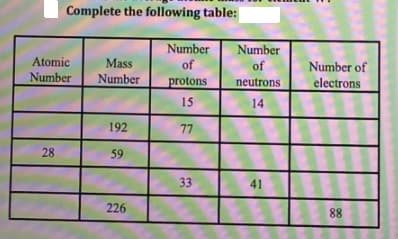
Complete the following table: Number Number Atomic Mass of of Number of Number Number protons neutrons electrons 15 14 192 77 28 59 33 41 226 88
Q: Symbol Protons Neutrons Electrons Mass # Charge 8. 10 8. 8. 18 9. 10 -1 10 10 10 00
A: Atomic number is equal to no. Of protons No. Of protons are equal to no. Of electrons in neutral…
Q: Fill in the blanks. Isotope 30Si4+ has: Number of protons Number of neutrons Number of electrons…
A: Given Symbol of isotope = Si4+30 Number of proton = ? Number of neutron = ? Number of electron…
Q: Nitrogen 11 is an isotope of nitrogen that has 7 protons and 4 neutrons. What are the atomic number…
A: First, we will learn some terms which are asked in the question, The number of protons is always…
Q: 4. Complete the following table. Atomic Number of Element Symbol Mass Number Neutrons Number Охудen…
A: 4. the filling spaces can be seen as: We know that, Mass number = number of proton(Atomic number) +…
Q: Fill in the missing boxes in the chart below: Nuclear Symbol Neutrons Atomic Number Electrons Mass…
A: An atom is the smallest particle that is capable of independent existence. When an atom loses…
Q: Each of the following isotopes is used in medicine. Indicate the number of protons and neutrons in…
A: Number of protons = Z Number of neutrons = A - Z Here, Z is the atomic number and A is the atomic…
Q: Complete the following chart, in order from left to right Mass Number Protons Neutrons Electrons Ion…
A: Correct option is B.
Q: Complete the following table for two of the naturally occurring isotopes of potassium, which is an…
A: Please find the attached images for answer
Q: 102. Copper has two naturally occurring isotopes. Cu-63 has a mass of 62.939 amu and relative…
A: Isotope: Element having the same atomic number but different mass numbers is called isotopes. For…
Q: SUBATOMIC PARTICLES AND ISOTOPES Complete Symbol Atomic Mass Number of Number of Number of Element…
A:
Q: The three stable isotopes of magnesium are Mg2", Mg5 and Mg. The relative atomic mass of magnesium…
A: Given three isotopes of Mg are : a). Mg24 b). Mg25 c). Mg26 Relative atomic mass of Mg = 24.3…
Q: Each of the following isotopes is used in medicine. Indicatethe number of protons and neutrons in…
A: Number of Protons and Neutrons for the following Isotopes are given below:
Q: • Complete the following table: Atomic lon Number of Number of Number of Mass Protons Electrons…
A: NOTE : Since you've posted a question with multiple sub-parts,we'll solve first three sub-parts for…
Q: what do you think the atomic number represents? WHICH OF THIS ANSWER IS CORRECT A)number of…
A: An Atom is made up of 3 sub-atomic particles. 1) Electron 2) Proton 3) Neutron
Q: 37, 37 The actual mass of a 17 atom is 36.9659 amu. Calculate the mass deficiency (amu/atom) for a…
A: Mass defect or deficiency is the difference between the predicted mass and the actual mass of an…
Q: 93. Determine the number of protons and neutrons in each isotope. (a) ㈜Na (b) Ra 2661 88 32 (d) '4N
A: To find: The number of protons and neutrons in each isotope
Q: For each atom give the following information: [1] the atomic number; [2] the mass number; [3] the…
A: General representation of an element : zXA Atomic number = Z Mass number = A Symbol of element = X…
Q: An atom contains 27 protons, 31 neutrons and 25 electrons. The atomic number and mass number for…
A: Atomic number of an atom is equal to the number of protons present in the nucleus or the number of…
Q: # of # of # of Atomic Mass Protons Neutrons Electrons Number Number 32 73 14 14 28 48 64 115 77 59
A:
Q: Complete the table below by filling the blanks with the correct value. Mass Atomic Number Symbol Mo…
A: Number of protons in an atom is equal to the atomic number of atom For a neutral atom, number of…
Q: Isotope symbot: (For the isotope symbol, type in without superscripts. For example, 184w would be…
A:
Q: Element Atomic Symbol Symbol Be 121 Sb 51 Atomic Mass Number Number 8 Protons Neutrons Electrons 51…
A: -> Atomic number = number of proton = number of electron (for element) -> Atomic mass = proton…
Q: How many protons, electrons and neutrons are in 34S2-? Number of protons ______________ Number of…
A: 34S2- Atomic No. = 16 Mass No. = 34 The atomic number specifies the number of protons, positively…
Q: Determine the number of protons and neutrons in 23/11 Na Number of Protons Number of Neutrons…
A:
Q: Consider a hypothetical element, "X". Suppose "X" has two naturally occurring isotopes with the…
A:
Q: 1. Complete the table Isotope Mass (amu) Relative Abundance (%) Silicon- 28 27.97693 92.21 Silicon-…
A: For this problem We have to complete the given table based on abundance of isotopes.
