Consider the following equilibrium: 2NOCI(g) 2NO(g) + Ch(g) with K = 1.6 × 10 1.00 mole of pure NOC1 and 0.981 mole of pure Cl are placed in a 1.00-L container. Calculate the equilibrium concentration of NO(g). Select one: O a. 9.81 × 10-!M O b. 1.02 M O c.4.04 x 10³M O d. 2.02 x 103 M O e.5.71 x 10 M search
Consider the following equilibrium: 2NOCI(g) 2NO(g) + Ch(g) with K = 1.6 × 10 1.00 mole of pure NOC1 and 0.981 mole of pure Cl are placed in a 1.00-L container. Calculate the equilibrium concentration of NO(g). Select one: O a. 9.81 × 10-!M O b. 1.02 M O c.4.04 x 10³M O d. 2.02 x 103 M O e.5.71 x 10 M search
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter12: Gaseous Chemical Equilibrium
Section: Chapter Questions
Problem 13QAP: Consider the following reaction at 250C: A(s)+2B(g)C(s)+2D(g) (a) Write an equilibrium constant...
Related questions
Question
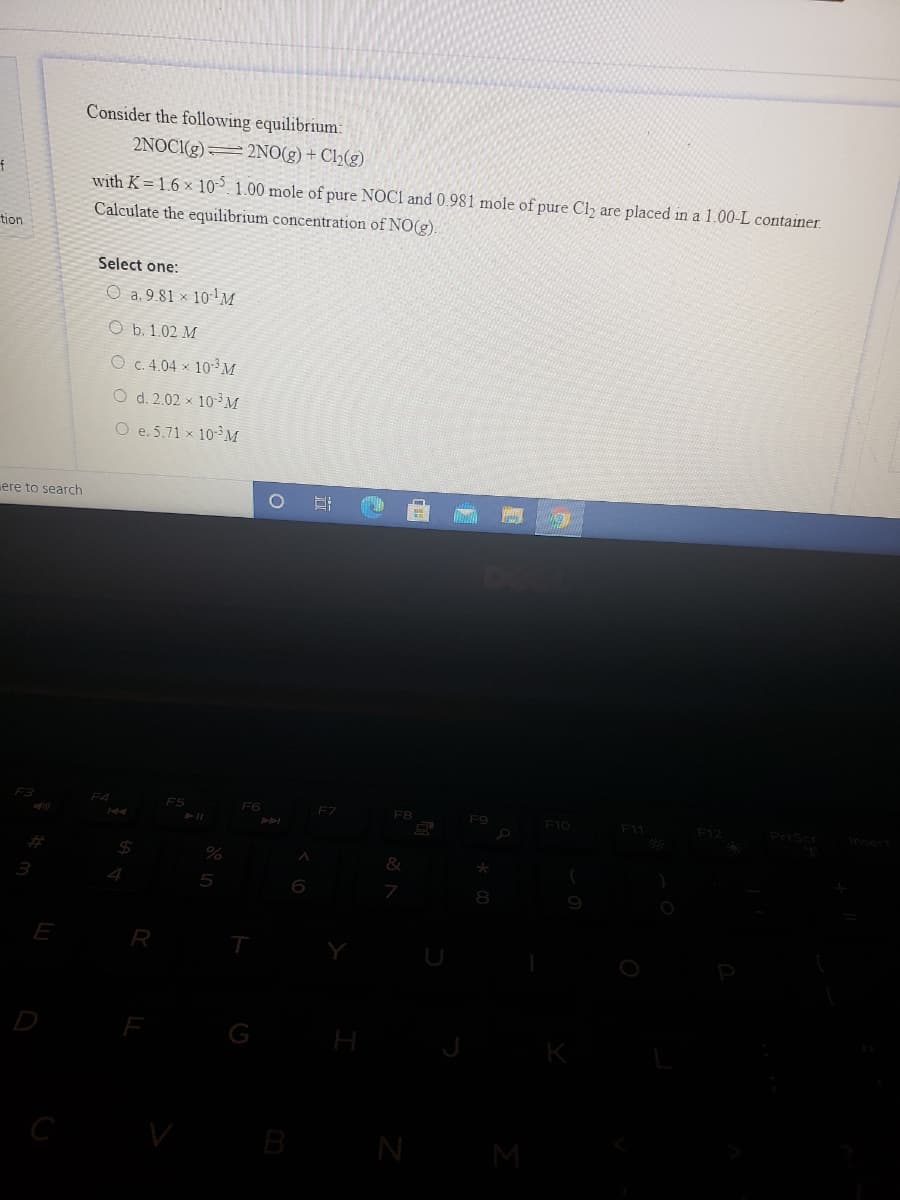
Transcribed Image Text:Consider the following equilibrium:
2NOCI(g)=2NO(g) + Ch(g)
with K = 1.6 × 10°. 1.00 mole of pure NOC1 and 0.981 mole of pure Cl are placed in a 1.00-L container.
Calculate the equilibrium concentration of NO(g).
tion
Select one:
O a. 9.81 × 10''M
O b. 1.02 M
O c. 4.04 × 10-³ M
O d. 2.02 × 10³M
O e.5.71 x 10 M
ere to search
F4
F5
F6
EZ
F8
F9
F10
F11
F12
Pri
%2$
4.
5
7
E
R
B
N
M
Expert Solution
Step 1
For the equilibrium Reaction -
2NOCl <-----> 2NO + Cl2
The , Equilibrium constant can be written as -
Kc = concentration of Products/concentration of Reactants
Kc = [NO]2[Cl2]/[NOCl]2
Here,
Kc = 1.6×10-5
[NO] = moles of NO = ?
[Cl2] = moles of Cl2 = 0.981 mole
[NOCl] = moles of NOCl = 1 mole
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning
