Q: Step by step please. also have to answer A-D
A: For the reaction., MnO4-(aq) + 5 Fe2+(aq) + 8 H+(aq)---> Mn2+(aq) + 5 Fe3+(aq) + 4 H2O (l)
Q: HO H,O OH + [acid or base] OH Acetylsalicylic Acid (aspirin) Salicylic Acid CH,OH ,A H,SO, (conc.)] ...
A: .
Q: HF is a weak acid and NH3 is a weak base. Which of the following shows the correct order of increasi...
A: Sodium chloride is formed from the reaction of a strong acid and a strong base. In the formation of ...
Q: What formula do you use to answer the question, Hydrogen and oxygen undergo a chemical reaction to f...
A:
Q: Give a reaction equation you could use to prepare an alkylamine from 2-phenyl-pentanoylazide. Show t...
A:
Q: (CH,(CH.),CH;) CH; reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (Co,) and g...
A: We first need to determine the limiting reagent. After determining the limiting reagent, we can calc...
Q: A sample of solid NH4NO3 was placed in an empty container. It decomposes according to the following ...
A:
Q: Although there are more electrons available to create bonds in F2 than in 02, the bond dissociation ...
A: In O2 according to MO theory highest occupied orbital is π*2px= π*2py anti bonding orbitals. 6 elect...
Q: Please answer all questions below. 1. Write the four least soluble anions. 2. If two soluble sal...
A: Anions having larger size and less size ( small charge density) are less soluble in aqueous solutio...
Q: Which of the following describes the final major product of the reaction of 2-propanone with excess ...
A: Carbonyl compound (aldehyde and ketone) reacts with an alcohol to form hemiacetal in the presence of...
Q: 1. Rank each set of three alcohols in terms of increasing acidity, "1" being least acidic, and "3" b...
A: - Inductive effect increases the acidic strength of the compound. More is the number of substituent...
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A: The given data is as follows: The mass of sodium bromide produced = 1.68 g The mass of hydrobromic a...
Q: A mixture of helium and hydrogen gas is expanded from a volume of 89.0 L to a volume of 90.0 L, whil...
A:
Q: e enthalpy change (AH) of a reaction is the transition state. Directly proportional to Independent o...
A: Enthalpy change of a reaction is the difference between the enthalpy of the product and the enthalpy...
Q: A practitioner will make a titration curve. The practitioner pipetted 10 mL of 0.1 M HCl solution an...
A: "Since you have asked multiple questions, we will solve the first question for you i.e (HCl vs NaOH...
Q: If K, for HCN is 6.2 x 10-10, what is K, for CN-? O 6.2 x 104 O none of these O 6.2 x 10-24 O 1.6 x ...
A: Given that, For the acid HCN , Ka = 6.2×10-10 We need to find Kb for its conjugate base CN-
Q: A reaction that requires less energy to reach the transition state will be a reaction. (A) (B) Faste...
A: Answer Reaction that requires less energy to reach the transition st...
Q: The formation of nitrogen monoxide from its elements has an equilibrium which favors reactants: N2 (...
A:
Q: Gaseous methane (CH,) reacts with gaseous oxygen gas (o,) to produce gaseous carbon dioxide (co.) an...
A:
Q: Explain why the following reaction is endothermic based on your knowledge of both bond enthalpy and ...
A:
Q: A chemist measures the amount of nitrogen gas produced during an experiment. She finds that 34.5 g o...
A:
Q: exothermic Is the reaction exothermic or endothermic? x10 endothermic up Does the temperature of the...
A:
Q: Essential oils are concentrated liquid containing metabolites extracted from plant sources and are w...
A: Given: Compound A molecular weight = 136.228 g/mol Compound B molecular weight = 168.270 g/mol Compo...
Q: Calculate the work, in joules, done by a gas as it expands at constant temperature from a volume of ...
A:
Q: Fou m Costants 12 C he llown Sreactions and correponding equilibenum constants Reaction A K-1) React...
A:
Q: 10) Calculate the moles of magnesium ( Show your calculation
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: When the following skeletal equation is balanced under basic conditions, what are the coefficients o...
A: Write the balanced redox reaction under basic conditions---
Q: A solution that has a hydrogen ion concentration equal to 9.61 x 10-7 has a pH =
A:
Q: mol
A:
Q: Wine goes bad soon after opening because the ethanol (CH,CH,OH) in it reacts with oxygen gas (0,) fr...
A:
Q: For a process, 153 J of heat was released by the system and 267 Jof work was done on the system. Cal...
A: Given, Work done on the system = 267 J Heat realised by the system = 153 J
Q: PT#15 3.) Directions: Calculate the empirical and molecular formula of the given. Question: A com...
A: Molar mass of Carbon = 12.011 Moles of carbon = 26.5/12.011=2.21mol Molar mass of H=1.00784 Moles of...
Q: For the following two compounds, the given names do not correctly follow IUPAC naming conventions. F...
A:
Q: Write T if the statement is true and F it is false.
A: 5) Typically, if we have pure product from a reaction, percent yields are understandably less than ...
Q: what is 100g/L when converted to mg/mL?
A:
Q: 1. A 55.0 ml sample of 0.102 M potassium sulfate is mixed with excess lead (II) acetate to produce p...
A: Potassium sulfate reacts with lead(II) acetate to form potassium acetate and lead(II) sulfate. The e...
Q: 1.0 x 10 11 moles of HCl are added to 1.0 L of H2O. What is the pH? O 10.0 O 11.0 3.0 o 7.0
A: Moles of HCl = 1.0 × 10^-11 mole Volume of H2O = 1.0 L
Q: Use the data given in the above question to first determine the rate law before answering the second...
A:
Q: ACTICE PROBLEMS "Atomic and Bonding Theories 1. Write the ground state electron configuration of eac...
A: The distribution of electron within different orbital or molecular orbital of a given element or mol...
Q: This question is based on the five compounds (A-E) given below. CH;-CH=CH-CH3 (B) (C) (A) CHCI=CHBR ...
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub- parts for ...
Q: how would you properly balance this reaction?
A: Sodium borohydride, NaBH4, is a reducing agent. It reduces aldehydes to primary alcohols and ketones...
Q: Q3] Explain formation of covalent bonds in the following compounds (using three theories) SO4-2, HF,...
A: Explain the formation of covalent bonds in the following compounds. SO4-2, HF, CCl4, C2Br2H2, NO, H2...
Q: Directions: Balance the chemical equation below and complete its mole ratio on the table below. C3H8...
A: Balanced equation: It can be defined as the chemical equation where the number of each atom is the s...
Q: You were asked to determine the acid content of vinegar and soda samples. You performed a simple aci...
A: To determine the acid content of vinegar and soda, a simple acid-base titration is performed. Aver...
Q: Balance the chemical equation below using the smallest possible whole number stoichiometric coeffici...
A: Given, Chemical equation : H2(g) + P4(s) ---------> PH3(g)
Q: Moarity How many gramo - use with uolume values to calculate mass I of solute are contained in each ...
A:
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A:
Q: What mass (in grams) of sodium acetate (MM=0.82 g/mol) is needed to prepare 500 ml of 75 mM acetate ...
A:
Q: 1. what is the solubility of ascorbic acid in water? (include references) 2. How much water is need...
A: Ascorbic acid is also known as hexuronic acid. It exists in D and L stereoisomers. It is one of the ...
Q: When the following skeletal equation is balanced under acidic conditions, what are the coefficients ...
A:
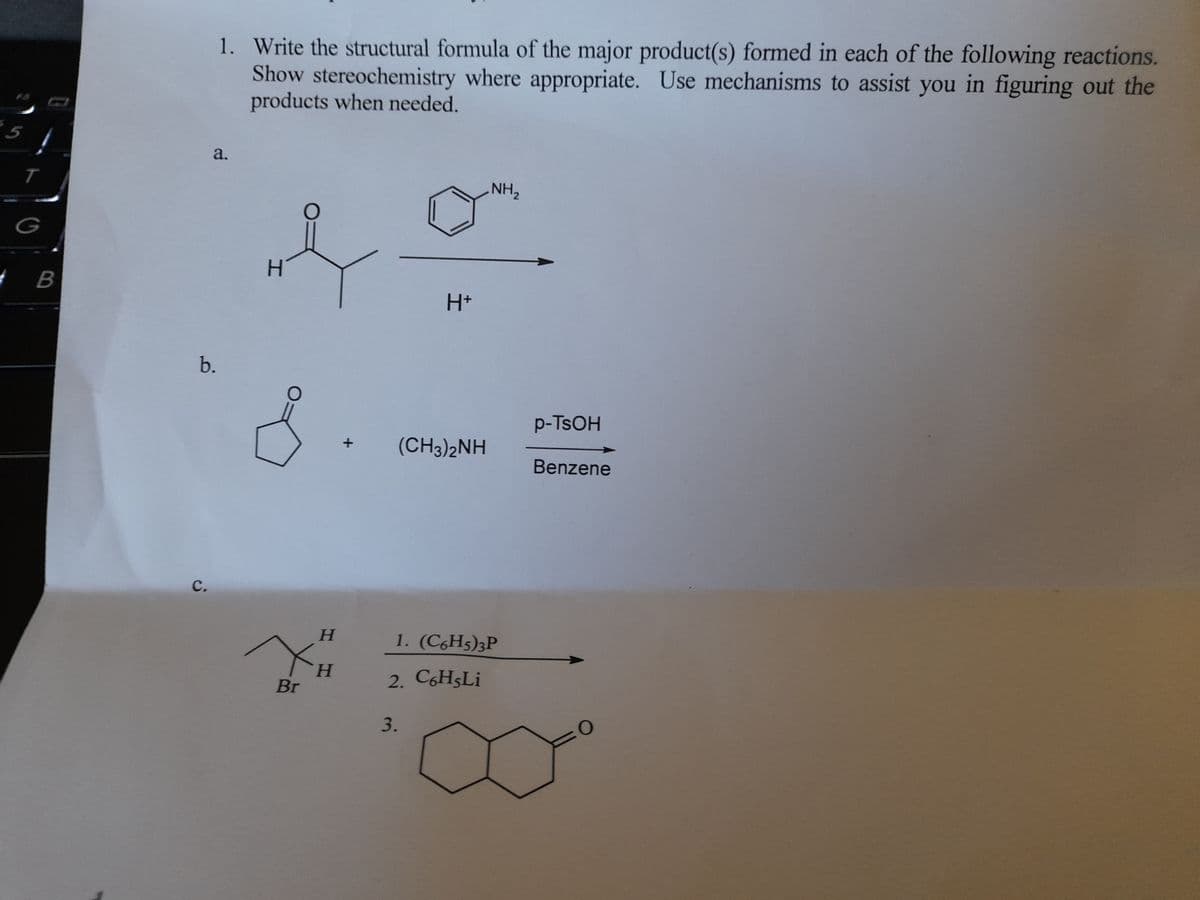
Just B and C pleases
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- (a) (i) 3-Methyl-2-butanol will react with sulphuric acid to give two isomeric alkenes in 3:1 proportions.(i) Write down the structures for these alkenes and assign appropriate systematic names to them. (ii) Name the most abundant isomer. (iii) Write down detailed mechanism for the formation of a minor product.What are the final products for these reactions? *mechanisms not necessaryThe synthesis above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order. Be sure to consider stereochemistry and regiochemistry.A. NaOHB. mCPBAC. OsO4, tBuOOHD. NaHE. H2, LindlarF. CH3CH2CH2BrG. CH3CH2ClH. NaNH2I. H3O+J. Na, NH3(l)
- CH₂ + Using the reagents below, list in order (by letter, no period) those necessary to prepare 2-butyne from acetylene. Note: Not all spaces provided may be needed. Type "na" in any space where you have no reagent. a. Br₂, heat, light b. Na, (-H₂) c. Nal(SN2) d. NH4NO3 e. 1) NaNH2, liq. NH3, 2) CH3Br f. (CH3)3COK, (CH3)3COH, heat g. NaOEt, EtOH, heat Step #1 Step #2 Step #3Show stereo chemistry and possible outcomes and the steps for the reaction.Give the reagents and reaction conditions for the conversion of (i), (ii), (iii), (iv) and (v) CH3CH2Br ➡ A ➡ CH3COOH (i) (ii) A ➡ CH3CHO ➡ B ➡ CH3CH(OH)COOH (iii) (iv) (v) Hence name the structures A & B. What type of reaction is step (i), (ii), (iii), & (iv). Give the mechanism of the conversion of CH3CHO to B.
- What would the final products look like? Pls specify stereochemistry if neededWhat will be the major product obtained from the reaction of Br2 with 1-butene if the reaction is carriedout ina. dichloromethane? b. water? c. ethyl alcohol? d. methyl alcohol?Complete the following reactions with the majority products and the missing reaction conditions. Indicate stereochemistry if applicable. Also indicate the type of reaction that took place (SN1, SN2, E1, E2, E1bc)
- What is the organic chemistry mechanisms to get to this final product? one reagant is NaOCH2CH3Hydration of 1,3-dimethylcyclopentene gives what major and what minor products?Give the reagents and reaction conditions for the conversion of (i), (ii), (iii), (iv) and (v) CH3CH2Br ➡ A ➡ CH3COOH (i) (ii) A ➡ CH3CHO ➡ B ➡ CH3CH(OH)COOH (iii) (iv) (v) Hence name the structures A & B.

