Consider the following half reactions Half-reactionE° (V) I2(s) + 2e- 2I-(aq) E 0.535V Pb2+(aq) + 2e- Pb(s) E-0.126 VZn2+(aq) + 2e- Zn(s) E-0.763V 1. The strongest oxidizing agent 2.the weakest oxidizing 3 the weakest reducing 4 the strongest reducing 5 will I2(s) oxidize Zn(s) to Zn2+(aq) Yes/No 6 Which species can be oxidized by Pb2+(aq) if none leave box blank Ps (Numbers 1 2 and 3 are answered already I only need 4 5 6
Consider the following half reactions Half-reactionE° (V) I2(s) + 2e- 2I-(aq) E 0.535V Pb2+(aq) + 2e- Pb(s) E-0.126 VZn2+(aq) + 2e- Zn(s) E-0.763V 1. The strongest oxidizing agent 2.the weakest oxidizing 3 the weakest reducing 4 the strongest reducing 5 will I2(s) oxidize Zn(s) to Zn2+(aq) Yes/No 6 Which species can be oxidized by Pb2+(aq) if none leave box blank Ps (Numbers 1 2 and 3 are answered already I only need 4 5 6
Chapter18: Introduction To Electrochemistry
Section: Chapter Questions
Problem 18.3QAP
Related questions
Question
Consider the following half reactions Half-reactionE° (V)
I2(s) + 2e- 2I-(aq) E 0.535V
Pb2+(aq) + 2e- Pb(s) E-0.126
VZn2+(aq) + 2e- Zn(s) E-0.763V
1. The strongest oxidizing agent
2.the weakest oxidizing
3 the weakest reducing
4 the strongest reducing
5 will I2(s) oxidize Zn(s) to Zn2+(aq) Yes/No
6 Which species can be oxidized by Pb2+(aq) if none leave box blank
Ps (Numbers 1 2 and 3 are answered already I only need 4 5 6
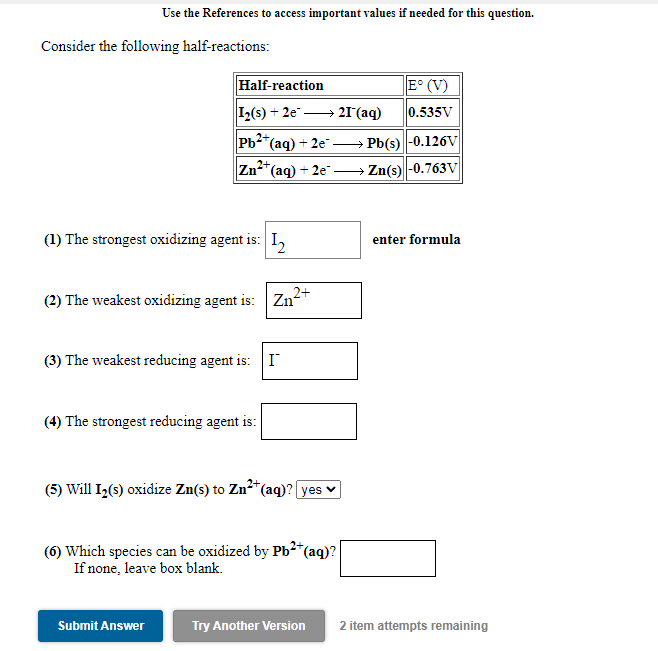
Transcribed Image Text:Use the References to access important values if needed for this question.
Consider the following half-reactions:
Half-reaction
E° (V)
I2(s) + 2e -
→ 21(aq)
0.535V
Pb2*(aq) + 2e → Pb(s) |-0.126V
Zn*(aq) + 2e –→ Zn(s) -0.763VV
(1) The strongest oxidizing agent is: I,
enter formula
2+
(2) The weakest oxidizing agent is: Zn-
(3) The weakest reducing agent is: I
(4) The strongest reducing agent is:
(5) Will I2(s) oxidize Zn(s) to Zn²*(aq)? yes v
2+,
(6) Which species can be oxidized by Pb²*(aq)?
If none, leave box blank.
Submit Answer
Try Another Version
2 item attempts remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning