d. If the chemical reaction uses 56.2 grams of MgCl2, then how many moles of Mg metal will be produced? e. If the chemical reaction uses 4.2 x 103 grams of Al metal, then how many moles of MgCl2 will be needed to fully react all of the Al metal? f. If the chemical reaction produces 25.3 grams of Mg metal, then how many moles of AIC13 will also be produced? g. If the chemical reaction produces 2.19 x 102 grams of AlCl3, then how many moles of MgCl2 were needed? h. If the chemical reaction produces 8.33 x 102 grams of AIC13, then how many moles of Mg metal were needed? If the chemical reaction produces 119.2 grams of Mg metal, then how many moles of Al metal were needed?
d. If the chemical reaction uses 56.2 grams of MgCl2, then how many moles of Mg metal will be produced? e. If the chemical reaction uses 4.2 x 103 grams of Al metal, then how many moles of MgCl2 will be needed to fully react all of the Al metal? f. If the chemical reaction produces 25.3 grams of Mg metal, then how many moles of AIC13 will also be produced? g. If the chemical reaction produces 2.19 x 102 grams of AlCl3, then how many moles of MgCl2 were needed? h. If the chemical reaction produces 8.33 x 102 grams of AIC13, then how many moles of Mg metal were needed? If the chemical reaction produces 119.2 grams of Mg metal, then how many moles of Al metal were needed?
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter11: Stoichiometry
Section: Chapter Questions
Problem 75A
Related questions
Question
100%
3MgCl2+2Al—>3Mg+2AlCl3
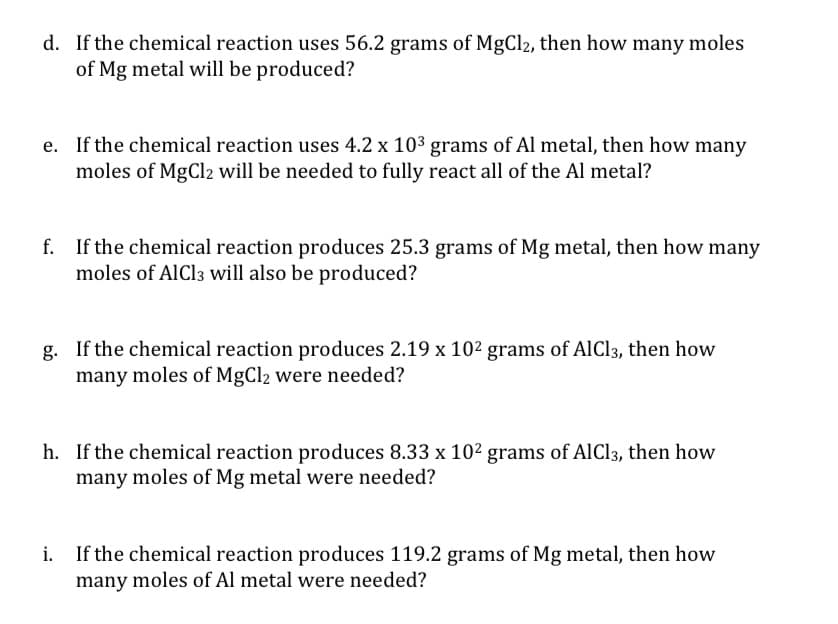
Transcribed Image Text:d. If the chemical reaction uses 56.2 grams of MgCl2, then how many moles
of Mg metal will be produced?
e. If the chemical reaction uses 4.2 x 10³ grams of Al metal, then how many
moles of MgCl2 will be needed to fully react all of the Al metal?
f. If the chemical reaction produces 25.3 grams of Mg metal, then how many
moles of AlCl3 will also be produced?
g. If the chemical reaction produces 2.19 x 102 grams of AlCl3, then how
many moles of MgCl2 were needed?
h. If the chemical reaction produces 8.33 x 102 grams of AlCl3, then how
many moles of Mg metal were needed?
If the chemical reaction produces 119.2 grams of Mg metal, then how
many moles of Al metal were needed?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning