Q: Give detailed Solution with explanation needed..please explain
A: Chemical properties of alkenes : Alkenes are having pi electron excess species Alkenes itself…
Q: Reduction of aldehydes and ketones by sodium borchydride or hydrogen forms all O water O ester O…
A:
Q: A 25.0 cm³ sample of 0.620 mol dm-³ nitric acid was placed in a beaker and 38.2 cm³ of 0.550 mol…
A: The number of moles (n) of a solute present in V cm3 of C mol dm-3 solution is calculated using the…
Q: Propose an efficient synthesis for the following transformation: th The transformation above can be…
A: Peracids oxidises the aldehydes to acids and ketones to ester by the migration of H and Alkyl group…
Q: The answer is not complete
A: 10.00 mL of 0.15 M Na2HAsO4 solution is added to 25.00 mL of 0.10 M NaH2AsO4 solution.We have to…
Q: Determine [OH], [H], and the pH of each of the following solutions. a. 1.2 M CsCl [OH-] [H+] pH = b.…
A:
Q: elp me answer this question: Q: Using your own data, is it possible to evaluate the ideas of the…
A:
Q: Name the following heterocyclic ethers. (a) (d) H CH₂CH₂ O (b) CH₂CH3 (e) H Br CI OCH₂CH3 (c) "O (f)…
A: The names of the above heterocyclic ethers molecules are based on the their parent heterocyclic…
Q: The reaction HCO3 CO2+ H is an important part of the carbonate system, and is governed by the…
A: (a). pH of a solution can be defined as the negative logarithm of the value of the molar…
Q: Are these two molecules... functional isomers? Positional isoners? Enantiomers? Diastereomers?…
A: The compounds with the same molecular formula but different bond connectivity are structural…
Q: Product Spinach Cabbage Kale Initial Dilution 10-1 10-1 none Plate Counted 10-2 10-2 10-1 Volume…
A: For Spinach, The colony-forming unit (CFU) per milliliter (ml) can be calculated as follows:…
Q: [References] Use the References to access important values if needed for this question. When HBr(g)…
A: We can solve this question using the stoichiometry of the reaction. As given in the question , for…
Q: A reaction profile (not to scale!) for the reaction 2 HI H₂ + I2 is shown below: E (kJ) HIA HI A 184…
A:
Q: Mathematically according to the Gibbs free energy equation, what are 3 ways a reaction could…
A: To solve this problem we have to use the Gibbs free energy equation . This equation is given as ∆G…
Q: 0- 1 0-P- O-CH₂ 0 04 -NH₂ H 09-0-962 6- HK Noble 0=P-O-CH₂ # 1) Haw would you designate this acid in…
A:
Q: 4. What is the value of K, (to two decimal places) for the following reaction at 300°C? CH₂OH(g) =…
A:
Q: 1. Use curved arrows to show electron movement in the reactant side and draw the product/s of the…
A:
Q: COH + Drawing - ≡ N ⓒ Di
A: Given a reaction of phenol with benzene diazonium dalt. We have to draw the product. ★ Benzene…
Q: Determine the mass of chlorine in the original sample as the difference in mass between the…
A:
Q: Provide the structures of the major organic products of the following reaction. Br NaNH, NH3…
A:
Q: Use the IUPAC Nomenclature System to name the following ester:
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: (b) HDAB The transformation above can be performed with some reagent or combination of the reagents…
A: Given that, a reaction scheme is shown below We have to carry out the above transformation.…
Q: Propofol (Diprivan) is a very common short-term anesthetic and sedative, and its structure is shown…
A: The mass spectra of a compound helps to find the chemical structure of the compound by the different…
Q: H I. H H H H } H 1 F HIN H. H- H H H H 3
A: NH3 can be act as Lewis base can accept proton. It also can act as nucleophile and carryout…
Q: 5. If 3.00 mL of water at 40.0 °C was added to a calorimeter containing 2.00 ml. of water at 20.0 °C…
A: To calculate the amount of heat gained by the calorimeter, we can use the equation: q = mcΔT where q…
Q: A 1.361.36 L buffer solution consists of 0.1310.131 M butanoic acid, C3H7COOH and 0.3150.315 M…
A: 1.36 L buffer solution consists of 0.131 M butanoic acid, C3H7COOH and 0.315 M sodium butanoate,…
Q: Obtain the rate law, including the rate constant 1) Exp. 1 Exp. 2 Exp. 3 Initial Concentrations A…
A:
Q: Use the precipitation interactive to answer the questions. Select the color of the solid that forms…
A: Given : Solution 1 : Pb(NO3)2 Solution 2 : K2CO3 When Pb(NO3)2 solution added to K2CO3 Solution…
Q: 20 15 33.5 1004 1.025 1.0045 34.0 1.0255 A 1.0275 34.5 1.026 1.0265 1.027 1.028 35.0 Falinitu (DOT)…
A: Solutions- From the above graph we find the values
Q: need help with number 3. I need help with the balanced chemical equation for part g. How to Balance…
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: At a certain temperature the equilibrium constant, Kc, equals 0.11 for the reaction: 2 ICl(g) I2(g)…
A:
Q: The following substances are pharmaceutical drugs. The pka values provided correspond to the acid…
A: The given reaction belongs to the Dolobid. Dolobid (C13H8F2O3) is the weak acid. Its conjugate…
Q: This is the TLC of syn-azobenzene (A for both plates since it was under UV light) and…
A: It is unusual for a more polar compound to have a higher Rf value and travel further than a less…
Q: At a certain temperature the equilibrium constant, Kc, equals 0.47 for the reaction: 2 ICl(g) ⇌…
A:
Q: A student wants to dry a solution of aniline in hexane. Which drying agent should he use for the…
A: To identify a drying agent to dry a solution of aniline in hexane.
Q: uestion 11 Which of the following is the correctly balanced equation for the complete neutraction of…
A: Note: Since you have posted multiple questions, we will provide the solution only to the first…
Q: What is the buffer range for the buffer made from mixing 70.0 mL of 0.15 M HCIO with 25.0 mL 0.20 M…
A: The effective range of the buffer can be calculated by using the pka values- Effective buffer range…
Q: 10) Calculate the following quantities: Vb Ma a) Volume (in ml) of 0.75 M HCl required to…
A:
Q: Draw the products when each diene is treated with one equivalent of HCI a. b. C. d.
A:
Q: (a) Draw a line drawing for the Lewis diagram given below. Hint start converting the Lewis to a…
A: In this question asking Lewis structure that shown the bond and electron pair on molecule according…
Q: 1. Give the empirical formulas and names of the compounds formed from the following pairs of ions:…
A: This question belong to Chemical Bonding and molecular of inorganic chemistry.
Q: The statements in the tables below are about two different chemical equilibria. The symbols have…
A:
Q: HỌ—CH2 H HO–CH2 O 0 0=2-0 H₂N HO–CH2 O HN OH H H ó H H CH₂ H OH OH N O. N N H H OH 0= N H H NH 'N O…
A: Nucleoside contains a sugar unit ( either ribose or deoxyribose sugar ) and a base ( purine or…
Q: Identify the weakest bond in Propene, CH3CH=CH2 AND EXPLAIN why it is the weakest. please explain
A: We have to identify the weakest bond in propene, CH3-CH=CH2. Introduction: We know that, a sigma…
Q: What is the balanced formula for iron(ii) permanganate
A: Answer: Chemically, the most common oxidation states of iron are iron(II) and iron(III).
Q: What is the correct formula triphosphorus hexaoxide
A: Given, Triphosphorus hexaoxideRequired, The correct formula
Q: Station #7 = H2SO3 (aq) For this acid, what does the white bead represent?________ blue…
A: Solutions- Represent the color of beads of H2SO3 (aq)
Q: (d) What is Geometric isomerism? (e) What is Optical isomerism?\
A: In organic chemistry, stereochemistry is a study of spacial arrangement of atoms. Isomers are two or…
Q: What is the pH of a solution made from adding 10.00 mL of 0.15 M Na₂HASO4 to 25.00 mL of 0.10 M…
A:
Q: Question 12 of 40 > Macmillan Learning The identity of an unknown monoprotic organic acid is…
A:
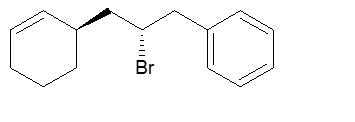
Which enantiomer is this molecule? R/R? S/S? R/S? S/R? Give your answer by indicating the configuration of the chiral carbons from left to right on the drawing.
Step by step
Solved in 3 steps with 3 images


