Complete the mechanism and draw the final product for the Grignard reaction below. added acid in workup MgBr: MgBr: Et20 -[MgBr]* H-0-H final product
Complete the mechanism and draw the final product for the Grignard reaction below. added acid in workup MgBr: MgBr: Et20 -[MgBr]* H-0-H final product
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter10: Oxidation And Reduction
Section: Chapter Questions
Problem 7CTQ
Related questions
Question
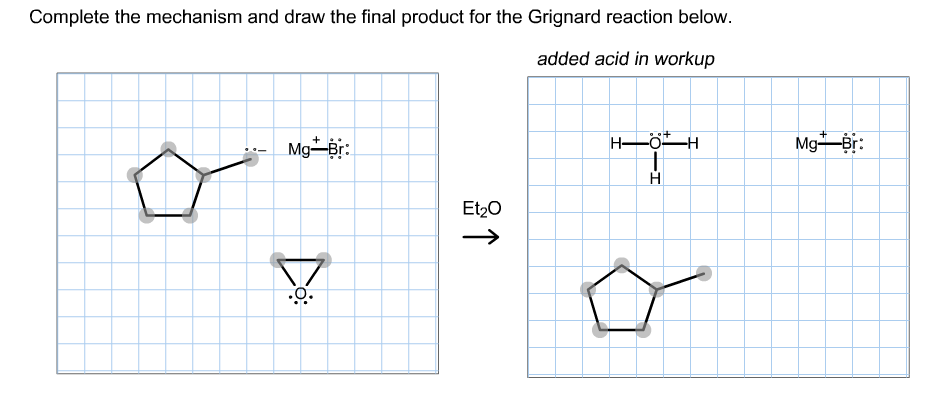
Transcribed Image Text:Complete the mechanism and draw the final product for the Grignard reaction below.
added acid in workup
MgBr:
MgBr:
Et20
![-[MgBr]*
H-0-H
final product](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F324696c5-32e2-4083-9c11-1b27590914e5%2F10de6690-db37-4b27-80a4-0636a8e16d69%2Fwe9wszq.png&w=3840&q=75)
Transcribed Image Text:-[MgBr]*
H-0-H
final product
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning