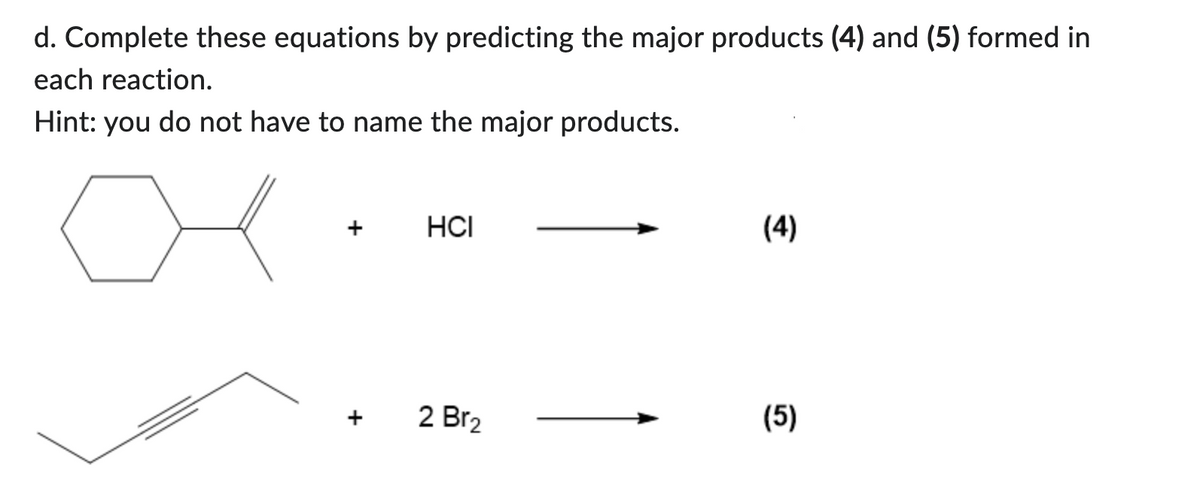
d. Complete these equations by predicting the major products (4) and (5) formed in each reaction. Hint: you do not have to name the major products. + + HCI 2 Br₂ (4) (5)
Q: Determine the mass (in g) of Na2CO3 that contains 5.03 × 1023 O atoms.
A:
Q: compound Q Which of the following is the most stable conformation of compound 1 CH₂ H H- CH3 CH3 'H…
A: Stereochemistry is branch of chemistry in which we deal with three dimensional arrangement of atoms…
Q: The following chemical reaction takes place in aqueous solution: FeSO4(aq)+Na₂S(aq) →…
A: The given reaction is---- FeSO4 (aq) + Na2S (aq) -------> FeS (s) + Na2SO4 (aq) Here reactants…
Q: Question 51 Add bonds (double or triple bonds) and lone pairs to the following incomplete structure…
A:
Q: What is the the equilibrium constant (Kc) for the reaction below, if the reaction mixture initially…
A:
Q: After dissolving NaOH in water, the final concentration of OH is 6.53x104. What is the p solution?…
A: Sodium hydroxide is strong base hence it dissociated fully in aqueous solution
Q: Question 55c Complete these equations by drawing the structure of the missing reactants (2)and (3)…
A: -> Reaction of HI with alkene produce alkyl iodide . -> Reaction of Cl2 with alkene give anti…
Q: The reaction 2HBr(g) ⇒ H₂(g) + Br₂(g) has K = 3.2 × 10-34 at 25 °C. If the reaction vessel initially…
A:
Q: Which one of the following complexes is responsible for the blue color obtained in the reaction…
A: Here we have to predict the reason for blue colourisation when iodate ion and sulphite ion reacts in…
Q: Equal volumes of 0.270 M aqueous solutions of nitrous acid ( HNO₂(aq)) and sodium acetate (NaCH3COO)…
A:
Q: Assuming no systematic errors, within what range are you 90% certain that the true mean value of the…
A: The given molarities of NaOH solution are, 0.4028, 0.4029 , 0.4023 , 0.4031 , 0.4026 , 0.4027 ,…
Q: How many moles of triglyceride do you have?
A: Given : mass = 30.85g Molecular mass = 787.22 g/mol Moles = given mass ÷ molecular…
Q: The reaction 2NO2(g) + F2(g) à 2NO2F(g) has a second order rate law, rate= k[NO2][F2]. Suggest a…
A: Chemical kinetics can be defined as the branch of chemistry that deals with rates of the chemical…
Q: Give the characteristic of a first order reaction having only one reactant. The rate of the reaction…
A: A first order reaction is which has only one reactant. In the question given some statements about…
Q: Consider the following reaction: 2CH3OH(g)→2CH4 (g) + O2(g), AH = +252.8 kJ
A: 2CH3OH(g) -----> 2CH4(g) + O2(g) ∆H = +252.8 kJ
Q: Suppose a 500. mL flask is filled with 0.50 mol of NO₂ and 1.5 mol of NO. This reaction becomes…
A: Answer: In the given problem statement equilibrium of decomposition of NO2 in a 500mL flask is…
Q: Draw the major product of this solvolysis reaction. Ignore any inorganic byproducts. CI CH3CH2OH
A:
Q: , Consider the unbalanced reaction below, which occurs in an acidic aqueous solution. MnO4 + 1 Ⓡ1₂ +…
A: Given -> MnO4- + I- ----> I2 + Mn2+ -> oxidation is process in which oxidation state of…
Q: Pinkman has a sample of 7.15 g apple vinegar. He conducted an acid-base titration on this sample.…
A: Tyyh
Q: 2. The addition of dimethylglyoxime, H₂C4H6O₂N₂, to a solution containing nickel(II) ion gives rise…
A: Given :
Q: 4. For each of i. ii. With reference to oxidation numbers, indicate whether choice. Use oxidation…
A: Redox reaction are those in which both oxidation and reduction reaction occurs simultaneously.…
Q: what products are formed from reaction of HBr with racemic 4-Methyl-1-hexene
A: Here we are required to predict the product of reaction of HBr with racemic 4-Methyl-1-hexene
Q: Gaseous butane CH3CH22CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2…
A:
Q: Chemistry Which of the following sets of quantum numbers (n,l,ml,ms) refers to a 4s orbital? Select…
A: Atom consist of sub atomic particles called electrons, protons and neutrons. Quantum number are…
Q: How do you draw the structure corresponding to the following IUPAC names? 1)…
A: Rule to draw structure-1) First make carbon skelton by using parent name.2) Then after attached the…
Q: 9) For the 2p, orbital, what is the most probable point (r, 8, d) where an e- will be found. R₁, Y₁…
A:
Q: 100 ml of 0.100M H₂A titrated with 0.100 M NaOH (Ka1 = 1.0 E-3; Ka2 = 1.0 E-7) Calculate the pH at…
A: Given data: Volume of H2A = 100 mL or 0.100 L Molarity of H2A = 0.100 M Molarity of NaOH = 0.100 M…
Q: Write a neutralization reaction for each acid and base pair. HI(aq)and NaOH(aq) Express your answer…
A: Since, In neutralisation reaction, Acid react with base to form water and salt known as…
Q: Consider the reaction of phosphoric acid and potassium hydroxide. In the complete ionic equation the…
A:
Q: NO3: Nitrate ion Incorrect
A:
Q: It takes 55.0 J to raise the temperature of an 11.0 g piece of unknown metal from 13.0°C to 24.8 °C.…
A:
Q: 8 Which smells the best? butanoic acid butane sodium butanoate ethyl butanoate They all smell…
A: Three questions based on introduction to organic chemistry that is to be accomplished.
Q: 4. Consider a compound with the formula CNS- a. Draw three possible Lewis dot structures, each one…
A: For the compound CNS- we have to draw three Lewis structures. In the three structures, the central…
Q: A 5.50 mL sample of an H3PO4H3PO4 solution of unknown concentration is titrated with a 1.050×10−2…
A: Given Volume of H3PO4= 5.50 mL Volume of NaOH required= 7.32 mL Molarity of NaOH = 1.050×10-2 M…
Q: Some soluble compounds are listed in the table below. Complete the table by filling in the name or…
A: Types of compound : 1) Ionic compound - The compounds which are formed by the combination of ions…
Q: The equilibrium constant, K, for the following reaction is 1.70×10-² at 608 K. COCI₂(g) →CO(g) +…
A:
Q: Give the IUPAC name for the following compound. H H3C, H H CH3 CH-CH2-CH2-CH3 CH H3C CH3 Select one:…
A:
Q: A buffer solution contains 0.205 M NaHSO3 and 0.405 M K₂SO3. If 0.0321 moles of nitric acid are…
A:
Q: Liquid hexane CH3CH24CH3 reacts with gaseous oxygen gas O2 to produce gaseous carbon dioxide CO2…
A:
Q: When 9.31 mL of 136 mM H2SO4 is added to a 2 L reaction flask that contains 171.31 mg Na(s),…
A: Given that: Volume of H2SO4 = 9.31mL = 9.31mL×1L/1000mL = 0.00931 L Molarity = 136mM =…
Q: Name each of the following alkanes. H3C-CH2-CH2-CH2-CH3 Spell out the full name of the compound.
A: The detailed explanation is given below.
Q: Although we tend to make less use of mercury these days because of the environmental problems…
A: 4 HgS(s) + 4 CaO(s) → 4 Hg(l) + 3 CaS(s) + CaSO4(s) mass of HgS = 10.7 kg
Q: Give the proper IUPAC name for the following. Use E/Z notation if applicable.
A: Rule of IUPAC- 1) Longest chain as parent chain.2) Numbering start from those side where more prior…
Q: A solubility of sugar in water 909 g/L at 25 °C. A student pours 1 kg of sugar into boiling water…
A: Sugar is a solid crystalline compound; when sugar is dissolved in water, solid molecules go into…
Q: For which one of the following reactions is Kp equal to Kc? CaCO3(s) CaO(s) + CO₂(g) 2NH3(g) 3H2(g)…
A:
Q: d. e b. C. a. 5 O 6. Draw at least 3 additional (l.e., in addition to the structure provided) valid…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: Predict the reactants of this chemical reaction. That is, fill in the left side of the chemical…
A: Given -> Products -> Ca(ClO3)2(aq) + H2O(l)
Q: Silver carbonate has a K-5.65x10-12 and a solubility of 0.0001122 M in water. Solid silver carbonate…
A:
Q: 7. Write the products to the following chemical reactions. H3CCH3 1. 2. CH4 4. H C CH₂ CH₂ 0₂ °CH3…
A:
Q: Macmillan Learning Complete the table. Number of electron groups Bond angle Outer atoms/Lone pairs 2…
A: vhh
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- a) Write down the products that will occur when you extract HBr from 2-bromo-3-methyl butane in a basic medium, State the reaction conditions. Show which product is the main product. b) Does the main product show the geometric isomer, so please write together. If it shows write the isomers. c) Write the product that will be formed when the main product reacts with KMnO4 in a basic environment in colda) Write down the products that will occur when you withdraw HBr from 2-bromine-3-methyl butane in a basic medium, State the reaction conditions. Show which product is the main product. b) Does the main product show the geometric isomer, so please write together. If it shows write the isomers. c) Write the product that will be formed when the main product reacts with KMnO4 in a basic environment in cold.Highlight all of the sites where the halogen could be added upon reaction with HBr.
- Was starting material A consumed during the NaBH4 reduction reaction? Brieflyexplain your answer.Can someone explain the answers to 28, 29, and 30? The answers are B, A, A. I understand lindar catalyst makes cis alkenes and Na/NH3 makes trans alkenes but I dont understand how the second step of Br2, OsO4 influences the stereochemistry between the 2 . Will rate quickly if helpful. Thanks!Based on the image attached, i) explain details on the nitration reaction of methyl salicylate. ii)Next, identify the products which one is 3-nitrosalicylate and 5-nitrosalicylate. Explain your answer.
- What are the starting materials, intermediate, and synthetic strategy for the attached molecule? For the synthetic strategy of the molecule, please show the stereochemistry we want to attain through the synthesis. I included an example of how the synthetic strategy should look.Outline the mechanism showing both products formed for the reaction of ammonia (2 mole equivalent) with 1-bromobutane (1 mole equivalent).Please show the machanism for the reaction. Make sure to include how NH2 is deprotonated and HCl formed. Thank you.
- complete this reaction Fe³= ........agent (from-to) S2O3= .........agrnt (from-to).Please propose a synthesis of the target molecule using as many steps or reagents and answer these questions. 1. Why use chemoselectivity as the functional group and not another? 2. Why regioselectivity? 3. Why stereoselectivity? 4. What are the changes in the oxidation state?What are the characteristics of the a good nucleophile for SN2? How does a chemist tell a strong versus weak or bulky versus not bulky nucleophile? What is the best type of solvent for this reaction? Please define the terms. Thank you.

