Q: Answer the following questions about a titration between 25.0 mL of a weak acid (Ka= 6.2*10-5) and 0...
A: The question is based on the concept of titration of weak acid and strong base. We have to calcula...
Q: Which property is shared by both acids and bases A) changes red litmus paper to blue B) feels slip...
A:
Q: can you help with product confirmation of rx?
A: A multiple choice question based on dehydration reaction, which is to be accomplished.
Q: 9. ABCI (s) #Ag' (aq) + Cl¯ (aq)? Addition of KCI (s) will shift the equilibrium in the backward dir...
A:
Q: H2O, H* What is the major product?
A: Acidic water helps in nucleophilic addition reaction, which gets attack at double bond.
Q: Which of the following, is an example of an endothermic reaction? 2Na + Cl2--> 2N2CI e C,H1206 + 602...
A: The reactions given are, 1) 2 Na + Cl2 -----> 2 NaCl 2) C6H12O6 + 6 O2 -----> 6 CO2 + 6 H2O 3)...
Q: Answer the following questions about an aqueous solution of tetrahydrofuran (C4H8O)with a concentrat...
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you. ...
Q: A solution consists of 0.450 mole of pentane, C5H12, and 0.250 mole of cyclopentane, C5H10 . What is...
A: A solution contains 0.450 mole of pentane and 0.250 mole of cyclopentane. Mole fraction of pentane =...
Q: H2SO4 solution was diluted with 475.0 mL of water? b.lf 20.0 mL of 0.0100 M AgNO3 reacts with excess...
A: Determination of no. of mol of H2SO4: Determination of concentration of final solution of H2SO4: ...
Q: Which atomic transition would release more energy in a hydrogen-like Aluminum atom (Z = 12): Transit...
A: A question based on Rydberg equation, which is to be accomplished.
Q: topic: catalyst affects the reaction rate Compare and contrast the two types of catalyst (homogenous...
A: Homogeneous catalyst1- The reactants and also the catalyst are present within the same phase.2- They...
Q: A compound X is to be determined by UV/Visible spectrophotometry. A calibration curve is constructed...
A: The light absorbed by the solution is known as absorbance.
Q: The time needed to plate out 1.00 g Ni(Ni2+) using a 15-amp current is about 3.7 minutes 7...
A: Mass of Ni(Ni^2+)=1.00g Current=15amp Time=?
Q: Q4. Rank the strength of the following acids based on the pH from strongest to weakest. A B D pH 0.5...
A: A question based on strength of acids, which is to be accomplished.
Q: Answer the following questions about the transition metals complexes with the chemicalformulas [Fe(H...
A: The answer to these above questions are as follows;
Q: The compound below undergoes an intramolecular Aldol condensation reaction to form ONE MAJOR PROUDUC...
A: In aldol reaction when an aldehyde or ketone have alpha Hydrogen reacts with any base it gives α, β-...
Q: Determine the number of atoms in a sample of 2.26 g Hg. O 6.78 x 10 21 atoms O7.53 x 10 22 atoms O 2...
A: Number of moles of a chemical element or a compound is used to measure the amount of element or comp...
Q: In an electrolysis experiment, a student passes the same quantity of electricity through two electro...
A:
Q: The gas-phase basicity of triphenylamine (876.4 kJ mol-1) is less thanthat of triphenylphosphine (94...
A: Given that The gas-phase basicity of triphenylamine is 876.4 kJ mol-1 The gas-phase basicity of ...
Q: What is the concentration, in mass percent (m/m %), of a solution prepared from 100. g NaCl and 150....
A:
Q: A helium filled weather balloon has a volume of 502L at 14.9 degrees Celsius and 751mmHg. It is rele...
A: From the combined gas law we know , (P1V1)/T1 = (P2V2)/T2 Where P1 = Initial pressure of system P2 =...
Q: 5
A: Ideal gas equation : PV = nRT P = 1.12 atm T = 200C = 293.15 K
Q: A sample of argon gas collected at a pressure of 546mmHg and a temp of 277k has a mass of 37.0 grams...
A: Pressure =546 mmHg=(546/760) atm=0.718atm Temperature =277 K Mass= 37.0 grams Volume=?
Q: On what events where the result of the gel electrophoresis may not be believable or invalid.
A: Gel electrophoresis is a technique used for DNA (deoxyribonucleic acid) analysis. In this technique,...
Q: Give the IUPAC name for each compound.
A: Alkanes are organic compounds containing carbon and hydrogen. These are very inert as there is only ...
Q: Determine the heat of vaporization of H2O: H2O(liq) → H2O(g) given the enthalpies of reaction bel...
A: Hess’s law states that when chemical equations are combined algebraically, their enthalpies can be c...
Q: Hannah and Matthew are studying the Arrhenius reaction below. They labeled the substances as follows...
A: Explanation to the correct answer is given below.
Q: 2. Which is the most acidic hydrogen in the following compound? Provide a brief explanation. H H- H....
A: Most acidic hydrogen is that form most stable conjugate base. Conjugate base is stabilized by -R an...
Q: Melting occurs in going from region VII V VI 1 atm I II IV III O III to II. I to II. OI to III. IIl ...
A: The given phase diagram can be used to predict different phase transition processes. Some of them ar...
Q: I need help with this
A:
Q: Pu-242 would be used for fusion reactors in nuclear fission in electrolytic cells ...
A: Pu-242 is an Isotope of plutonium
Q: H* Кey A- H* A- H* Dissociated H* ion A- H* H* A Dissociated A ion H* A- A- A H НА Does the diagram ...
A:
Q: How much heat energy is needed to melt 750.0 grams of ice. it needs to be in kj
A: We have to calculate how much heat energy is needed to melt 750 grams of ice.
Q: Describe how would you distinguish the folla (a) Benzene and cyclohexane (b) Phenol and toluene (c) ...
A: Organic compound is large class of chemical compounds. In this atoms of carbon are covalently linked...
Q: 33. A radioisotope is all the following except an isotope with different number of neutrons fr...
A: An isotope with different number of neutrons from others.This is true about radioisotope. Isotopes ...
Q: 2.2CO + O2 → 2CO2 Carbon monoxide will burn in air to produce CO2 according to the following equati...
A: First find the moles of CO from given information. And we know that , 1 mol of any gas contains 22.4...
Q: What is the mass (in grams) of an unknown gas (molecular weight = 295 g/mol) that is contained in a ...
A:
Q: Nbalance Za3L2 + Nf2QU ----> ZaQu + Nf3L Za= +2 QU= polyatomic -2 Nf= +1 metal L= nonmetal -3 cha...
A: To balance the equation, Za3L2 + Nf2QU ----> ZaQu + Nf3L
Q: A sample of cleansing powder weighing 4.0 grams was diluted with water and filtered to remove the su...
A: Given that: total mass of cleansing powder = 4.0 g volume of HCl = 35 mL = 0.035 L molarity of HCl ...
Q: 20. The Coordination & Oxidation numbers for Copper in K2[CuC14] respectively are:_ & (K has +1 and ...
A: The coordination number defines the number of atoms ,ions, or molecules that a central atom or ion h...
Q: Suppose 26.0 mL of 0.100 M HCl(aq) were spent to completely neutralize 10.0 mL of an ethylamine solu...
A: M1V1 = M2V2 26*0.100=M2 *10.0 M2 = 26.0x0.100/10.0 =0.26Molar is the molarity of ethylamine solution...
Q: 6. Positron (+10B) emission of a large isotope can give increased nuclear stability by: a) increasin...
A: Answer:- the emission of positron particle results in the decrease of number of protons by one unit ...
Q: A polymer has an overall density of 1.056x103 kgm-3. The density of the crystalline phase is 1.12x10...
A: Percentage crystallinity is the quantification of heat associated with melting of sample polymer.
Q: When a sample of aqueous hydrochloric acid was neutralized with aqueous sodium hydroxide in a calori...
A: The amount of heat required to raise the temperature of the sample is given by: q = mc∆T where 'q' i...
Q: 17) What type of amino acid is tyrosine? A) Polar neutral B) Nonpolar ncutral C) Acidic D) Basic
A: So we will determine the type of amino acid tyrosine is! Why tyrosine belongs to that catagory.
Q: Which of the following is NOT a unit of pressure? О А. psi о В. atm C. pascal D. candela O F. torr
A: Units of measurement are defined as the fixed magnitude or quantity that can be defined and adopted ...
Q: AsAP in clearly. Thank you
A: The details solution for this reaction is provided below in attach image.
Q: Please answer completely will give rating surely
A: Acetoacetic ester is an ester compound. It contains an active methylene group between the two carbon...
Q: at 300k which of the following gases would be expected to have the greatest average kinetic energy?
A: Average kinetic energy of per mole of gas is given by the following equation. K.E.=(3/2)RT where ...
Q: 4) In order to form an octet, an atom of nitrogen will: A) lose 6 electrons B) gain 3 electrons C) l...
A:
Provide a reasonable synthesis for each of the following using the provided starting material and any
other organic or inorganic reagents.
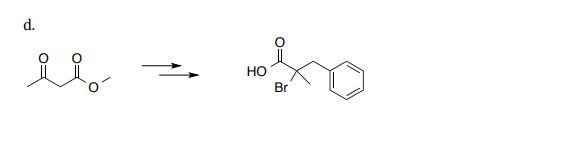
Step by step
Solved in 3 steps with 3 images


