Q: Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: Which of the following is the product of the reaction shown in Image 17? a. (C6H5)3COH b.C6H5CH2OH…
A: It is based on the concept of reactivity of the Grignard reagent. Here we are required to find the…
Q: A 1.252 g sample of cyclohexanol (C6H12O) was burned in a constant-volume bomb calorimeter where the…
A:
Q: 4. What is the relationship between the following two molecules? H3C- a. They are constitutional…
A: GIVEN the relationship beteen the above given compounds are given below
Q: ▼ Part A CH3 CH3-CH-CH₂-CH₂ CH3 Spell out the full name of the compound. Submit Part B Request…
A: IUPAC nomenclature rules for alkanes: • Identify the longest continuous carbon chain. • Identify the…
Q: Calculate the ratio [C6H5NH2]/[C6H5NH3+] if the solution has the following pH value. (Assume that…
A: It is based on the concept of buffer solution. Here we are required to find the ratio of…
Q: What is the pH of a buffer that results when 5.18 mole of H₂PO4 is mixed with 4.33 mole of NaOH and…
A:
Q: A chemist dissolves 134. mg of pure sodium hydroxide in enough water to make up 170. mL of solution.…
A: Answer: pH tells us about the power of hydrogen ion concentration and its value is equal to the…
Q: 4. List the first ionization energy with units for each element. You may use the table in your…
A: The first ionization energy is the minimum amount of energy required to remove the most loosely…
Q: Question 4 Calculate the frequency of the light emitted by a hydrogen atom during a transition of…
A: Please find your solution below : Planck's equation is an expression used to calculate energy of a…
Q: When 12.0 mL of a 9.90x105 M ammonium sulfide solution is combined with 25.0 mL of a 3.30x10 4 M…
A: Answer: A precipitate begins to form when reaction quotient Q becomes equal to solubility product of…
Q: C₂H6O(1) +O₂(g)→CO₂(g) + H₂O(g) Express your answer as a chemical equation. Identify all of the…
A:
Q: Y Spell out the full name of the compound. Submit Part D Request Answer CH₂-CH CH₂=CH-CH-CH₂-CH-CH3…
A: According to the IUPAC nomenclature, First longest chain of carbon atom is identified. Numbering is…
Q: What is the molecular weight, formula, valence, and species in Calcium Phosphate?
A: For calcium phosphate, we have to determine molecular weight formula valence species present
Q: Explain why the dialysate gave a positive reaction with silver nitrate and a negative reaction with…
A: Dialysate is a medical solution that is used in dialysis to remove excess water, toxins, and waste…
Q: What Al(III) species is the major species in aqueous solution at pH 14.0 ? a) [Al(H2O)6]3+ b)…
A: To solve this problem, I used my knowledge of: The chemistry of aluminum in aqueous solution,…
Q: tep 3: A series of proton transfer steps results in the ydroxyamine. The curved arrows are drawn for…
A: We have to write down the missing curved arrows, intermediate and product for the given set.
Q: Find [CO32-] of a 0.070 M carbonic acid solution. Express your answer using two significant figures.…
A: Given Concentration of carbonic acid = 0.070 M
Q: In the Friedel-Crafts alkylation of benzene, dialkylation is often a significant by-product. In the…
A: Given that, in the Friedel-Crafts alkylation of benzene, dialkylation is often a significant…
Q: Lab: 1) A 5.89 gram sample of vinegar is titrated with 0.100M NaOH. If the initial buret reading is…
A: For question 1: The initial and final burel readings are 0.54 mL and 48.96 mL respectively. The…
Q: Spectrum #2: 3-bromopropene PPM
A: The question is based on the concepts of Organic spectroscopy. we need to assign the peaks to the…
Q: A chemist prepares a solution of potassium chloride (KCl) by measuring out 0.60 g of KCl into a 100.…
A: Given Mass of KCl = 0.60 g Volume = 100 ml
Q: Order these chemical species by increasing pH of an 0.1 M aqueous solution of each. That is, imagine…
A: According to conjugate acid base principle, a strong acid has a weak conjugate base and a weak acid…
Q: Be sure to answer all parts. The reaction of peroxydisulfate S₂08² (aq) +31 (aq) →2SO4 (aq) + 13(aq)…
A:
Q: Which of the choices correctly presents the equilibrium law for the following reaction? 2BrCl(g) Br2…
A: Since,Equilibrium constant or Kp is the ratio of the equilibrium partial pressure of product over…
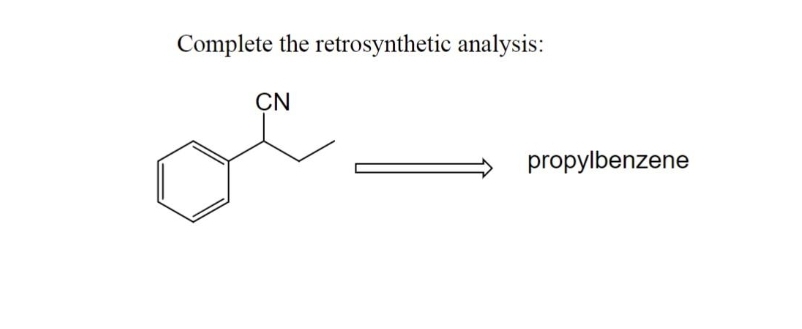
Q: (iv) H₂C-CH₂ O || H 1) Na CN 2) HCI (aq) HCI/H₂O heat
A: Carbonyl compound give nucleophilic addition reaction . Hydrolysis of cyanide group produce carboxyl…
Q: Drawing Question 5 Based on the atomic weight, density and atomic radius for the hypothetical alloy…
A: Given data is for the hypothetical alloy Atomic weight (g/mol) = 43.1 Density (kg/m3 ) = 6400…
Q: Would you classify this reaction as endothermic or exothermic? If you had to describe the flow of…
A: ⇒The Reaction that gives off or release energy is called exothermic reaction. ⇒The reaction which…
Q: Calculate the work done when a gas at a pressure of 2.4 atm and constant temperature expand in…
A:
Q: Be sure to answer all parts 2,2-dimethylcyclopentanol You have available (A) and…
A: Givn that the available compounds to prepare 3,3-dimethylcyclopentene are A :…
Q: Calculate the standard enthalpy of formation for diamond, given that Reaction C(graphite) + O₂(g) →…
A: Given
Q: (e) Calculate the pH of a buffer solution in which [HC₂H₂O₂] = 0.36M and [C₂H₂O₂ 1=0.31M.
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: 14 12 12 11 |12| 1L April 2, 1995 A 1L 1L 1L 1 1 1 1 1 1 December 1, 2000 B What are the quantum…
A:
Q: Calculate the ph of a solution prepared by mixing 20mL of 0.25M h3po4 (ka1 = 7.11 x 10^-3, Ka2 =…
A: For H3PO4: moles H3PO4 = Molarity x Volume in liters = 0.25 M x 0.02 L = 0.005 moles Since H3PO4…
Q: The work done to compress a gas is 69.0 J. As a result, 29.0 J of heat is given off to the…
A:
Q: Hydrofluoric acid and water react to form fluoride anion and hydronium cation, like this: HF (aq) +…
A: Concentration/Molarity = w/(M x V) w = Mass of Solute M = Molar mass of solute V = Volume of…
Q: Given this reaction: Predict which direction the reaction proceeds in: A H₂O + H₂O H3O+ + OH B Goes…
A: The question is based on the concept of chemical equilibrium and acidity. we need to identify the…
Q: ch row of the table below describes an aqueous solution at 25 °C. e second column of the table shows…
A: Given, Based on pH scale, The solutions which have pH greater than 7 ( pH>7 ) are basic…
Q: Calculate DU for the following reaction at 1 atm and 25oC: CH4 (g) + I2…
A:
Q: The reaction N₂O4(g) 2NO₂(g) has a Kp = 0.140 at 25 °C. In a reaction vessel containing the gases in…
A:
Q: (v) O CH3 NaBH4 CH3OH
A: NaBH4 is less reactive but more selective reducing agent than LiAlH4. It selectively reduces some…
Q: Excess sodium is reacted with 4.00 L of chlorine gas that has a density of 1.88 g to form sodium…
A:
Q: Indicate the products of the reaction C6H5-CO-CH3 + CH3-COOOH (peroxyetanoic acid)
A:
Q: Examine the AH values given for a series of gaseous reactions that are listed below. Reaction 1:…
A: Examine the ΔH values given for a series of gaseous reactions that are listed below.Reaction 1:…
Q: 1. Draw the structure and interaction that occurs between the following R groups of amino acids in…
A: a)The interaction that can occur between the R groups of asparagine and serine is a hydrogen bond.…
Q: se 7.10 Write a chemical equation for solid cadmium hydrogen carbonate decomposing to yield solid…
A: Balanced chemical equation: Balanced chemical equation can be define as the reaction in which number…
Q: Identify the alkene obtained on dehydration of 3-ethyl-3-pentanol.
A: Here, we have to find the product alkene obtained on the dehydration of 3-ethyl-3-pentanol.
