Consider the solubilities of a particular solute at two different temperatures. Temperature (C) Solubility (g/100 g H,O) 20.0 36.2 30.0 67.7 Suppose a saturated solution of this solute was made using 44.0 g H,0 at 20.0 °C. How much more solute can be added if the temperature is increased to 30.0 °C? mass: g
Consider the solubilities of a particular solute at two different temperatures. Temperature (C) Solubility (g/100 g H,O) 20.0 36.2 30.0 67.7 Suppose a saturated solution of this solute was made using 44.0 g H,0 at 20.0 °C. How much more solute can be added if the temperature is increased to 30.0 °C? mass: g
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter1: Matter And Measurements
Section: Chapter Questions
Problem 53QAP: Magnesium sulfate (MgSO4) has a solubility of 38.9 g/ 100 g H2O at 30C. A solution is prepared by...
Related questions
Question
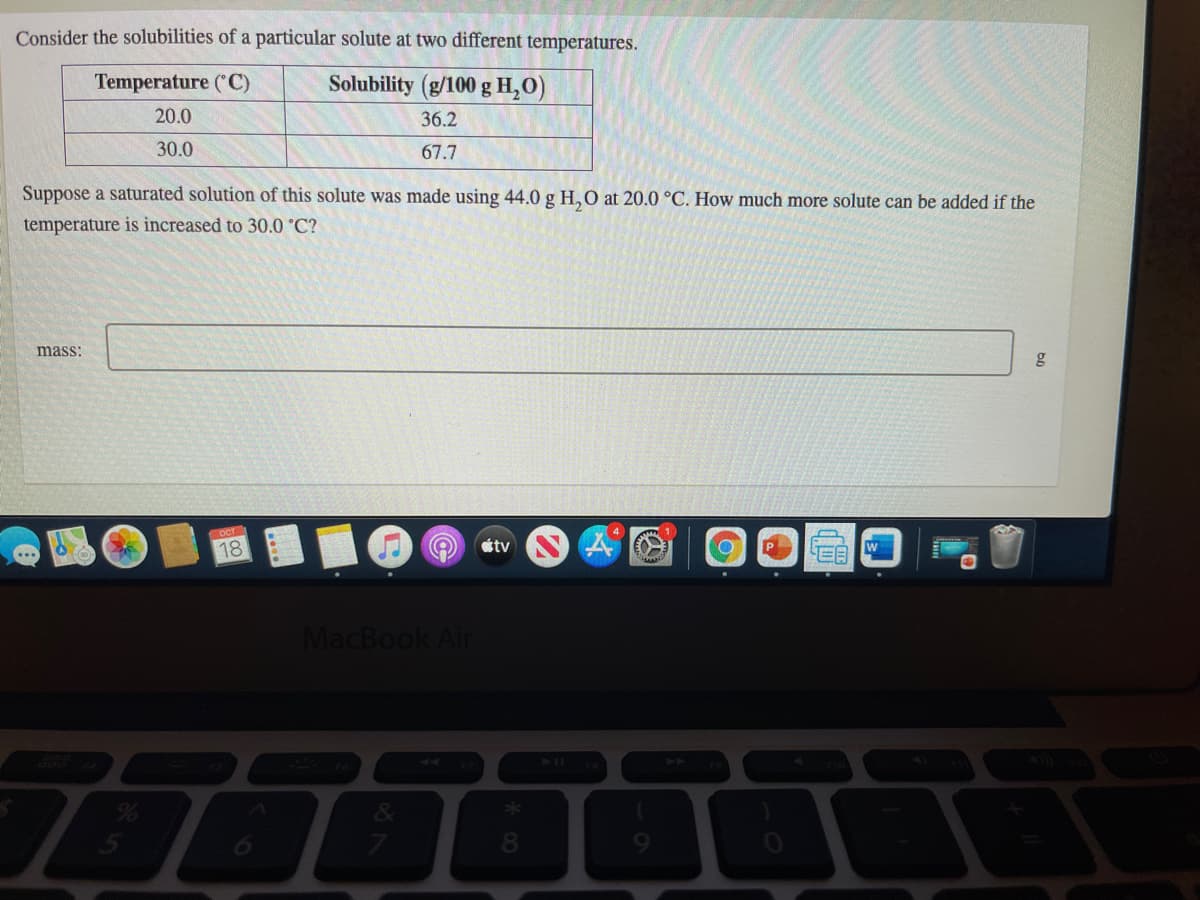
Transcribed Image Text:Consider the solubilities of a particular solute at two different temperatures.
Temperature (C)
Solubility (g/100 g H,O)
20.0
36.2
30.0
67.7
Suppose a saturated solution of this solute was made using 44.0 g H,0 at 20.0 °C. How much more solute can be added if the
temperature is increased to 30.0 °C?
mass:
g
18
tv
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning