Data Sheet sample calculation for the solubility of KNO, using your actual mass from tube #1. Complete gives the recommended mass of salt to dissolve in each of the four test tubes. Show a your graph on the back of this sheet. Solution # Recommended mass KNO, Solubility Actual mass of KNO, (g per 5 ml. H₂O) Saturation. temperature 8 KNO, (g) 100 g H,O (°C) 1 2.0 2.1971 51.3 2 4.0 11.1568 62.5 3 6.0 6.0112 72.7 83.6 4 8.0 8.06 Sample Solubility Calculation: Questions 1. According to your data, how does the solubility of KNO, change with increasi- temperature? Does this indicate an endothermic or exothermic dissolving process? Explain. 2. Using your graph, indicate if each of these solutions would be saturated or unsaturated: a) 110 g of KNO, in 100 g of water at 40 °C b) 60 g of KNO3 in 100 g of water at 70 °C c) 140 g of KNO3 in 100 g of water at 60 °C According to your graph, what mass of KNO3 will dissolve in 100 g of water at 30 °C?
Data Sheet sample calculation for the solubility of KNO, using your actual mass from tube #1. Complete gives the recommended mass of salt to dissolve in each of the four test tubes. Show a your graph on the back of this sheet. Solution # Recommended mass KNO, Solubility Actual mass of KNO, (g per 5 ml. H₂O) Saturation. temperature 8 KNO, (g) 100 g H,O (°C) 1 2.0 2.1971 51.3 2 4.0 11.1568 62.5 3 6.0 6.0112 72.7 83.6 4 8.0 8.06 Sample Solubility Calculation: Questions 1. According to your data, how does the solubility of KNO, change with increasi- temperature? Does this indicate an endothermic or exothermic dissolving process? Explain. 2. Using your graph, indicate if each of these solutions would be saturated or unsaturated: a) 110 g of KNO, in 100 g of water at 40 °C b) 60 g of KNO3 in 100 g of water at 70 °C c) 140 g of KNO3 in 100 g of water at 60 °C According to your graph, what mass of KNO3 will dissolve in 100 g of water at 30 °C?
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter14: Chemical Equilibrium
Section: Chapter Questions
Problem 94AP
Related questions
Question
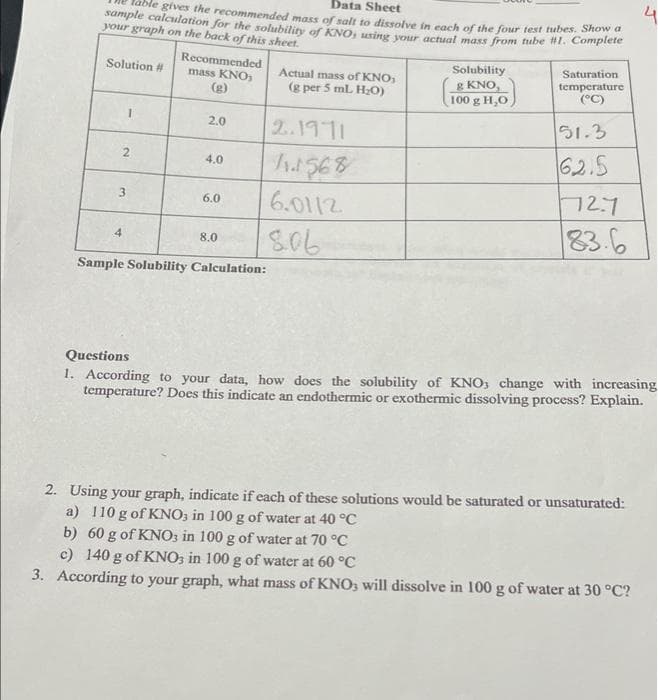
Transcribed Image Text:Data Sheet
4
ble gives the recommended mass of salt to dissolve in each of the four test tubes. Show a
sample calculation for the solubility of KNO, using your actual mass from tube #1. Complete
your graph on the back of this sheet.
Solution #
Recommended
mass KNO3
Actual mass of KNO,
(g per 5 mL H₂O)
Solubility
& KNO,
100 g H₂O
Saturation
temperature
(°C)
(g)
1
2.0
51.3
2.1971
2
62.5
4.0
11.1568
3
72.7
6.0
6.0112
83.6
4
8.0
806
Sample Solubility Calculation:
Questions
1. According to your data, how does the solubility of KNO, change with increasing
temperature? Does this indicate an endothermic or exothermic dissolving process? Explain.
2. Using your graph, indicate if each of these solutions would be saturated or unsaturated:
a) 110 g of KNO3 in 100 g of water at 40 °C
b) 60 g of KNO3 in 100 g of water at 70 °C
c) 140 g of KNO3 in 100 g of water at 60 °C
3. According to your graph, what mass of KNO3 will dissolve in 100 g of water at 30 °C?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning