Concentration 3006 2901 2892 Absorbance std 0.540 0.540 0.540 Absorbance sx 0.555 0.526 0.540 100.3 mg X2 Sample weight Specs 95 - 105% Concentration = 0.0403 mg/mL Dilution factor = 2500 mL Formula: % potency = (Abs sx / Abs std)(Conc)(Dilution factor) x 100 100.6 mg 100.1 mg X1 X3 Weight of sx
Concentration 3006 2901 2892 Absorbance std 0.540 0.540 0.540 Absorbance sx 0.555 0.526 0.540 100.3 mg X2 Sample weight Specs 95 - 105% Concentration = 0.0403 mg/mL Dilution factor = 2500 mL Formula: % potency = (Abs sx / Abs std)(Conc)(Dilution factor) x 100 100.6 mg 100.1 mg X1 X3 Weight of sx
Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
Using the data and formula given below, compute for the potency of Lot No. 3006, 2901 & 2892:
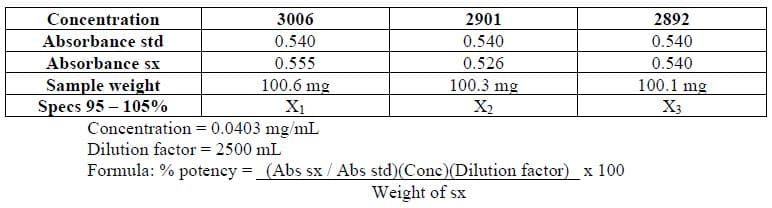
Transcribed Image Text:Concentration
3006
2901
2892
Absorbance std
0.540
0.540
0.540
Absorbance sx
Sample weight
Specs 95 - 105%
Concentration = 0.0403 mg/mL
Dilution factor = 2500 mL
0.555
0.526
0.540
100.6 mg
X1
100.3 mg
100.1 mg
X)
X3
Formula: % potency = (Abs sx / Abs std)(Conc)(Dilution factor) x 100
Weight of sx
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