Q: Fill in the table. Element Number of Protons Number of Neutrons Number of Electrons Mass Number…
A: Fill in the blanks--
Q: Fill in the following table: Elemental symbol Atomic Number Mass Number # protons 59 27 # neutrons #…
A: Mass number = Number of protons + Number of neutrons Since an atom is electrically neutral Number of…
Q: (a) 2.8 19. Complete the following table. Number Number Number of Name o the Atom Species Atomic…
A: The atomic number of the species is equal to the number of protons and electrons present in the…
Q: Complete the table Symbol of element Atomic Mass number Number of Number of Number of protons (+) a)…
A: Solution This is a pretty simple question if you have a periodic table handy. Use the Table of…
Q: Atomic Atomic Atomic number Protons Neutrons Electrons symbol mass 102 70 89 225 Мо 53 81 206 100…
A: Atomic symbol Atomic number Protons Neutrons Electrons Atomic mass Yb 70 70 102 70 172 Ac…
Q: Complete the following table: Symbol #of Protons #of Electrons # of Neutrons "He 20N 2 27A
A: 1) The ion given is 4He+. Since the atomic number of He is 2. Hence number of protons = atomic…
Q: Use the element box for potassium to fill in the missing information. 19 K Protons = Type here…
A: Given: Element box of Potassium:
Q: Number of Number of Isotope Symbol Number of protons neutrons electrons 39K+
A:
Q: 1. Nuclear Symbol Protons Neutrons Electrons 107 47A 109 47Ag
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost…
Q: Element Atomic Number Mass Number Number of Protons Number of Neutrons Number of Electrons Ge Si Ni…
A:
Q: Complete the following table. Element Symbol Nuclear Symbol Protons Neutrons Electrons 46 35 Br 79…
A:
Q: Atomic Number Atomic Mass = : # of Protons : # of Protons + # of neutrons :: #of electrons : #of…
A:
Q: Write the complete symbol, including mass number and atomic number, for each atom. contains 28…
A: Any element is represented as Xatomic numbermass number Mass number is given as , mass number…
Q: 18. Complete the following table: Atomic Number of Number of Total Number of Isotope Name Neutrons…
A:
Q: Question attached
A: Symbol 69Ga+3 Atomic number = 31 Number of protons…
Q: See if you can complete the following table using only what is on this page. Element Symbol Nuclear…
A: Answers to all the blanks in the table is given below.
Q: Which of the following represents a pair of isotopes? Atomic Number | Mass Number 1. 6| 14 A II. 7|…
A: The question is based on the concept of atomic structure. We have to identify which of the given…
Q: Number of Inotope Name Owimass number) Total Electric Number of Neutrons Atomic Number of Number…
A: There are different isotopes with different atomic masses but same atomic number - (1) Zinc 66 -…
Q: Number of... Atomic Mass Isotope Name Isotope Symbol Protons Neutrons Electrons Number Number…
A: Atomic number: The atomic number of a chemical element is the number of protons found in the nucleus…
Q: Isotope Name Nuclear symbol Atomic Number Numbers Mass Number of protons Number Number of of…
A:
Q: There are two stable isotopes of nitrogen: Nitrogen-14 and Nitrogen-15. Nitrogen-14 has an atomic…
A: Isotope is defined as the atom of same element having same atomic number but different mass number…
Q: Subatomic Particles Atomic Mass Element Number Number Protons Neutrons Electrons Iron 30 27 13 19…
A:
Step by step
Solved in 4 steps

- Briefly explain the term ‘chemoselectivity’how do I round 0.004198 to 3 sig figsIn a vacuum chamber filled with argon gas at 100mTorr, a plasma is ignited resulting to 1% ionization. This is already a "hot plasma" by lab processing standards. Calculate the number density of plasma electrons (should be about the same as the argon ion density due to quasineutrality) in this 1% ionized plasma.
- In a vacuum chamber filled with argon gas at 100 mTorr, a plasma is ignited resulting to 1% ionization. This is already a "hot plasma" by lab processing standards. Calculate the number density of plasma electrons (should be about the same as the argon ion density due to quasineutrality) in this 1% ionized plasma.What is analogue in medicinal chemistry?Define brieflyPlease calculate the energy of inward movement of chloride ions given: Na+ F=23100 cal/V*mol Vm = -60mV R =2cal/mol*K T = 300K Inside 50mM Outside 150mM A. -2051.2 cal/mol B. 2051.2 cal/mol C. -727cal/mol D. 727cal/mol



