Dariana Using the Arrhenius equation to calculate Ea from k versus T da... The rate constant k for a certain reaction is measured at two different temperatures: temperature 2.7 x 109 311.0 C 7.3x 109 444.0 C Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy Efor this reaction Round your answer to 2 significant digits. kJ E mol X
Dariana Using the Arrhenius equation to calculate Ea from k versus T da... The rate constant k for a certain reaction is measured at two different temperatures: temperature 2.7 x 109 311.0 C 7.3x 109 444.0 C Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy Efor this reaction Round your answer to 2 significant digits. kJ E mol X
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter12: Kinetics
Section: Chapter Questions
Problem 63E: Hydrogen iodide, HI, decomposes in the gas phase to produce hydrogen, H2, and iodine, I2. The value...
Related questions
Question
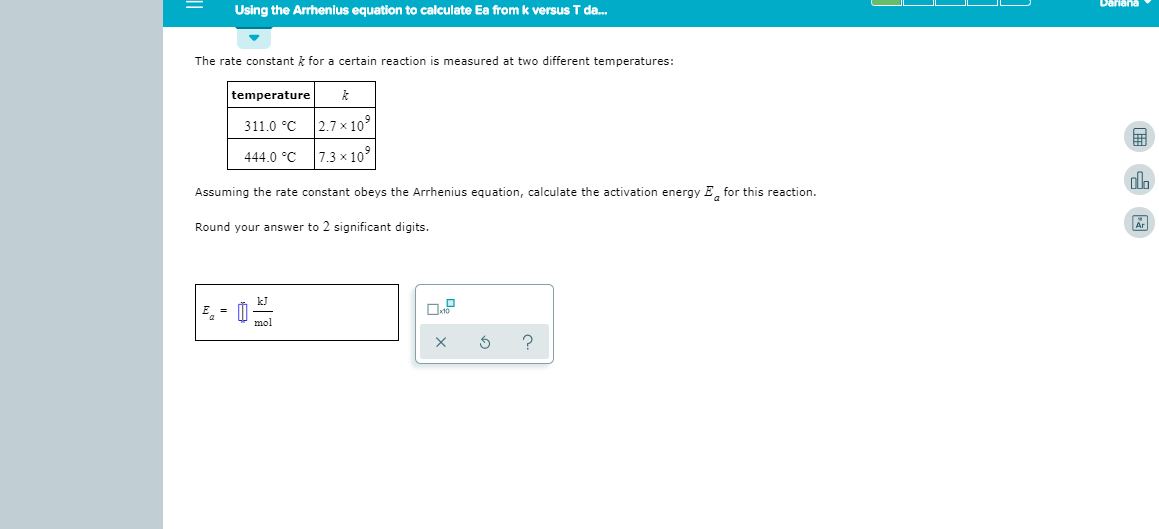
Transcribed Image Text:Dariana
Using the Arrhenius equation to calculate Ea from k versus T da...
The rate constant k for a certain reaction is measured at two different temperatures:
temperature
2.7 x 109
311.0 C
7.3x 109
444.0 C
Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy Efor this reaction
Round your answer to 2 significant digits.
kJ
E
mol
X
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning