Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
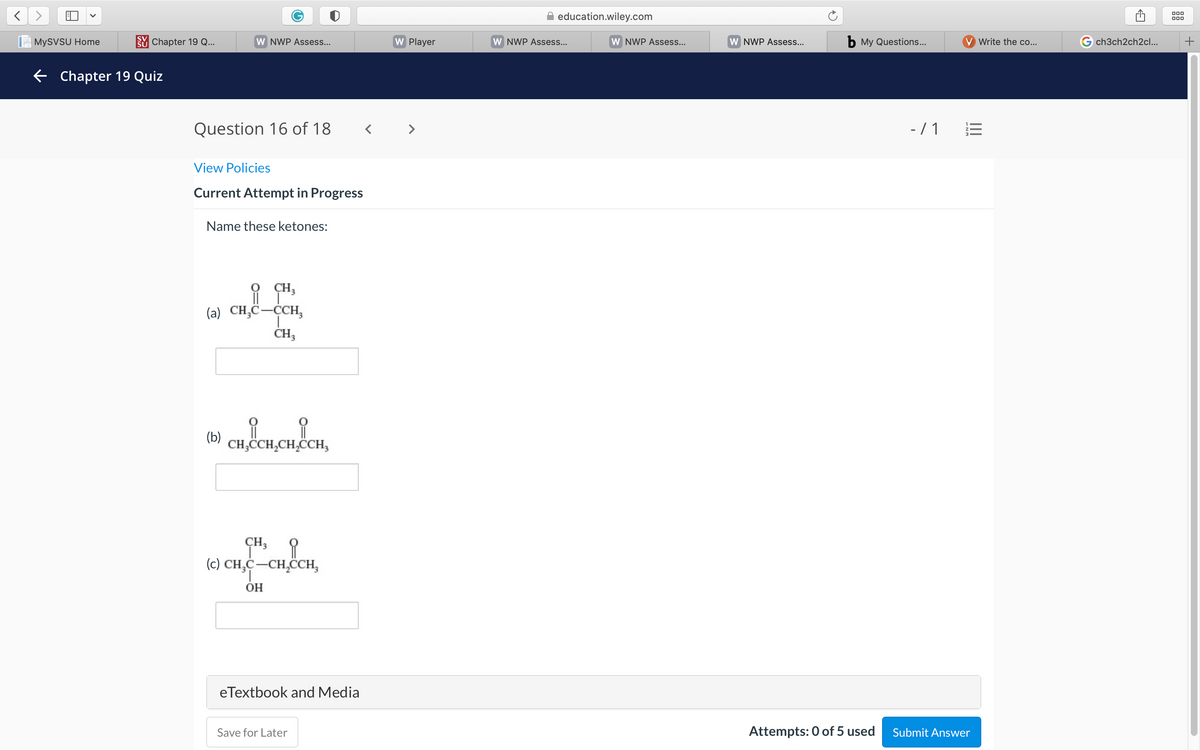
Name these
Transcribed Image Text:education.wiley.com
00
000
MYSVSU Home
SY Chapter 19 Q...
W NWP Asses...
W Player
W NWP Assess...
W NWP Assess...
W NWP Assess...
b My Questions...
V Write the co...
ch3ch2ch2cl...
E Chapter 19 Quiz
Question 16 of 18
>
- / 1
View Policies
ent Attempt in Progress
Name these ketones:
O CH;
(a) CH,C-CCH,
(b)
CH,CH,CH,CCH,
CH3 1
(c) CH,C-CH,CCH,
1.
OH
eTextbook and Media
Save for Later
Attempts: 0 of 5 used
Submit Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

