Daclatasvir is formulated at the dihydrochloride salt. Which functional group in Daclatasvir will be protonated? (structure shown below) OA Carbond
Daclatasvir is formulated at the dihydrochloride salt. Which functional group in Daclatasvir will be protonated? (structure shown below) OA Carbond
Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 7.6P
Related questions
Question
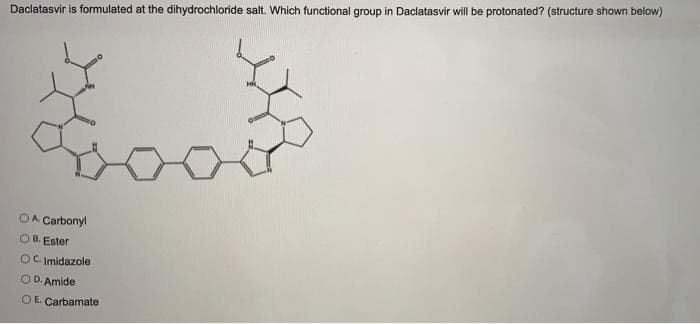
Transcribed Image Text:Daclatasvir is formulated at the dihydrochloride salt. Which functional group in Daclatasvir will be protonated? (structure shown below)
OA Carbonyl
B. Ester
OC Imidazole
OD. Amide
O E. Carbamate
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

