Consider a 4-m-diameter spherical tank initially filled with liquid nitrogen at 1 atm and 196°C. The tank is exposed to 20°C ambient air with a heat transfer coefficient of 25 W/m² °C. The temperature of the thin-shelled spherical tank is observed to be almost the same as the temperature of the nitrogen inside. Disregarding any radiation heat exchange, determine the rate of evaporation of the liquid nitrogen in the tank as a result of the heat transfer from the ambient air, since the heat of vaporization of 198 kJ/kg at 1 atm. Tair = 20°C N, vapor 1 atm Liquid N₂ -196°C
Consider a 4-m-diameter spherical tank initially filled with liquid nitrogen at 1 atm and 196°C. The tank is exposed to 20°C ambient air with a heat transfer coefficient of 25 W/m² °C. The temperature of the thin-shelled spherical tank is observed to be almost the same as the temperature of the nitrogen inside. Disregarding any radiation heat exchange, determine the rate of evaporation of the liquid nitrogen in the tank as a result of the heat transfer from the ambient air, since the heat of vaporization of 198 kJ/kg at 1 atm. Tair = 20°C N, vapor 1 atm Liquid N₂ -196°C
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Kreith, Frank; Manglik, Raj M.
Chapter3: Transient Heat Conduction
Section: Chapter Questions
Problem 3.12P
Related questions
Question
i need clear ans by hand and solve very very fast in 20 min and thank you | ᴅʏʙᴀʟᴀ ?✨؛
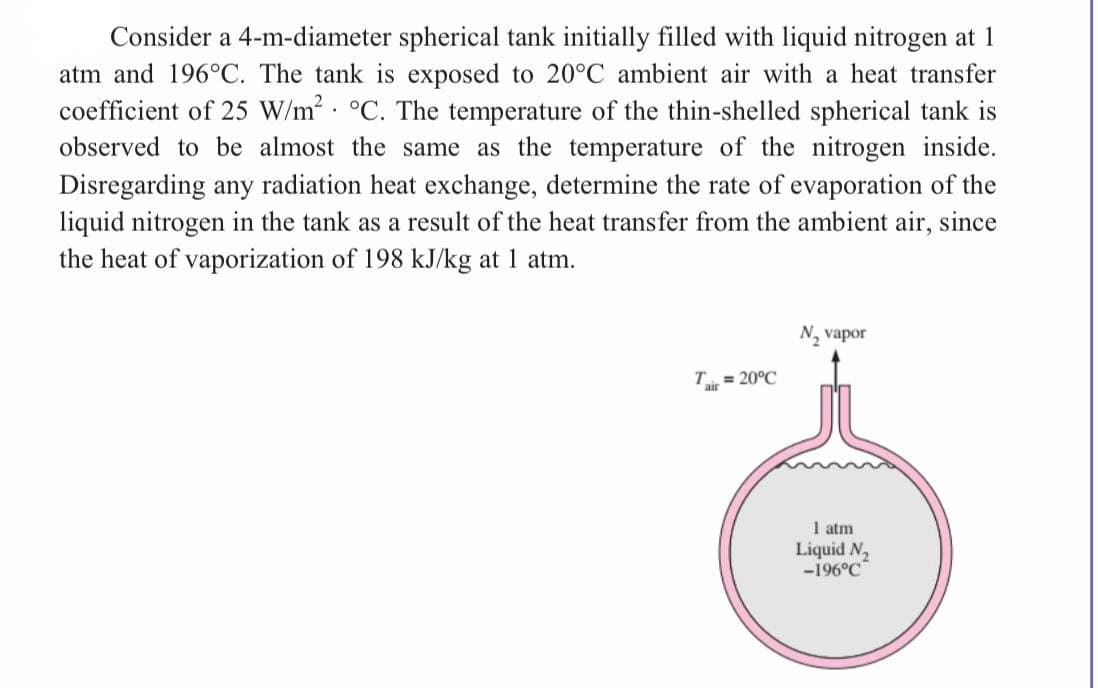
Transcribed Image Text:Consider a 4-m-diameter spherical tank initially filled with liquid nitrogen at 1
atm and 196°C. The tank is exposed to 20°C ambient air with a heat transfer
coefficient of 25 W/m² °C. The temperature of the thin-shelled spherical tank is
observed to be almost the same as the temperature of the nitrogen inside.
Disregarding any radiation heat exchange, determine the rate of evaporation of the
liquid nitrogen in the tank as a result of the heat transfer from the ambient air, since
the heat of vaporization of 198 kJ/kg at 1 atm.
.
Tair = 20°C
N₂ vapor
1 atm
Liquid N₂
-196°C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning