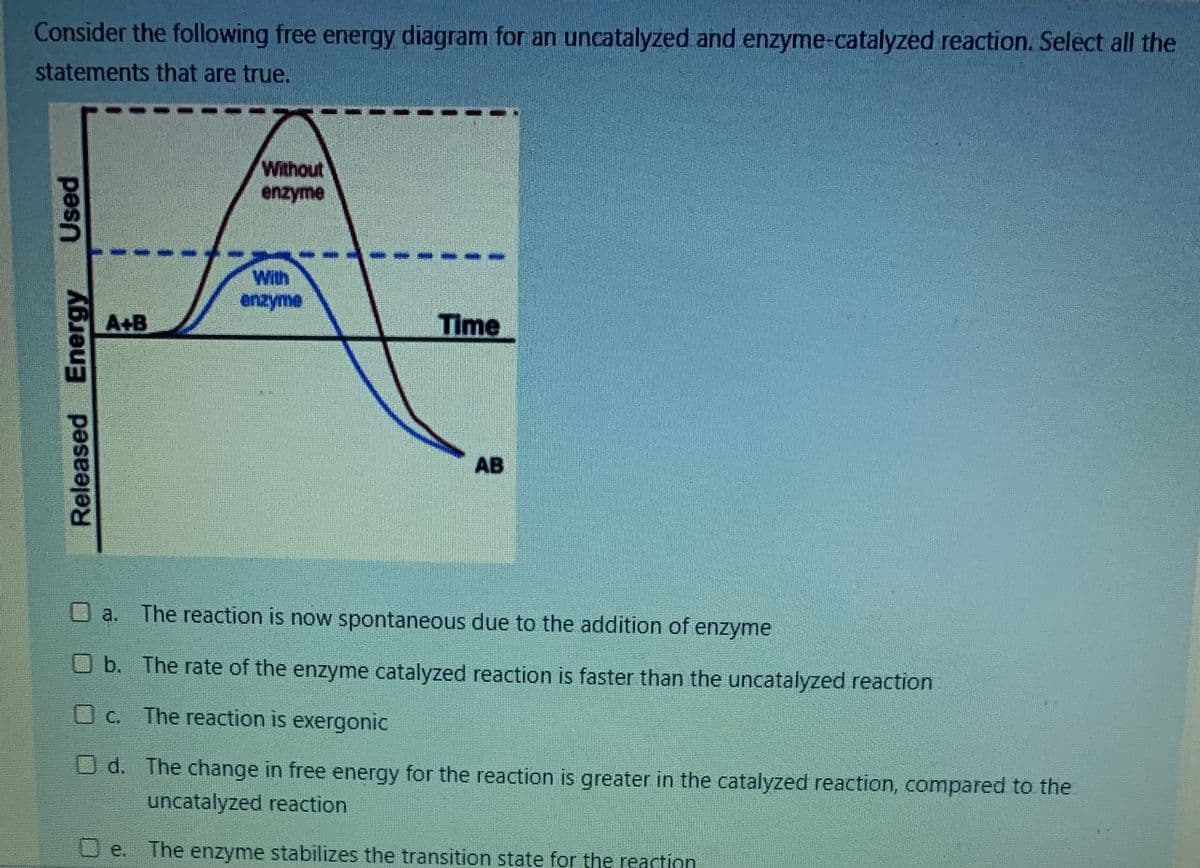
Consider the following free energy diagram for an uncatalyzed and enzyme-catalyzed reaction. Select all the statements that are true. Without enzyme pes
Q: Examine the figure below, which compares the energetics of a catalyzed and uncatalyzed reaction…
A: Most of the chemical reactions in cells cannot undergo quickly enough by themselves. They need…
Q: Effects of temperature and enzymes on free energy and activation energy Use the graph to determine…
A: Enzymes Biological catalysts of the chemical reactions. They are neither consumed nor permanently…
Q: Consider the following data set of enzymes A, BC and D which catalyze the same reaction. Enzyme A km…
A: Introduction: Catalysts are those substances that increase the rate of the reaction without…
Q: Given the following enzyme catalyzed reaction, identify the class and subclass of the enzyme…
A: Depending on the activity and the reaction they catalyse enzymes have 6 different classes…
Q: Sketch the complete reaction free energy diagram for an enzyme-catalyzed conversion of a single…
A: The reactants must overcome a kinetic barrier to proceed with the conversion to products in every…
Q: Use the energy graph shown here to define and explain the importance of the following four terms.…
A: Definition of biochemical metabolic pathway of enzymes In biochemistry, a metabolic pathway is a…
Q: An enzyme catalyzes the following reaction. Which of the following inhibitors would you expect to be…
A: Enzymes are biocatalyst. They are protein in nature (except-ribozymes), thermolabile in character…
Q: Students conducting research observe the rate of an enzyme-catalyzed reaction under various…
A: Enzymes are the biological catalysts that affect the rate of reaction. In most cases, enzymes are…
Q: Many organisms are able to live in extremely cold or hot temperatures, for example, the thermophilic…
A: There are many organisms, such as extremophiles, which live in extreme conditions. For example,…
Q: Define
A: Enzyme kinetic is a branch of Biochemistry. There are several factors that enzyme catalysis are…
Q: Briefly explain the Michaelis-Menten model of enzyme kinetics.
A: Enzymes are commonly composed of protein molecules that catalyze the biochemical reaction by…
Q: Calculate the factor by which the enzyme will decrease the rate of the reaction with the following…
A: The enzyme is a biological catalyst that increases the rate of reaction by changing the mechanism…
Q: Which image depicts the exergonic reaction spontaneously moves forward with the use of an enzyme? -…
A: The activation energy is the energy required to start a reaction. And enzyme lowers the activation…
Q: The change in the amount of product B in a reaction which is catalyzed by Enzyme A is given in the…
A: Michaelis menten constant, Km= It is the substrate concentration required to produce half maximum…
Q: Use the data below to determine the maximum velocity [in mM/s] of a certain enzyme-catalyzed…
A: We must know the Michaelis Menten equation: V=Vmax [S]Km+ [S]
Q: Draw the Michaelis-Menten Plot and Lineweaver-Burke Plot of an enzyme in the following situation.…
A: Enzymes are biocatalysts which increase the rate of biochemical reactions. Enzymes will not…
Q: Enzyme Activity vs. Drops of Enzyme 0.030 0 025 0.020 0 015 0.010 0.005 0.000 10 Enzyme (drops) 15…
A: The rate of a chemical reaction increases when the substrate concentration increases. Enzymes speed…
Q: In an enzyme-catalyzed reversible reaction what happens when a) rate of change of enzyme-substrate…
A: Enzyme is a biochemical substance mane up of proteins mostly that increases the rate of a…
Q: Suppose that an uncatalyzed reaction is spontaneous because AG has a value of -10 kcal/mol. An…
A: The spontaneous reaction is considered as the reaction which favors the formation of the product as…
Q: Use the plot provided to estimate Km and Vmax values for both with and without inhibitor of the…
A: Those proteins which help to speed up the chemical reaction are referred to as enzymes. The rate of…
Q: Identical reactions are catalyzed by the enzyme hexokinase with a Km of 0.2 mM in le cells and by…
A: In a classic Michaelis-Menten graph, the y-axis represents reaction rate and the x-axis represents…
Q: Using enzyme kinetics, illustrate the cooperative behavior of allosteric enzymes (plot of reaction…
A: Enzyme is basically biocatalyst that increase the rate of chemical reaction without itself being…
Q: Which of these statements about enzyme-catalyzed reactions is false? At saturated levels of…
A: Enzyme : E Substrate: S Product: P These both form E-S complex via S binding at the active site of E…
Q: Of the following constants in Enzyme kinetics, this is catalytic efficiency: О а. Vmax O b. Kcat/Km…
A: Introduction: Enzymes are proteins that increase the rate of the reaction that occurs in the living…
Q: An enzymes catalyzed reaction is studied in the presence and absence of an inhibitor. The following…
A: Enzymes are proteins which accelerate the rate of an enzyme catalyzed reaction. The rate of an…
Q: Explain why the diagram that you chose is for an enzyme that would catalyze the reaction, and…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: Reaction 25.2 Glucose (aq) + PO43- (aq) ⇄ Glucose-PO43- (aq) Reaction 25.2 is nonspontaneous and…
A: The Gibbs free energy change is the change in free energies of reactants and products in the…
Q: Which of the two graphs, below, represents an enzyme catalyzed version of the reaction Y-->X?…
A: A catalytic reaction is a reaction where the reactants will form the products with the help of a…
Q: Use the Michaelis-Menten plot to answer this question. What is the estimated value of Vmax of the…
A: Vmax : Reaction rate- when the enzyme gets fully occupied by substrate. Vmax- Maximum velocity
Q: Which of these statements about enzyme-catalyzed reactions is false? The activation energy for the…
A: An enzyme accelerates the rate of a chemical reaction several times as compared to the uncatalyzed…
Q: effectors of enzyme kinetics
A: Enzyme kinetics is the process of understanding the chemical reactions which are catalyzed by the…
Q: Please handraw this graph with all the necessary detailed information: Imagine that I text enzyme…
A: The graph is as shown in the white-board.
Q: Select the graph that correctly illustrates the effect of a positive modifier (effector) on the…
A: In case of allosteric enzymes they usually have more than one active site that involves binding of…
Q: Two enzyme catalyzed reactions, RA (red) and Rg (blue), have kinetic profiles shown below. Report…
A: In enzyme kinetics, the maximum rate of an enzyme-catalyzed reaction is Vmax, where Vmax is the…
Q: Describe the reactants and products of the following enzyme catalyzed reaction. Names of molecules…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: You have a reaction that is very slow on its own, but is accelerated in the presence of an enzyme.…
A: 1. Enzyme increased Kcat - Kcat is the maximal velocity of the catalyzed reaction divided by the…
Q: Explain how the following changes affect the rate of an enzyme-catalyzed reaction in the presence of…
A: In uncompetitive inhibition, the enzyme inhibitor binds to the enzyme-substrate complex. This type…
Q: The turnover number of an enzyme catalyzed :reaction is equal to .K2 .a O .K1 .b O .K3/1 .c O .K3 .d…
A: A general enzyme catalyzed reaction is designated with the mechanisms shown below: E+S↔ES→E+P Here,…
Q: OH CH3CHCO2- CH3CCO2-
A: In the given reaction lactate is converted into pyruvate.
Q: In enzyme catalysed reactions, the energy level of the enzyme/substrate (or ES) complex is higher…
A: The enzyme is synthesized in the body and reactions are carried out in presence of enzymes. They are…
Q: Use the plot (below) to estimate values of KM and Vmax for an enzyme-catalyzed reaction. Use 1…
A: Michaelis Menten (MM) kinetics was developed by Canadian physician Maud Menten and German biochemist…
Q: Which of the listed effects would be brought about by any enzyme catalyzing the following simple…
A: Introduction: Those compound that increases the rate of the reaction without undergoing any change…
Q: Classify the enzyme that catalyzes each of the following reactions:
A: Enzymes are bio-catalyst. They lower the activation energy of a enzyme catalyzed reaction. Enzymes…
Q: The effect of an inhibitor I on the rate of a single-substrate, enzyme-catalyzed reaction was…
A: The Michaelis-Menten equation for enyzme kinetics is : VO = Vmax[S] / Km + [S] The Lineweaver-Burk…
Q: Explain how the observation that reaction (B) runs faster than reaction (A) is related to the enzyme…
A: Michaelis-Menten model of enzyme kinetics is a very useful model which describe the relationship…
Q: he figure displays the relationship between initial rate of product formation and reactant…
A: in this Figure At low concentration rate of product formation is directly proportional to…
Q: Consider the following free energy diagram for an uncatalyzed and enzyme-catalyzed reaction. Select…
A: The process associated with the formation of bonds as well as conversion of a reactant into another…
Q: . Would you expect an "enzyme" designed to bind to its target substrate as tightly as it binds the…
A: Enzymes are a special class of proteins that can catalyze a biochemical reaction. There are six…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Which of the following is TRUE concerning the induced fit model of enzyme catalysis? * (One correct answer only) A. The active site can be influenced by molecules binding elsewhere on an enzyme B. The initial binding of enzyme and substrate is the most tightly bound conformation C. The induced fit must occur prior to the initial binding of enzyme and substrate in order for the reaction to proceed D. The binding of enzyme and substrate is weakest in the transition stateWhich of the following is NOT true? a. For many enzymes, Vmax is essentially equal to k3. b. Km is the reaction rate at 1/2 Vmax. c. At steady state the formation of ES is equal to the breakdown of ES. d. A Lineweaver-Burke plot generates a straight line. e. An Eadie-Hofstee plot generates a straight line.Which of the following is true under the following conditions: an enzyme displaying Michaelis-Menten kinetics where the enzyme concentration is 10 nM, the substrate concentration is 45 mM, and the Km is 50 µM? a) The enzyme has low catalytic efficiency for the substrate. b)The rate of catalysis is near half-maximal velocity. c)The enzymatic reaction is near maximal velocity. d)Halving the substrate concentration has little effect on the catalytic rate. e) There is not enough information provided.
- Evaluate the following statements concerning enzyme kinetics. Which one of the statements is false? a. Enzyme saturation fluctuates. b. In an uninhibited enzymatic reaction system, adding an excess of substrate will increase the reaction velocity beyond Vmax. c. The Vmax of an enzyme kinetics graph represents the point at which the enzyme is saturated with substrate. d. Non-competitive inhibition of an enzymatic reaction can be overcome by adding more unaltered enzyme. e. The activation energy of a reaction can be reduced by the presence of an enzyme.In a Lineweaver-Burk graph, the lines representing the uninhibited and inhibited enzyme catalyzed reaction meet each other on the x-axis. The type of inhibition which is occurring is: a) competitive b) noncompetitive c) uncompetitive d) allosteric CO2 exerts direct activity upon hemoglobin by: a) blocking oxygen from binding to the heme group b) displacing BPG from the central cavity c) oxidizing Fe+2 to Fe+3 which does not bind oxygen d) forming an N-terminal carbamate which favors the T-state The dominant motif found in hemoglobin and myoglobin is: a) helix-turn-helix b) twisted beta sheet c) beta barrel d) random coil Which of these is an ketohexose? a) fructose b) glucose c) ribose d) erythrose Which of these is a constitutional isomer of d-glucose? a) fructose b) galactose c) l-glucose d) ribose Which of these is an enantiomer of d-glucose? a) d-fructose b) d- galactose c) l-glucose d) d-ribose Which of these is a diastereomer of…Which of the following statements is/are true about enzyme-catalyzed reactions? A. The reaction is faster than the same reaction. B. The free energy change of the reaction is opposite from the reaction in the absence of the enzymes. C. The reaction always goes in the direction toward chemical equilibrium. D. A and B only E. A, B and C
- The graph shows the reaction coordinate of an enzymatic reaction of substrate to product a) which number correlates with the overall Keq of the reaction going from S to P? b) which number correlates with the velocity of the reaction WITHOUT enzyme (ie. starting with only substrate)? C) which number correlates with the overall velocity of the reaction WITH SATURATED enzyme D) the initial velocity of this reaction increases by a specific factor in the presence of enzyme. The difference between which two numbers best correlates with this enhancement in velocity? 1) 3 and 1 2) 3and 2 3) 3 and 4 4) 3 and 5 5) 3 and 6In competitive inhibition, increasing concentrations of the inhibitor will have the following effect on the kinetics of the enzyme: A. Km will decrease. B. Vmax will stay the same. C. The reaction will cease because the inhibitor binds irreversibly. D. Km / Vmax will stay the same.The enzyme glutamine synthetase catalyzes the following reaction: glutamate + ATP + NH3 → glutamine + ADP + phosphate Which of the following statements about this reaction is correct? he reaction is only exergonic if enzyme is added b. The reaction is endergonic whether or not enzyme is added c. The reaction is exergonic whether or not enzyme is added d. The rate of oxygen production will be unaffected e. The reaction is only endergonic if enzyme is added
- Which of the following statements about a plot of V0 vs. [S] for an enzyme that follows Michaelis-Menten kinetics is false? a. As [S] increases, the initial velocity of reaction V0 also increases. b. At very high [S], the velocity curve becomes a horizontal line that intersects the y-axis at Km. c. Km is the [S] at which V0 = 1/2 Vmax. d. The shape of the curve is a hyperbola. e. The y-axis is a rate term with units of μm/min.The enzyme glutamine synthetase catalyzes the following reaction: glutamate + ATP + NH3 → glutamine + ADP + phosphate Which of the following statements about this reaction is correct? a. The reaction is endergonic whether or not enzyme is added b. The reaction is only endergonic if enzyme is added c. The reaction is only exergonic if enzyme is added d. The rate of oxygen production will be unaffected e. The reaction is exergonic whether or not enzyme is addedWhich of the followingdescribe superior properties of enzymes (biological catalysts) over traditional chemical catalysts? a. They are mostly and generally operative under mild temperature, pressure, and pH conditions b. They are regulated only by substrate concentration c. They do not effect the reaction equilibrium, but lower the reaction's activation energy d. They are recycled at the end of the reaction Choose all that apply

