Q: Explain why the atomic mass listed on the periodic table is 126.9 amu for an atom of iodine and, for...
A: Isotopes are the compounds which have same atomic number but different mass number.
Q: In the following reaction, potassium chlorate decomposes to potassium chloride and oxygen gas. How m...
A:
Q: Sulfuryl chloride, SO2Cl2, decomposes when heated. SO2Cl2(g) → SO2(g) + Cl2(g) In an experiment...
A: Given reaction is followed first order kinetics
Q: Complete the table below by writing the symbols for the cation and anion that make up each ionic com...
A: Cation: The species which acquires a positive charge on it. Anion: The species which acquires a neg...
Q: Phosphorous trichloride and phosphorous pentachloride equilibrate in the presence of molecular chlor...
A: Determine Kp of the given reaction---
Q: skeletal equation
A:
Q: What product is formed when the alcohol is oxidized with K2Cr2O7? In some cases, no reaction occurs ...
A: This question belong to Reagents (Oxidation-Reduction Reaction) . K2Cr2O7 are the Powerful Oxidising...
Q: What is the boiling point of a solution made by dissolving 10.0 g sucrose ( C12H22O11C12H22O11 ) in ...
A:
Q: 67- g/ Give a reaction mechanism OH EtO,CN=NCO,Et ʼNO2 CH2NO2 PPH3 92%
A: This reaction is Mistonbou inversion reaction where inversion of a nucleophilic centre takes place ...
Q: Which of the following assumptions are made by the kinetic molecular theory of gases? I. Gas molecul...
A: in this question find out the correct option for kinetic theory of gasses
Q: A patient is suspected of having low stomach acid, a condition known as hypochloridia. To determine ...
A: A doctor titrate 15.00 mL of gastric juice with 24.7 mL of 1.27×10-4 M KOH solution. We have to find...
Q: 14 Answer the exercise indicated in the image The reaction rate was investigated as a function of th...
A:
Q: Br For the reaction conditions shown above, predict the mechanism(s) for the major reaction pathway(...
A: Given reaction is : Predict the mechanism of the given reaction = ? Options are : a). SN2 b). E2...
Q: 9 43% l TEW LTE 12:49 PM Done 14 of 14 Bal's Law NChane gas scuplesa wume of 12iters at 770 torr pre...
A: Given, The volume of chlorine gas at 720 torr is 1.2 Liters. The volume at 1 atm pressure is:
Q: 7. Draw three valid Lewis structures for HNPH and predict the most likely HNP and NPH bond angles. E...
A: In Lewis Structure all the octet of atoms are complete but phosphorous Do not follow any octet rule...
Q: Diethyl ether[(CH3CH2)2O]is an imperfect anesthetic in part because it readily combusts in air to fo...
A: Diethyl ether is an imperfect anesthetic in part because it readily combusts in air to form CO2 and ...
Q: a 165lb man was found to contain 3.27mg of cobalt (Co). how many atoms of cobalt is this?
A:
Q: Calculate the enthalpy of the following reaction for the gas phase salts: Needed facts: K = Coulombs...
A: The energy of the electrostatic attraction (E), a measure of the force’s strength, is inversely prop...
Q: A monoprotic weak acid, HA, dissociates in water according to the reaction HA(aq) = H*(aq) + A¯(aq) ...
A: Given, the dissociation equation for the weak acid, HA: HA (aq) ⇌ H+ (aq) + A- (aq) Given the equili...
Q: 2. A galvanic cell consists of a lead anode immersed in a 1.00 M solution of Pb(NO3)2 and a platinum...
A:
Q: In an energy diagram for an exothermic chemical reaction which of the following is true? Group of an...
A: Given that : We have to identify which of the following statements is true in an energy diagram for ...
Q: the concentration of H3O+ of seawater is 3.16 x 10-9. what is the pH of seawater?
A: Ph depends on concentration of H+ ions...
Q: 3. Calculate the concentration of Cl- in the 10mL unknown NaCl solution. The Ksp for Silver Chlorid...
A: Answer is below
Q: e weight percent
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: 2) If doubling the concentration of nitrate ion in the reaction above causes the rate of reaction to...
A: The relation between the concentration of the reactants and the rate of reaction is expressed by the...
Q: a liquid cough syrup has 3.0mL of active ingredient in 50.0mL of solution. if 10.0mL is the dose, ho...
A: The above question asks to calculate the volume of active ingredient that is present in the dose. It...
Q: QUESTION S A 10 ml of HBr solution is titrated with a 1M NAOH solution. Some drops of phenolphthalei...
A:
Q: 22 What class of enzyme catalyzes each of the following reactions? (a) OH CH3CHCO,- CH3CCO,- (b) Он ...
A:
Q: hát was the funclion of the anhydrous calcium chloride? Illustrate this function by completing this ...
A: Extraction is the method here to get your compound that is in aqueous medium with the help of an org...
Q: Perform these operations in scientific notation with the appropriate number of significant figures. ...
A:
Q: sodium hydrogen carbonate (NaHCO3) was reacted with an excess of 5.00 M hydrochloric acid (HCI). The...
A: Reaction 1: NaOH (s) + HCl (aq)----> NaCl(aq) + H2O(l) Reaction 2: NaOCO3 (s) + HCl (aq)----> ...
Q: How many secondary hydrogens are there in the following compound? > View Available Hint(s) O 4 Submi...
A:
Q: The chemical reaction of silver nitrate with sodium carbonate produces silver and sodium nitrate. a...
A: (a) Balanced Chemical equation between AgNO3 and Na2CO3 Na2CO3(aq) + 2AgNO3...
Q: An analyst weighs the same tablet three times on a well calibrated analytical balance and obtains th...
A: Environmental changes, such as in temperature or humidity. Exposure to harsh conditions, such as cor...
Q: which class of enzymes catalyzes the given reaction
A:
Q: Enough of a monoprotic weak acid is dissolved in water to produce a 0.0178 M solution. The pH of the...
A:
Q: Convert 8.5 mg/mL in μg/μL
A: mg/mL is equal to μg/μL
Q: 25) Triglycerides are esters of fatty acids with
A: Given, Triglycerides are esters of fatty acids with ________. Ester bond: Ester bonds Forms between ...
Q: Now suppose, instead, that 33.653 g of a volatile solute is dissolved in 470.0 g of water. This solu...
A:
Q: Draw the major organic product of the following reaction. Inorganic byproducts like water or salts d...
A: Organic reactions are those in which organic reactant react to form organic products.
Q: Sulfuric Acid is added to sodium chloride. The reaction yields sodium sulfate and hydrochloric acid....
A: Reactants are the species present on left side of the chemical equation and products are present on ...
Q: 3. Given that: 2CH;OH (I) + 302 (g) → 2CO2 (g) + 4H2O (I) AH = -1452 kJ A 2.00 g sample of methanol,...
A:
Q: Just nitrosyl bromide NOB1 (0.64 mol) was placed in a 1.00 L flask. When heated it readily decompose...
A: Nitrosyl bromide, NOBr decomposes into nitrogen monoxide, NO, and molecular bromine, Br2. The equili...
Q: Describe molar heat fusion and melting point 2. Study how to solve problem involving molar heat fusi...
A: We have to tell Molar heat of fusion and melting point.
Q: d. lonic compounds tend to 29. What is the couloumbic energy of MgO? ECouloumbe = (231 aJ) 2) %3D a ...
A:
Q: 2 NOBr(g) 2 NO(g) + Br,(g) AH = 16.1 kJ How does the equilibrium shift after the following stresses?...
A: Given that , 2NOBr(g)-----> 2NO(g) + Br2(g)
Q: Vanumber of H ion per moleculeXMa) (Vb)(number of OH - ion(s) per molecule(Mb), then 40.00 mlL of O....
A:
Q: O2 (g) CO: (g) H:O (1) CH6 (1) ->
A:
Q: I structure bE
A:
Q: For the reaction below, which species is the oxidizing agent? H216) + Cl2) → 2 HCl) Cl26) HCl Questi...
A: Oxidising agent: those species which oxidise other and reduce itself. Reducing agent: those species ...
Step by step
Solved in 2 steps with 2 images

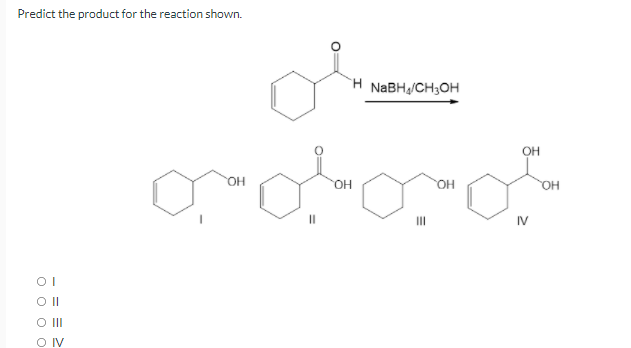
- Indicate the result of the reaction:(3E)-pent-3-en-2-one + 1) LiAlH4 + 2) H3O+Which compounds from 5a-d will react with hydroxide in the fashion shown. Which reaction will be the fastest and slowest? Explain using knowledge of carbonyl chemistry, pka and leaving groups.Show the major products of these reactoions, and please show the complete reaction



