Q: 1a. How many moles of M9SO, are contained in 50. mL of a 3.0 M solution? 1b. How many grams of CaCl,...
A: Formula of molarity is:- Molarity = moles ÷ volume(L)
Q: 3. Calculate the kg/cmol for Ca*2 (review the calculations from Lab 1). If a soil can retain 25 cmol...
A: Answer and explanation is given below hope you like it.
Q: Compound A has a heat of combustion of -2300 kJ/mol. Compound B, a stereoisomer of Compound A, has a...
A:
Q: A sample of pure t-butyl alcohol weighing 7.512 g was found to have a freezing point of 24.7 °C. Whe...
A:
Q: Determine the entropy difference between solid and liquid states for a substance melting at 100 °C a...
A:
Q: Find out what mass of CaSO3 must have been present initially in order for a factory to produce 1.39...
A:
Q: Which solution would have the lowest boiling point elevation? 3.0 m NaCl O 3.0 m CaBr2 O 4.0 m Na3PO...
A:
Q: What is the pH of a 1.2 M pyridine solution that has Kp = 1.9 x 10 9? The equation for the dissociat...
A: The concentration of pyridine = 1.2 M Kb = 1.9 × 10-9 The dissociation equation of pyridine : C5H...
Q: a ketone and an ether?
A:
Q: ionic
A:
Q: What is the pH of a solution containing 1.089 mol L-1 of a weak acid with pKA = 7.27 and 0.712 mol L...
A:
Q: Examine the given Beer's law standard curve for an unknown dye measured in a 1.0 cm cuvette. Absorba...
A: According to Beer's Law, A = (εl).c For a graph between A (Absorbance) and c (concentration), Slop...
Q: Annotate the given IR spectrum. Does this IR spectrum match one of chalcone?
A: IR spectrum of a chalcone
Q: 4. Which sample represents the smallest number of moles? a. 1L H2 at STP b. 1L Argon at STP C. 1L of...
A:
Q: The typical grade of Pine Point ore is 3% for Pb, and 7% for Zn. Suppose the sample pictured in the ...
A: Given , typical grade of pine point ore is 3% pb . 7% Zn And total weight of the sample = 13.6kg C...
Q: empirical formula f
A:
Q: Find the Ka of a weak acid ( HA ) that has a concentration of 0.020 M. The pH is 2.74.
A: Weak acid [H+] and Ka are related as [H+] = ( Ka x C)1/2 Where C = concentration of acid = 0.02 ...
Q: A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains 6....
A: As given that heat flow out of the water that means heat is lost by it and it's temperature would ...
Q: In separate titrations, equal volumes of 0.1 M HCl and 0.1 M HC2H3O2 (weak acid) are titrated with 0...
A: Answer - The correct option is (d) The pH at the equivalence point is greater for HC2H3O2 than for H...
Q: Given the potential difference between the terminals of a battery as 5 Volts, calculate the number o...
A:
Q: A 25.0 ml sample of 6.00 M HCl is titrated with 25.0 ml of 6.00 M NaOH solution. What is the pH of t...
A: Here acid base titration occurred, here the strength and volume of HCl and NaOH is same, so acid tot...
Q: In EDTA titrations, a,4- is an important param as a function of [H']. (Note: The definition of dyt- ...
A: To derive an expression of αY4- as a function of H+ in EDTA titrations
Q: explain how to count H-bond acceptors and donors for Lipinski Rule one more time.
A:
Q: What is the density of the 2Mg nucleus in g/mL, assuming that it has the typical nuclear diameter of...
A:
Q: What is the molarity of a solution of nitric acid if 0.216 g of barium hydroxide is required to neut...
A: 2HNO3(aq) + Ba(OH)2(aq) -----> Ba(NO3)2 +2H2O(l) Mass of Ba(OH)2 = 0.216 g Volume of HNO3 = 20 mL...
Q: Calculate the ionic strength of 0.040 M Na2SO4.
A: Dear student, Since you have posted multiple questions, the answer for first question is given below...
Q: Balance the molecular equation and write the complete ionic and net ionic equation Ca(NO3)2 (aq) + ...
A:
Q: Mercury (II) oxide creates a beautiful red precipitate. It is also a VERY toxic substance, which abs...
A:
Q: What is the name of the sugar shown here? OH OH O L-xylose O D-xylose O D-erythrose O L-erythrose O ...
A: To identify the name for given Sugar.
Q: -y positron emission?
A:
Q: Name of Experiment: Flame atomic Absorption spectroscopy (FAAS): Determination of Lead in environmen...
A:
Q: How many electrons, protons, and neutrons are there in the ion of the hypothetical 445A-3+ element 1...
A:
Q: Rewrite this measurement with a simpler unit, if possible. kg-L's 4.2 kg s Note: If you can simplify...
A: Here simply use division rule , eliminate same unit when they are in ratio . So final measurement wi...
Q: ne equilibrium concentrations for
A:
Q: At a certain temperature, the pH of a neutral solution is 7.90. What is the value of Kw at that temp...
A: Given -> pH = 7.90
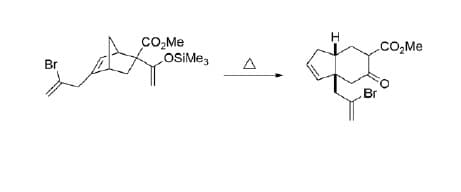
Q: Buli SnMeg
A: EXPLANATION : Given reaction is 2,3-sigmatropic rearrangements reaction. 2,3-sigmatropic rearra...
Q: This reaction generates 7.70 L of oxygen at a temperature of 18.7°C and a pressure of 0.92 atm, what...
A: Here the decomposition reaction of KClO3 solid gives KCl solid and O2 gas. The volume of O2 produced...
Q: Write the balanced chemical equation for the reaction of the weak acid HCN with water. Include the p...
A: The reaction of the weak acid HCN with water, The balanced chemical equation is :
Q: Below is an unbalanced molecular equation that describes what happens when solutions of cobalt (II) ...
A: The unbalanced molecular equation is., Co(ClO4)2 (aq) + Na3PO4 (aq) → NaClO4 (aq) + Co3(PO4)2 (...
Q: A raindrop has a mass of 50. mg and the Pacific Ocean has a mass of 7.08 x 100 kg. Use this informat...
A:
Q: What is the conjugate acid of HPO2²- ? Express your answer as a chemical formula.
A: • We need to write the conjugate acid of HPO22-
Q: Identify the acids, bases, conjugate acids, and conjugate bases in the reaction below. "C." stands f...
A: Given :- CN1-(aq) + HC2H3O2(aq) <----> HCN(aq) + C2H3O21-(aq) To identify :- acid, base, co...
Q: What is the pH of a solution containing 0.57 mol L-1 of a diprotic acid with pKA1 = 5.36 and pKA2 = ...
A: Answer: 2.80
Q: The rate of deposition of copper (mass deposited per unit time) on the cathode depends on: a) The su...
A:
Q: Calculate ∆H for the reaction 4NH3 (g) + 5O2 (g) → 4NO (g) + 6H2O (g), from the following data. 1. N...
A: 4NH3 (g) + 5O2 (g) → 4NO (g) + 6H2O (g) 1. N2 (g) + O2 (g) → 2NO (g) , ∆H = -180.5 kJ 2. N2 (g) + 3H...
Q: A student uses a pipette to dilute 1.00 mL of a standard iron solution to 50.0 mL. If the µg what wo...
A:
Q: Write the cell notation for an electrochemical cell consisting of an anode where Zn (s) is oxidized ...
A:
Q: What gas should we use to separate two compounds with very similar boiling points
A: solution is shown below.
Q: Gaseous methane (CH) reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (CO,) and...
A: Given data : Mass of methane (CH4) = 0.96 g Mass of oxygen gas (O2) = 6.8 g
Q: H2SO4 CH;CHCH2OH CH3 H2SO4 2 CH,CHCHCH,CH3 CH3 Majo CHaCH-CHCH3 +2KOH Br Br
A: ->H2SO4 is a dehydrating agent which form alkene. ->Alco KOH is used for elimination reaction....

Step by step
Solved in 2 steps with 1 images

- Ramachandran plots for two amino acids differ significantly from that shown in Figure 2.23. Which two, and why?What happens to Cl2 how am I suppose to know Ca gets a 2+ and attachs to OH please ecplain how to do this Im confused!.D) Explain your answer. This is the most important part. Please type on an attached page. A complete explanation will require you compare the IMFs of each of these, make a judgement as to the order of the BP’s, and thoroughly explain your answer. The best answers will explain why BP is related to IMF’s. For a thorough answer, you will need to calculate or look up all the molar masses.

