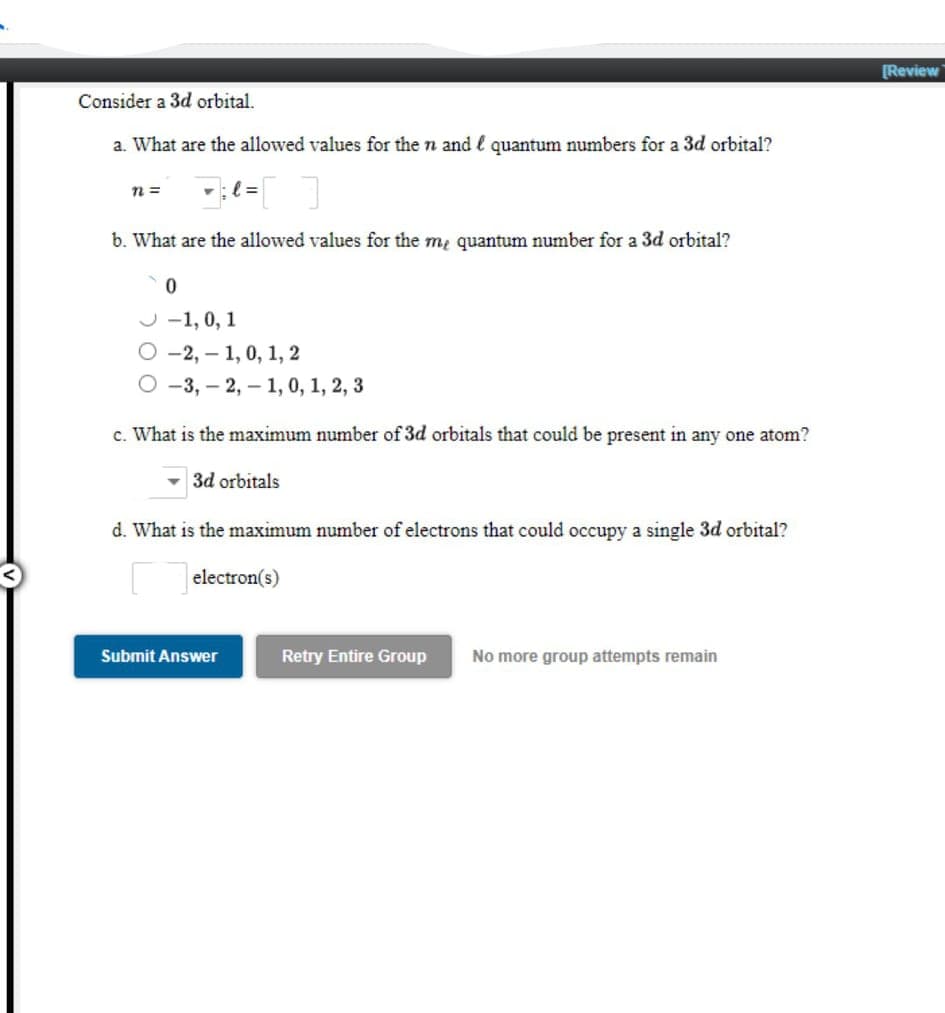
Consider a 3d orbital. a. What are the allowed values for the n and l quantum numbers for a 3d orbital? n = b. What are the allowed values for the me quantum number for a 3d orbital? J -1, 0, 1 О-2,— 1, 0, 1, 2 О -3,- 2, — 1, 0, 1, 2, 3 c. What is the maximum number of 3d orbitals that could be present in any one atom? 3d orbitals d. What is the maximum number of electrons that could occupy a single 3d orbital? electron(s)
Consider a 3d orbital. a. What are the allowed values for the n and l quantum numbers for a 3d orbital? n = b. What are the allowed values for the me quantum number for a 3d orbital? J -1, 0, 1 О-2,— 1, 0, 1, 2 О -3,- 2, — 1, 0, 1, 2, 3 c. What is the maximum number of 3d orbitals that could be present in any one atom? 3d orbitals d. What is the maximum number of electrons that could occupy a single 3d orbital? electron(s)
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter6: Electronic Structure And Periodic Properties Of Elements
Section: Chapter Questions
Problem 43E: How many electrons could be held in the second shell of an atom if the spin quantum number ms could...
Related questions
Question
Transcribed Image Text:[Review
Consider a 3d orbital.
a. What are the allowed values for the n and l quantum numbers for a 3d orbital?
b. What are the allowed values for the m, quantum number for a 3d orbital?
J -1, 0, 1
О-2, — 1, 0, 1, 2
-3, – 2, – 1, 0, 1, 2, 3
c. What is the maximum number of 3d orbitals that could be present in any one atom?
- 3d orbitals
d. What is the maximum number of electrons that could occupy a single 3d orbital?
electron(s)
Submit Answer
Retry Entire Group
No more group attempts remain
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning