: Nicotine is a diamino compound isolated from dried tobacco leaves. Nicotine has two rings and...
Related questions
Question
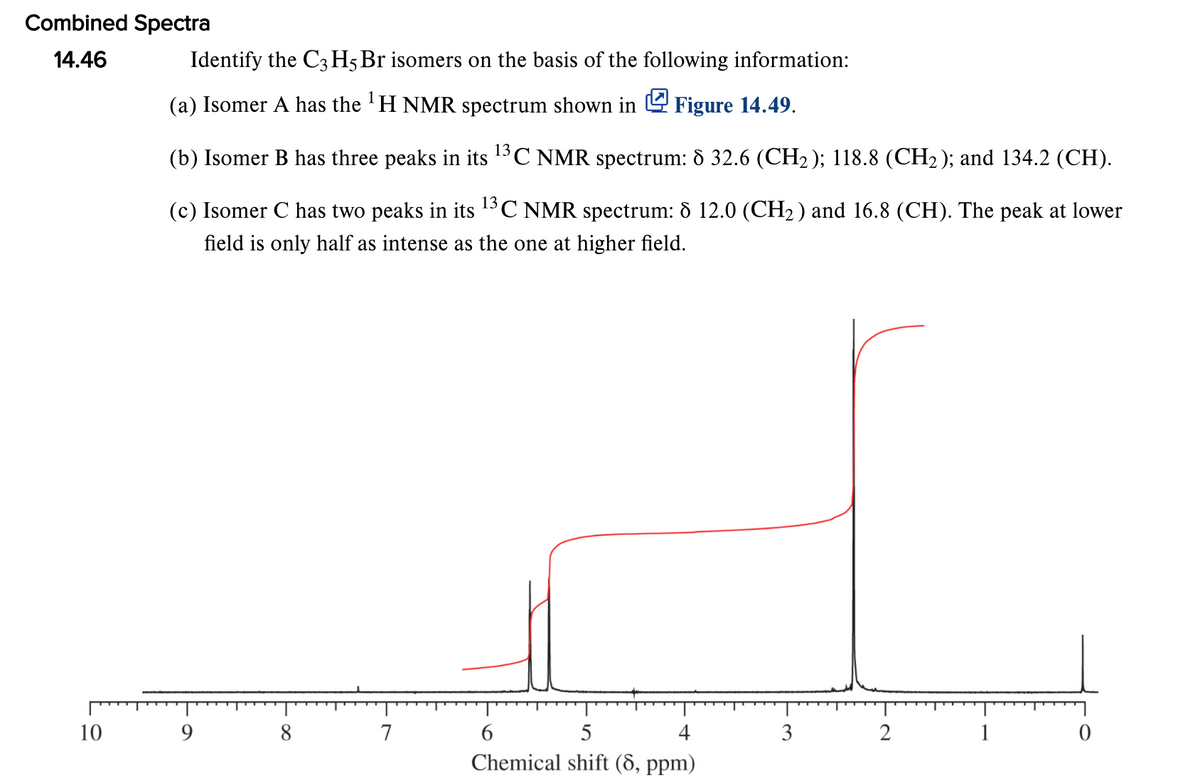
Transcribed Image Text:Combined Spectra
14.46
Identify the C3 H5 Br isomers on the basis of the following information:
(a) Isomer A has the 'H NMR spectrum shown in 4 Figure 14.49.
(b) Isomer B has three peaks in its 13C NMR spectrum: 8 32.6 (CH2 ); 118.8 (CH2 ); and 134.2 (CH).
(c) Isomer C has two peaks in its 1°C NMR spectrum: 8 12.0 (CH2) and 16.8 (CH). The peak at lower
field is only half as intense as the one at higher field.
10
9.
8
7
6.
5
4
3
2
1
Chemical shift (8, ppm)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

