Q: A2 1. An alkyne with molecular formula C5H10 2. A ketone with molecular formula C4H8O 3. A ketone…
A: these are bond line structures . carbon atom present at end point and joints.
Q: Br #1. HgC CH3 Br name CI #a. 1-bromo-1-chloro -1-fluoro-a- methyl butane diagram
A:
Q: Draw all the alkyl halides with molecular formula C 5H 11Cl formed when pentane (CH 3CH 2CH 2CH 2CH…
A: The monohalogen derivative of alkanes are usually reffered to as haloalkane or alkyl halide. They…
Q: w Name the following compounds (IUPAC) но OH HO SH HO HO - CH, H,C
A: IUPAC nomenclature is helpful in giving names to organic molecules according to rules. These are…
Q: Name: Condensed Molecular Structural Formula Formula Sketch Ethers Dimethyl ether CH,-O-CH, C,H,O…
A: Ethers are the class of compounds having general formula ROR where R is an alkyl group. The sketch…
Q: Identify the functional groups in leukotriene C4, a major contributor to the inflammation associated…
A: The functional group is the group of atoms which act as a single unit and it exhibits its own…
Q: MCQ 20: Useful alkenes for chemical industries is supplied by A. oil refineries B. coal mines C. oil…
A: In oil refineries the gases which are produced are oxides of nitrogen,carbon monoxide,hydrogen…
Q: MCQ 5: Bromine water does not react with benzene at A. high temperature B. low temperature C. room…
A: The question is based on the concept of organic reactions. we are talking about the reaction of…
Q: Compound X has the formula C30H50BrCIO2. (There are no triple bonds in X.) It reacts with excess H2…
A: Double Bond Equivalent (DBE). One DBE = one ring or one double bond. DBE can be calculated using the…
Q: What is the molecular formula of each drug?
A: a) CHLOROQUINE drug used for antimalarial medication MOLECULAR FORMULA: C18H26ClN3 MOLAR MASS: 319:…
Q: show the major products
A: Alcohols react with tosylchloride in the presence of pyridine to form tosylates. Tosylates undergo…
Q: pls predict the products H30* CCH Br CH;SO Acetone
A: The reactions given are,
Q: 13 Cl₂CH-CH₂ 1,1-dichloroethane C₂H₂Cl₂
A: IR spectra deals results from the interaction of infrared radiation with matter by absorption. IR…
Q: 3. Which structural formula below represents an unsaturated hydrocarbon? Explain. H H H H H-C-H…
A:
Q: 1. CH₂-CH₂-CH₂-CH₂-CH₂ + H₂O 2. H₂C-CH=CH₂ + HBr/H₂O₂ 3. H,C-CH (OH)-CH, +H* 4. Nitrobenzene +? ?…
A:
Q: b. p-TSOH (CH3)2NH Benzene C. H. 1. (C6H5)3P H. Br 2. C6HSLI 3.
A: The reaction is like stork enamine reaction. Here in first step the acid protonates the carbonyl…
Q: 1.1 Provide IUPAC names of the following aromatic compounds: QU. NO₂ NO₂ CH3 Br NH₂ OCH3 f) COOH OH
A: IUPAC nomenclature is used for naming the organic compound as recommended by international union of…
Q: No Spacir NH2 g- NO2 h. NO NO OH Br NO
A: (g) The parent structure is benzene ring. The functional groups present are methyl and amine…
Q: 1. Give the IUPA name for the following compounds: H,C-C-C=C-CH3 Но- a. OH b. С.
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: C5H10O has both aldehyde and ketone isomers. Draw and name all isomers that are aldehydes, and then…
A: Answer;- This question is answered by using the simple concept of writing the isomers which have…
Q: of a) d) MCPBA NaHB4, ethanol H,0* b) CH,CH,MgBr, ether H,O* e) PPH3 c)
A:
Q: OH он toluene H OH 130 °C OH HO, ÓH Ph "I |
A:
Q: 56) Which of the following compounds is not aromatic? N: I. N: II. N I. N IV. A) I B) II C) III D)…
A: From the given compounds, which is not aromatic has to be given.
Q: Which molecule(s) contain(s) ketone functional group? a. CH3 OH H. 0=C осн OH CH3 C. CH3 OH O none…
A: Anwer of the question given below,
Q: Which of the following is most likely not a gas at room temperature? A) O2 B) He C) H2 D)…
A: Given Species 1. O2 2. He 3. H2…
Q: NH2 C1 27. What is the correct IUPAC name for the compound above? a. 2-chloroaniline b.…
A: Answer:- This question is answered by using the simple concept of naming of organic compound using…
Q: Draw the structure of the product expected in the following Sonogashira reactions:
A: Note: As per the Q&A guidelines, I am answering only the first three subparts of a question. So…
Q: please indicate their IUPAC names. а. b. C. d. f. g. h. е. NH2 OCH3 i. j. H3CO k. I. m.t n. o. р.
A: According to the rules of IUPAC nomenclature 1. Select the longest carbon chain as a parent chain.…
Q: MCQ 145: Ethane C,H„belongs to A. alkane B. alkene C. alkynes D. aromatic hydrocarbons
A:
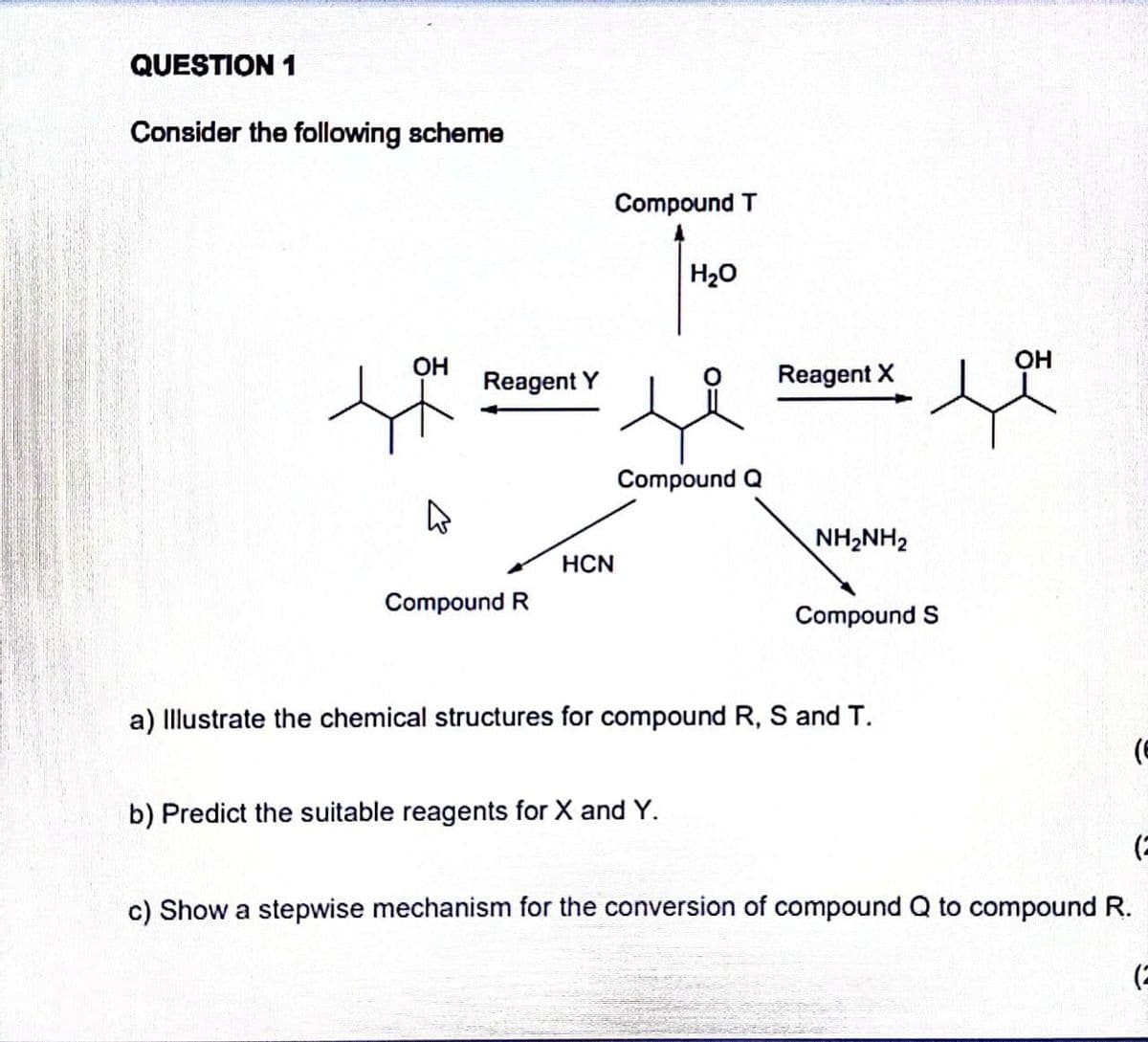
Q: Compound E PBr3 Compound A H₂SO4 Heat Compound B (Major) KMnO₂/ H* Heat i 2-butanone OH + HCN…
A: All the reactions have been completed in the following step.
Q: What is the formal charge on the aldehyde carbon of acetaldehyde? H O H-CC A. 0 b) 1 c) 2 d) 3 e) 4…
A: Formal charge of an atom is calculated by
Q: Formaldehyde (H,CO) :0: I
A: Intermolecular forces of attraction in formaldehyde molecules:
Q: Q13: Write the name of this compound and recall how each of the three can be introduced to benzene:…
A: Answer of the question given below,
Q: What are the products of each aromatic substitution reaction? H FeBr, H, H + Br2 a. H. H CH, AICI,…
A: Aromatic substitution It is the substitution reaction in which hydrogen on an aromatic ring is…
Q: dentify the type of functional group that is most likely give a molecular ion having m/z = an odd…
A: Compound having odd number of nitrogen atoms generally give molecular ion peaks at odd number in the…
Q: Q 31: Haloalkanes react with water in silver nitrate solution A. alcohol B. yeast C. ester D. ketone
A: The question is based on concept of nucleophilic substitution reactions. we have to determine the…
Q: Compound X has the formula C30H50BrCIO2. (There are no triple bonds in X.) It reacts with excess H2…
A: The answer is as follows:
Q: Identify the products or reactants. Draw the structures. a. isobutyl bromide + Mg/ether -> A b. A +…
A: Alkyl halide reacts with Mg forms Grignard reagent (organometallic catalyst). In which alkyl acting…
Q: 56) Which of the following compounds is not aromatic? N: I. N: I. N II. N IV. || A) I B) II C) III…
A: Aromaticity is the property of a molecule or compound in which the delocaization of the pi-bonds or…
Q: Ch,cl2 C2h5ONa HN, indole
A: Ethoxy is the strong base so it will bastract acidic H+ from indole.
Q: LOH H2N + any compound of <2 carbons
A:
Q: among th 1. Which of the following compounds is a functional group isomer of C2HSOH, ethanol (ethyl…
A: The term isomer comes from two words, 'iso' means same and 'mer' means parts. The compounds that…
Q: n compounds are aromatic? (A) (B) (C) (D) (E) (F) B. (G) (H) (1) H.
A:
Q: Which steps/ reagents are required to convert structure A to structure B? Please specify each step.…
A: Here we have to convert the compound i.e structure-A into structure-B by multistep reactions.
Q: ÇH3 CH3 CH3 H3CCH2 1. limonene 2. muscone 3. nicotine H. H3C. CH3 CH3 CH3 H3C. O-CH3 „CH3 H-O 6.…
A: A) Double bonds except aromaticity.
Q: Select one: а. (CH2OH)2 o b. CH4 с. СН3ОН
A: Which of the given species have the highest viscosity
Q: ch compound: (a) CH2(COOB
A: To draw the product formed in the crossed aldol reaction of phenylacetaldehyde (C6H5CH2CHO) with…
Q: Thionyl choride, SOCl2, is a reagent commonly used to convert the –OH group of an alcohol to a…
A: Here we have determine the formal charge present on the S atom in the following Lewis structure of…
Q: Acyl chloride H20 RCOO™NA* ROH RNH2 3 4
A:
Q: SKO024 ANALYTICAL & ORGANIC CHEMISTRY TUTORIAL 9 HALOALKANES 1. Name the following compounds: a. b.…
A: Iupac naming depand main functional group and halogen atom always behave as a substitute group
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- a) Write out the first 3 steps only(to the tetrahedral intermediate shown) in the 6-step arrow pushing mechanism showing how ethyl propanoate is hydrolyzed in acid to form propanoic acidand ethanol. b) NaOH/H2O also serveto hydrolyze an ester; briefly explain why NaOH/H2Ois generally preferable.When (CH3CH2)3CBr is added to CH3OH at room temperature, the product is(CH3O)C(CH2CH3)3. Propose a mechanism(s) for the reactions leading to theseproducts and use curved arrows to show the movement of electrons.Oxidation of cholesterol converts the alcohol to a ketone. Under acidic or basic oxidationconditions, the C“C double bond migrates to the more stable, conjugated position. BeforeIR and NMR spectroscopy, chemists watched the UV spectrum of the reaction mixture tofollow the oxidation. Describe how the UV spectrum of the conjugated product, cholest-4-en-3-one, differs from that of cholesterol
- Predict the expected product for each reaction and provide IUPAC name for the correct starting material to yield the desired epoxide: 1. Name of compound 2. Answer A, B, C, or DDeduce the structure of each compound from the information given. All unknowns in this problem have molecularformula C8H12.(a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC¬(CH2)6¬COOH. Draw the structure of Wa.) Give an acceptable name for compound C. (b) Draw the organicproducts formed when C is treated with each reagent: [1] H3O+; [2] −OH,H2O; [3] CH3CH2CH2MgBr (excess), then H2O; [4] LiAlH4, then H2O.
- Answer the following: (i) Haloalkanes easily dissolve in organic solvents, why? .. (ii) What is known as a racemic mixture ? Give an example. (iii) Of the two bromoderivatives, C6H5CH(CH3)Br and C6H5CH(C6H5)Br which one is more reactive in SN1 substitution reaction and why?Identify the reagent e, f, g and hPlease help with F-M, the questions Complete the following reactions which form ethers (A and B) and cyclic ethers (C-E) as major products
- Q2. What are the products of the following reactions? Q3. 2-Bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C. Identify A, B and C. Which is predominant? Compound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr), which reacts violently with D2O togive 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (CH3COCH3)followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2SO4 gives E (C10H16), which decolorizestwo equivalents of Br2 to give F (C10H16Br4). E undergoes hydrogenation with excess H2 and a Pt catalyst to giveisobutylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout3.provide the starting materials needed to synthesize each compound.