Q: Part B Zn + O₂ → Express your answer as a balanced chemical equation. 0 ΑΣΦ ***** ? A chemical…
A:
Q: 4. All of the following word equations represent single displacement reactions. Complete the word…
A:
Q: Combustion of hydrocarbons such as hexane (C6H₁4) produces carbon dioxide, a "greenhouse gas."…
A:
Hh.91.
Step by step
Solved in 2 steps with 2 images

- Does it safe to use this sunscreen K on acne prone skin. And can we rinse off it by only using facial gel mixed with water to rinse off it without use oil. Do I need to use micellar water for removing cosmetic before rinse off it with facial gel Sunscreen K has lists of ingredients below (descending order)…. AQUA DIMETHICONE CYCLOPENTASILOXANE PROPYLENE GLYCOL NIACINAMIDE DIETHYLHEXYL CARBONATE ZINC OXIDE CETEARYL ALCOHOL POLYMETHYL METHACRYLATE SODIUM POLYACRYLOYL-DIMETHYL TAURATE TITANIUM DIOXIDE ETHYLHEXYL METHOXYCINNAMATE BUTYL METHOXYDIBENZOYLMETHANE GLYCERYL STEARATE BIS-PEG/PPG-20/5 PEG/PPG-20/5 DIMETHICONE METHOXY PEG/PPG-25/4 DIMETHICONE HYDROGENATED POLYDECENE BUTYLENE GLYCOL OCTOCRYLENE PEG-40 STEARATE PHENOXYETHANOL HEXYL LAURATE POLYHYDROXYSTEARIC ACID CAPRYLIC/CAPRIC TRIGLYCERIDE STEARIC ACID CETYL ALCOHOL PEG-10 DIMETHICONE CETEARETH-20 TOCOPHERYL ACETATE STEARYL ALCOHOl PHENYL TRIMETHICONE TRIETHOXYCAPRYLYSILANE TRIDECETH-10 CHLORPHENESIN DISODIUM EDTA DIMETHICONOL…give at least 3 confirmatory/identification test for sulfanilamide and the chemical reactions involvedGiven that your mixture containing benzoic acid and aminobenzene dissolved in n-hexane solvent. Suggest a method on how to separate these two substances. Available reagents: A. Hydrochloric acid B. Sodium hydroxide C. Distilled water D. n-hexane
- Explain this difference in potency and speed of onset by pointing out the main differences in functional groups between morphine and heroin.1. outline a procedure for separating a mixture of naphthalene (a neutral compound), toluic acid (an acidic compound), and p-bromoaniline (a basic compound). Assume that toluic acid and p-bromoaniline are completely insoluble in water. 2. The solubility of Lorazepam (a tranquilizer found in Emotival , Lorax, Wypax, and other prescription medicines) is 0.08mg/mL and 2.62mg/mL, in water and chloroform, respectively. Estimate the partition coefficient of lorazepam between chloroform and water. 3. The solubility of benzoic acid at 25 degrees Celsius in water and diethyl ether is 3.35 mg/mL and 0.353 g/mL, respectively. Estimate the percent recovery of benzoic acid in the organic phase, if a solution of benzoic acid in water is extracted with an equivalent volume of ether. [HINT-pay attention to the units] 4. The solubilities of cholesterol in water and chloroform are 0.550mg/100mL and 1.00g/5.25mL, respectively. Estimate the partition coefficient of cholesterol between chloroform and…a. Give at least three characteristics of dichloromethane that makes it a good extracting solvent for the alkaloid.b. Why is it necessary to remove a stopper from a separatory funnel when the liquid is being drained from it through a stopcock?c. What are emulsions? Why do they form during extractions? How is the formation of an emulsion minimized? How are emulsions removed?
- Predict the structures and provide an alternate reagent as a substitute foracetic anhydride.In detail, explain how to separate a mixture of naphthalene, byphenyl-4-carboxylic acid, and 4-chloroaniline.Give at least three characteristics of chloroform that make it a good extracting solvent for the alkaloid.
- Briefly describe the process of Fischer esterification of methyl salicylate.(a) Write the structures of main products when aniline reacts with the following reagents :(i) Br2 water (ii) HCI (iii) (CH3CO)2O/pyridine(b) Arrange the following in the increasing order of their boiling point :C2H5NH2, C2H5OH, (CH3)3N(c) Give a simple chemical test to distinguish between the following pair of compounds : (CH3)2NH and (CH3)3NWhat is the predominant form of cyclohexylamine at pH 9.5? Approximately what fraction is in this form?

