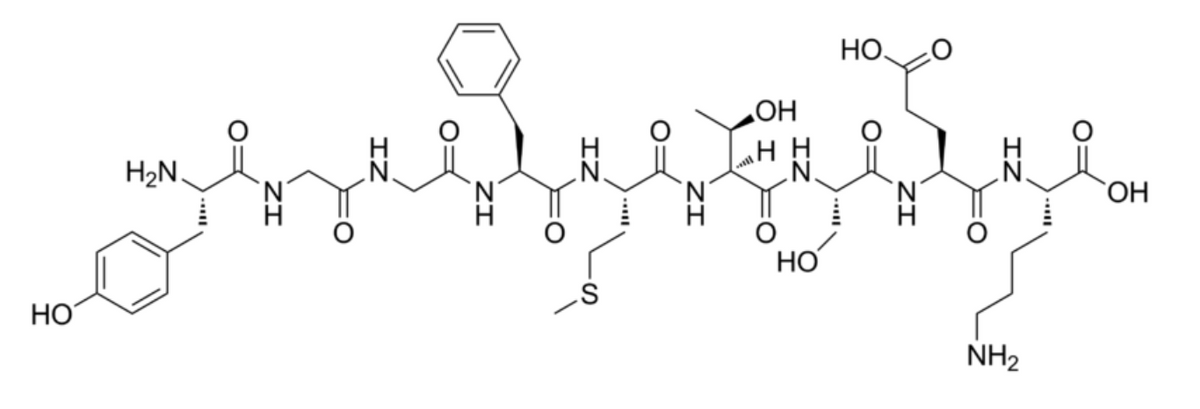
Consider the oligopeptide whose structure is shown below: How many peptide bonds are there? Give the complete name of the oligopeptide What is the net charge of the oligopeptide at pH 7? Explain.
Q: 3. Draw both hemiacetal forms of d-ribofuranose and both hemiacetal forms of d-xylofuranose.
A: Alpha and beta anomers of sugars form hemiacetal structures. Hemiacetals have structure that…
Q: Draw the arrow-pushing mechanism
A: The transfer of amino acid residue from aminoacyl-tRNA to peptidyl tRNA is an important reaction in…
Q: is albumin a macromolecule? if yes which type?
A: The biological macromolecules can be divided into four classes: the nucleic acids, the proteins, the…
Q: Consider the peptide L-E-T-S-P-R-A-Y Which amino acid(s) is/are basic? A. Y B. P C. R D. L E.…
A: An amino acid is an organic substance that combines to create proteins, the body's building blocks.…
Q: need help to 1)hand draw the dipeptides that contains Glutamic acid and Proline. 2)hand draw the…
A: Amino acids are biomolecules that have an amino group, a carboxyl group and a side group attached to…
Q: A) Write out both strands of the recognition sequences for BamHI and Sau3A, show what they would…
A:
Q: Discuss why fluoroacetate can be called a mechanism-based inhibitor. what is a mechanism based…
A: Introduction Enzymes are known as bio-catalyst. They increases the rate of a chemical reaction by…
Q: Is the BSA a good stadard to use in the bradford assay?
A: Introduction Proteins are the most abundant macromolecule present in our body. Proteins are made up…
Q: Compare and contrast Hb and Mb in terms of the following: • Physiological roles • Structures • O2…
A: Hemoglobin and Myoglobin are used as classic models for studying protein-ligand interaction. Both…
Q: What are the monomers of DNA & RNA? What 3 parts do the monomers contain?
A: Introduction: Nucleic acids are large biomolecules that carry hereditary information for cellular…
Q: Substance that transports fatty acid from the interrnembrone space to the mitochondrial matrix? A.…
A: Long chain fatty acids must be transported into the mitochondrial matrix for beta oxidation. As they…
Q: Draw the Citric acid cycle pathway and identify the different types of organic reaction mechanisms.…
A: Citric acid cycle involves 8 biochemical reactions that occur in a cyclic manner. The acetyl CoA…
Q: Consider the following peptide sequence: Met-Ser-Val-Thr-Ile-Lys-Ala-Cys-Leu-Ser-lle-Tyr-Phe-Ser…
A: Structure of a protein or a peptide is described in four levels: primary, secondary, tertiary and…
Q: A.We have seen from history the role science has played in the prevention and control of diseases.…
A: Epidemiology : It is the study of diseases in a population, studying the reason , how it spread, why…
Q: Starting from glutamine ,glycine, aspartate, N-10 formal-ThF ,how many ATP equivalents are required…
A: Glutamine : It is the most abundant amino acid in the body.It is synthesized in the muscles and the…
Q: What is the dominant force behind the interaction between side chains of 2 Cys? A H-bond B…
A: Cysteine is a sulphur containing amino acid. It has side chain with thiol group (-SH).
Q: Which of the following statements regarding hydrogen bonding in secondary structures is true?…
A: A weak chemical link that exists between a hydrogen atom that is partly positively charged and an…
Q: c) A lysine residue in the active site of UstD is involved in forming a covalent Schiff base linkage…
A: UstD is an enzyme that decarboxylates specific acidic amino acids and allows the transfer of the…
Q: You've discovered a novel biosynthetic pathway that makes the molecule caffeine. The pathway can be…
A: In biochemistry, a metabolic pathway is series of enzyme catalysed reaction where product of one…
Q: describe how 18O from water can end up in C18O2. Feel free to draw structures and reference any…
A: We know that the end product of glycolysis is pyruvate which to enter citrate cycle cycle for…
Q: In biochemistry,what is the main function of amide group and carboxyl group on amino acids?which one…
A: Proteins are made up of amino acids. When few amino acids are joined, the formed structure is called…
Q: Lable out the main difference of Primary, secondary, tertiary, and quatrenary protein structure in…
A: As per the central dogma of molecular biology, DNA contains the code that is necessary for the…
Q: 1. Why are eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) important? 2. What are the…
A: Lipids are compounds that are soluble in organic solvents and insoluble in polar solvents such as…
Q: Ceruloplasmin is a blue-colored monomeric oxidase found in mamma- lian blood plasma. It contains…
A: Ceruloplasmin is a copper containing globulin which helps in the transport of copper in the…
Q: Predict the amino acid sequence based on the conditions below. Use the one-letter abbreviation of…
A: Amino acids are building block of polypeptide chain. its alpha carbon contains carboxyl group, amine…
Q: QUESTION 5 What was the distance (in cm) traveled by the SAMPLE C in the TLC below: 10 6 8 7 56 D 2…
A: TLC is a separation technique in which silica is used as a stationary phase and the non-polar…
Q: Calculate the pH of a blood plasma sample with a total CO₂ concentration of 25.7 mM and bicarbonate…
A: The acid dissociation reaction taking place here is given below, where carbonic acid (H2CO3) gives…
Q: Q3.1 Describe two ways in which oligosaccharides in human milk provide benefits to newborn babies.…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which of the which of the following forces are involved in maintaining the primary structure of a…
A: There are 4 levels of protein structure: Primary structure - covalent bonds are responsible for…
Q: What is the subject of the figure: What is the main message of the figure? Give a concise, complete…
A: Subject of the figure is point mutation of a base which leads to different translated product of a…
Q: can you explain using a drawing presenting the factors like pH, temperature, substrate…
A: Enzymes are protiens which act as a biochemical / biological catalysts by speeding up the rate of…
Q: 1. Consider the enzyme pyruvate carboxylase. a. What pathway(s) does this enzyme function in? b.…
A: “Since you have posted a question with multiple sub-parts, we will solve the first four sub-parts…
Q: What is the significance of acetyl-CoA to lipid metabolism?
A: The synthesis and breakdown of lipids in cells is known as lipid metabolism. It involves the storing…
Q: The following data describe the catalysis of cleavage of peptide bonds in small peptides by a…
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: Assumed that you have isolated the protein cytochrome C from a cell lysate. Discuss the advantages…
A: Cytochrome C Cytochrome C is a protein that is loosely bound to the inner membrane of mitochondria…
Q: What possible quiestions can be askeed under the following question: How can one inestigate the tyoe…
A: AZT is a anti-HIV drug. It is a nucleoside analogue. AZT is incorporated in the replicating DNA in…
Q: How does dithiothreitol (DTT) differ from B-mercaptoethanol (BME) as a reducing agent?
A: A reducing agent is a substance that reduces the substrate and itself gets oxidised. The reducing…
Q: PROTEINS 1. Give the general structure of amino acid. 2. Give the 20 amino acids (include structure,…
A: Amino acids are the building blocks of protein. They are joined by peptide bonds to make a…
Q: Ion channels can open or close based on electrochemcical gradient.
A: Ion channels are proteinaceous pathways that allow the passage of ions across a membrane at rates…
Q: Energy is stored long-term in the bonds of _____ and used short-term to perform work from a(n)…
A: A molecule is defined in chemistry as a grouping of distinct atoms. Water molecules (H2O) and carbon…
Q: 4. Cis peptide bonds are rarely found in proteins because they are about 8 kJ/mole less stable than…
A: Proteins are unbranched polymers constructed from 20 standard α-amino acids. They have four levels…
Q: What would you predict about the ratio of hydrophilic to hydro- phobic amino acid residues in a…
A: Before getting into the calculations, we have to assume that our globular protein have a spherical…
Q: What does it mean if a compound is said to have “high phosphoryl transfer potential”?
A: ATP is known as the energy currency of the cell. ATPs are required by the cell for various reactions…
Q: Which of the following would you expect to happen if amino acids 579 to 582 (YIYV) in the PDGF-Beta…
A: PGDF beta-receptor contain multiple domains that activate the various pathways inside the cell. The…
Q: 14) Which of the following statements is true under the conditions provided: the enzyme…
A: For a one-substrate enzyme-catalyzed reaction, the Michaelis-Menton equation shows the quantitative…
Q: Which of the following statements about alpha-helix and beta-sheet structure are true? More than one…
A: A protein's function depends on its structure. There are four levels of protein structure: primary,…
Q: 1. Why does nitric acid stain the skin yellow? 2. How does the Xanthoproteic test differ from the…
A: Nitric Acid : It is a nitrogen oxoacid having a formula HNO3 , in which the nitrogen atom is…
Q: Based on the functional groups in estrogen and testosterone, which would be more hydrophobic.…
A: Estrogen and testosterone are sex hormones.They are secreted by the gonads and regulate the…
Q: 3 a. b. Name and very briefly describe the four levels of protein structure. Hemoglobin has a…
A: As per the central dogma of molecular biology, DNA contains the code that is necessary for the…
Q: So when drugs are bound to plasma proteins, the effects are NOT intensified?
A: Drugs are chemical substances that have the capability to improve physical and mental illness.…
Consider the oligopeptide whose structure is shown below:
-
- How many peptide bonds are there?
- Give the complete name of the oligopeptide
- What is the net charge of the oligopeptide at pH 7? Explain.
- If you were asked to fragment the oligopeptide with just one enzyme or one chemical reagent, what enzyme or chemical reagent will you use to give the most number of fragments? Show where the peptide bond cleavage will occur.
Step by step
Solved in 3 steps

- In 2-page worth of words (around 500), discuss in depth the risks and hazards of CYCLAMATE in the body. What will be the reactions and the harmful products and their effects? Why is this a concern?True or false: starch decomposes in the presence of high concentrations of I2.Health risk of exposure to Bisphenol A (BPA)
- diagramatical representation of exposure of Bisphenol A effecting human healthwhat is hydroxychloroquine? should you publicly speak on intake of hydroxychloroquine? Does it matter that former President Trump took this drug and told the world about it?Triacylglycerols in adipose tissue do not enter the bloodstream as triacyglycerolsexplain

