Q: Consider the reversible formation of the Cu(NH3)42+ complex ion. Cu²+ (aq) + 4NH3 (aq) = Cu(NH3)4²+…
A: Given Consider the reversible formation of the Cu(NH3)4 +2 complex ion. Cu +2 (aq) + 4NH3 (aq)…
Q: Which one of the following changes could double the rate of the reaction 2 A(g) + B(g) + 3 C(g) → 3…
A: Given, 2A(g) + B(g) + 3C(g) ----> 3D(g) + E(g) Rate law expression for the above reaction, Rate =…
Q: Study the following sketch of a molecular orbital (MO) in a homonuclear diatomic molecule. This MO…
A: Given =molecular orbital diagram is given. we have to find name of it and its energy.
Q: 3. gon ata CI lourd AICI3
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: The half-life for the second order reaction A → Products is 4.57 min. What is the value of the rate…
A:
Q: P-V work (w = -PΔV) occurs in the following system. Assuming all gases are ideal such that PΔV =…
A: Given: Temperature = 296 K R = 8.314 J/mol·K w = -PΔV
Q: A mixture of 87.3 mole % methanol and 12.7 mole % isopropyl alcohol is vaporized at 101.325 kPa…
A:
Q: Consider the following chemical equilibrium: H₂(e) + Co, (e) H₂0(e)+CO(e) Now write an equation…
A: Answer: KP represents the value of equilibrium constant for pressure of gases at equilibrium while…
Q: Complete the reactions below
A: Given that, We have to complete the above given reactions. Introduction: Nucleophilic substitution…
Q: A solution contains 1.21×10-2 M calcium acetate and 5.84×10-³ M silver nitrate. Solid sodium…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: Under what conditions will enthalpy tend to dominate the equilibrium state of a chemical system?…
A: The dominance of enthalpy or entropy in determining the equilibrium state of a chemical system…
Q: How much heat in kJ is required to convert 24.3 grams of ice at -8.2 Celsius to steam at 100.0…
A: 24.3 grams of ice is heated from - 8.2 °C to steam at 100.0 °C.We have to determine the amount of…
Q: Give the structure of the major product of the following reactions a. (CH3)2CHCOCH₂CH3 + N₂H4 + KOH…
A: Different sets of substrate gives different products when reacted with different reagents. And thus…
Q: Indicate which one of the metals should be the anode in all reactions. Choose one: O Al(s) O Ni(si O…
A: A question based on electrode potential. 5 options of metal are given from that the most appropriate…
Q: Solid cobalt(II) acetate is slowly added to 150 mL of a 0.0653 M potassium hydroxide solution. The…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: Each of the insoluble salts below are put into 0.10 M hydrobromic acid solution. Do you expect their…
A: The question is based on the concept of Le-chatlier principle. It States that when a system at…
Q: Calculate the pH in the titration of 100.0 mL of 0.20 M HClO (Ka = 4.0 x 10-8) with 0.20 M KOH after…
A: Given , Ka=4.0×10-8
Q: Rank the samples of gas described in the table below in order of increasing average speed of the…
A: The average speed of the molecules is given by the formula, vavg=8RTπM T is the temperature in…
Q: If you complete and balance the following equation in acidic solution Mn²+ (aq) + NaBiO3(s) → Bi³+…
A: Balance chemical equation: Balance chemical equation can be define as the reaction in which number…
Q: You are required to produce a digital poster to answer the following questions based on the compound…
A:
Q: Identify the relationship H HO CH3 H3C Br -I H between the following compounds. H H3C. во фон Br -I…
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
Q: If the pH of a solution is 4.5, what is the [H3O*] of the solution? Hint [H3O+] = 10-PH 90.0 M…
A:
Q: Give the organic family the compound shown here belongs to. NH₂ C-C-C-C-C-C-C The organic family the…
A: The organic compound given contains –NH2 group.
Q: How much ammonia is evolved when 34.0 grams of ammonium chloride are added to 37.0 grams of…
A:
Q: 13 Write and name the other product of each of the following reactions:…
A:
Q: A chemical engineer is studying the following reaction: N₂(g) + 3H₂(g) → 2NH₂(9) At the temperature…
A: The reaction involved is N2 (g) + 3H2 (g) <-----> 2NH3 (g) Kp of the reaction = 0.0043…
Q: Please indicate the reagents and the mechanism to form this product (synthesis problem) H₂W
A: The question is based on organic synthesis. we need to synthesize the product from the starting…
Q: 5. This reaction has been allowed to reach equilibrium, 2 NH, (g) = N₂ (g) + 3 H₂ (g) Then…
A:
Q: A chemical engineer is studying the following reaction: HCH3CO₂(aq) + CH3NH₂(aq) → CH3CO₂(aq) +…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: 2. Benzoic acid is a weak acid. Which of the following solutions will have the highest pH? O a.…
A: The highest pH solution among the given options will be b. 0.0010 M benzoic acid in 0.0025 M sodium…
Q: 8. -O-K* -OH O₂N. H₂SO4 NH NaHCO3 H3C CN M-Z hv
A: Given that, We have to complete the above given reactions. Introduction: Nucleophilic substitution…
Q: Assuming 100% dissociation, calculate the freezing point (1) Colligative constants can be found in…
A: Kf of water = 1.86 K kg mol-1 Kb of water = 0.512 K kg mol-1 Van't Hoff factor (i) of AgNO3 = 2…
Q: A strong absorption of infrared radiation is observed for ¹H³5Cl at 2991 cm-1 a) Calculate the force…
A: Infrared spectroscopy comes under vibrational spectroscopy. From the value of the wave number one…
Q: The rates of heterogeneous reactions typically depend on the solid and a liquid. surface area…
A: We know, there are two types of reactions - Homogeneous and Heterogeneous Reactions.
Q: A hydrocarbon (a compound that contains carbon and hydrogen only) contains 82.8% carbon and its…
A:
Q: Calculate the enthalpy change, ΔH , for the process in which 21.1 g of water is converted from…
A:
Q: 1. What reagent(s) are needed to complete the following 0 1) 2) A) 1) LIAIH4, 2) H3O+ HO. B) 1)…
A: two reaction are given , we have to tell missing reagent and product
Q: Consider the insoluble compound silver hydroxide, AgOH. The silver ion also forms a complex with…
A:
Q: 4. [25'] Consider the reaction 2NO₂ (g) → N₂O₂ (g) a) Calculate the value of AG at 298.15 K. b)…
A: ∆G° of a reaction can be calculated by taking the difference of ∆G° of product and ∆G°of reactant.…
Q: 1. Determine the weight of KCIO3 heated. 2. Determine the weight of oxygen gas lost using the…
A: In the presence of a MnO2 catalyst, KClO3 decomposes to form potassium chloride and oxygen gas on…
Q: How many joules of heat does it take to melt 117 grams of ice that has an initial temperature of…
A: To solve this problem, I used the concepts of specific heat, heat of fusion, and the calculation of…
Q: In the Heat of Neutralization experiment, we will measure the heat of reaction (ΔH) for the…
A:
Q: 16-57 Starting with either benzene or toluene, how would you synthesize the following substances?…
A: We have to synthesized the below given compounds from benzene or toluene. 2-Bromo--4-nitrotoluene…
Q: What mass of aluminum (in g) would be required to completely react with 1.15 L of 0.350 M HCI in the…
A:
Q: Show that for a reaction that obeys the general rate law, rate = K[A]" a graph of log(rate) versus…
A: The given data shows that at the start of the reaction, the concentration of SO2Cl2 is 0.1000 M. The…
Q: 4. For the reaction, PC13 (4) + Cl₂ (g) = PCI; (s) (A) [PC3][Cl₂]/[PCla] what is the expression for…
A: Note: Here, I am answering question number 4 as you have asked. Equilibrium constant expression…
Q: Tartaric acid is the white, powdery substance that coats tart candies such as Sour Patch Kids.…
A:
Q: Sketch the ultraviolet absorption spectrum of 1,3-Butadiene, matching peaks to electronic…
A:
Q: Anhydrous CuCl2 (0.12g) and triphenylphosphine oxide Ph3PO (0.5g) are reacted together to form what…
A: The reaction between anhydrous copper(II) chloride (CuCl2) and triphenylphosphine oxide (Ph3PO)…
Q: At 500 K, hydrogen and iodine can form hydrogen iodide in the gas-phase reaction: 2HI(g) H₂(g)…
A: The equilibrium constant (K) is a mathematical representation of the ratio of concentrations of…
Predict the product(s) of the following reactions:
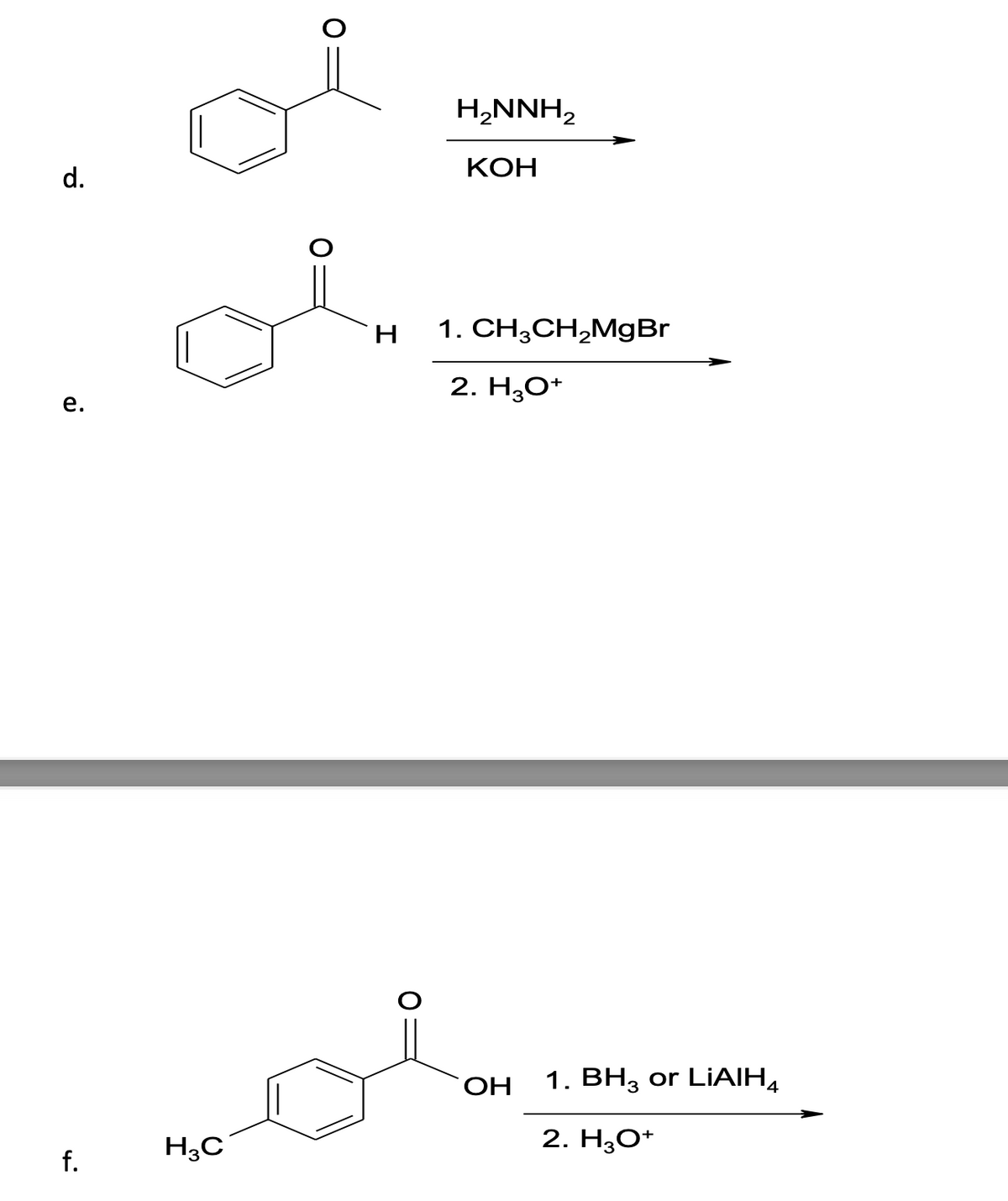
Step by step
Solved in 4 steps with 4 images


