Q: Direction: Balance the following chemical reactions. 7. COCl2 + H2O ->HCl + CO28. CS2 + O2 ->C...
A:
Q: All of the following statements are true EXCEPT || A. gamma emission can accompany nuclear decay, su...
A: Hi, since you have posted multiple questions we will answer the very first question for you. Kindly ...
Q: In each of the following sets of elements, indicate which element shows the most active chemical beh...
A: Active chemical behavior means which reacts very fastly either by losing electrons or by gaining ele...
Q: The Ksp for In(OH)3 is 1.3 x 10-37. ...
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you. ...
Q: Which of the following reactions would have the most positive AS° value?
A: Entropy is defined as degree of randomness or disorderness and to get the most positive value of ent...
Q: Draw a curved arrow mech. Start with removing the OPP and constructing a ring.
A: Please find the mechanism below.
Q: 1 of 20 An amino acid is dissolved in solution at a pH which is the same as its isoelectric point Mo...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: The Ksp for In(OH)3 is 1.3 x 10-37. ...
A: Solubility of the substance is the maximum amount of solute that can be dissolved in a given amount ...
Q: (а) The reaction of (R)-2-bromopentane with NaOH in ethanol produced a racemic mixture. Br H.. NaOH ...
A:
Q: If water were a linear molecule, (a) would it still be polar, and (b) would the water molecules stil...
A: answer
Q: In this reaction, which compound is the Bronsted Lowry acid? NO, + CH,OH, HNO, + CH,он
A:
Q: For the reaction CO(NH2)2 +H2O(l) -> CO2(g) + 2NH3(g) The ∆S^2 and ∆H^2 is 354.8KJ/mol and 119.2K...
A: Dear student I have given answer to your question in the image format.
Q: What is the [H+] concentration in a solution with a pH of 4.9
A: The pH is defined as the negative log to base 10 of concentration of hydronium ion. The Hydronium i...
Q: Why should perfume bottles be stored tightly capped and away from heat and direct sunlight?
A: Evaporation is the process by which water changes from a liquid to a gas or vapor. The process in wh...
Q: 1. Which of the following is not the unit of concentration? a. Mole/m³ b. Molar c. N/m³ d. pmm 2. W...
A: Since we only answer one question at a time, we'll answer the first one. Please resubmit the questio...
Q: What are the pertinent chemical equations in the standardization of sodium thiosulfate solution?
A: We'll answer the first question since the exact one wasn't specified. Please submit a new question s...
Q: Find the molecular formula of the following compounds 2. Cortisol is a steroid hormone that is used ...
A:
Q: Calculate the standard entropy change for the reaction given below, 2 H202 (1) – 2 H20 (1) +
A: Question in image first will be solved only.
Q: Please answer the question with explanations and clarity
A:
Q: 4NH, (g) + 50, (g) -4NO (g) + 6H;0 () how many moles of excess reactant will be left when 4.0mol NH,...
A: The given reaction may be written as: 4NH3(g)+5O2(g)⇌ 4NO+6 H2O(l) The above given reacction is an r...
Q: A. Predict the major product of the reaction. CI Ph (РСС) OH CH,Cl2 B. Draw a detailed arrow-pushing...
A: Pyradinium chlorochromate (PCC) is an oxidizing agent which oxidizes alcohol to aldehyde and ketones...
Q: help
A: CH2I2 + Zn(Cu)
Q: Calculate the pH of a buffer solution prepared by dissolving 0.400 mol of cyanic acid (HCNO) and 0.8...
A: Given Number of mole of HCNO Acid = 0.400 mole Number of mole of sodium cyanate Salt = 0.8...
Q: Which of the following structures have 1+ formal charge on the central atom? H H Н: Вe: H н:B:Н Н:N:...
A:
Q: Electron transtons teas place in excilbd hydrogen atoms One such trangition is s n-2 to m7 aoro Se c...
A:
Q: Which of the following molecule(s) belong to CooV point group? 1. CO 2. H2 3. HCI 4. 02 * O 1 and 3 ...
A: We have to ask which of the molecule belong to C∞V point Group 1. CO 2. H2 3. HCl 4. O2 An...
Q: Chemistry Question
A:
Q: What is the solubility product of calcium hydroxide?
A: Solubility is defined as the property of the substance to get dissolved in the solution. Solubility ...
Q: Please answer the question with explanations and clarity
A: Williamson's ether synthesis :- Alkyl halides on reaction with sodium alkoxide forms ethers. R-X + R...
Q: help
A: Addition of deutirium to alkene is cis (or) syn addition Cis-compound →cis addition Meso ...
Q: When a 22.9 ml. sample of a 0.347 M aqueous nitrous acid solution is titrated with a 0.309 M aqueous...
A: The volume of the sample = 22.9 mL The molarity of the sample = 0.347 M The molarity of Ba(OH)2 ...
Q: Please answer the question with explanations and clarity
A: Williamson ether synthesis in presence of base
Q: 1.00 mole of a ydW gas goes from 300 K and 1 atm to 500 K and 25 atm. What would be its AS, AH and A...
A: Internal energy is the amount of energy contained in the thermodynamic system. It was represented by...
Q: What is the secondary criteria pollutant according to NAAQS ?
A: NAAQS stands for national ambient air quality standards. In the United States, the allowed levels of...
Q: How many minutes are required to deposit 1.42 g Cr from a Cr³⁺(aq) solution using a current of 2.50 ...
A: Given: W = 1.42 gm I = 2.50 A Mol. Wt. of Cr = 52 'n' factor of Cr = 3 Equivalent wt. of Cr = 5...
Q: Methyl isocyanate, CH3NCO, is used in the manufacture of insecticides, such as carbaryl (Sevin). In ...
A: Answers are explained below.
Q: Find the molecular formula of the following compounds 9. A class of compounds called sodium metaphos...
A:
Q: Identify the most acidic proton below
A: The methylene group attached to the electron-withdrawing carbony group is called the active methylen...
Q: Heat Calculations Q=(M) (C) (AT) A 42.0 g piece of metal has a heat content of 600 Joules. The initi...
A: These type of questions are solved with the help of formula: q=mc(T2-T1) If q is negative, it means ...
Q: A mixture of gases contained only three types of gases (Ne, Ar and He) has a total pressure of 0.900...
A: A mixture of gases is one of the properties of the gas when they do so, they become a solution a hom...
Q: Write the required compounds in the places where there are questions in the reactions below.
A:
Q: Refer to Figure 2 on the cover sheet. What is the Electron Geometry, the Molecular Geometry and the ...
A: ANSWER IS DISCUSSED BELOW :
Q: 2. Which is a false statement regarding non-competitive enzyme inhibitor? (a) It binds to an alloste...
A: 2.) In non-competitive inhibition, the inhibitor does not bind on the active site, it rather binds o...
Q: If you decrease the volume of a system containing the following reaction at equilibrium, identify wh...
A: The direction of equilibrium can be predicted on the basis of Le-chatlier principle.
Q: Please answer the question with explanations and clarity
A:
Q: Calcium hydroxide is sometimes used in water treatment plants to clarify water for residential use. ...
A: We have Calcium hydroxide as 0.025 M, Aluminium sulphate as 0.125 M of 25 ml. We have to calculate t...
Q: A sample of gas occupies a volume of 625 ml at 22°C and 1.12 atm. a) Calculate the new pressure in a...
A: Ideal gas law is also called the general gas equation. It is the equation that represents the hypoth...
Q: Help me please
A: In the presence of a strong base, E2 elimination reaction take place, it will abstract proton from t...
Q: Write the applications of the glass electrode. (Please write it correctly and concisely.)
A: Applying concept of glass electrode.
Q: A. Draw a reaction coordinate diagram that is consistent with the shown chemical equations and obser...
A: Answers are explained below.
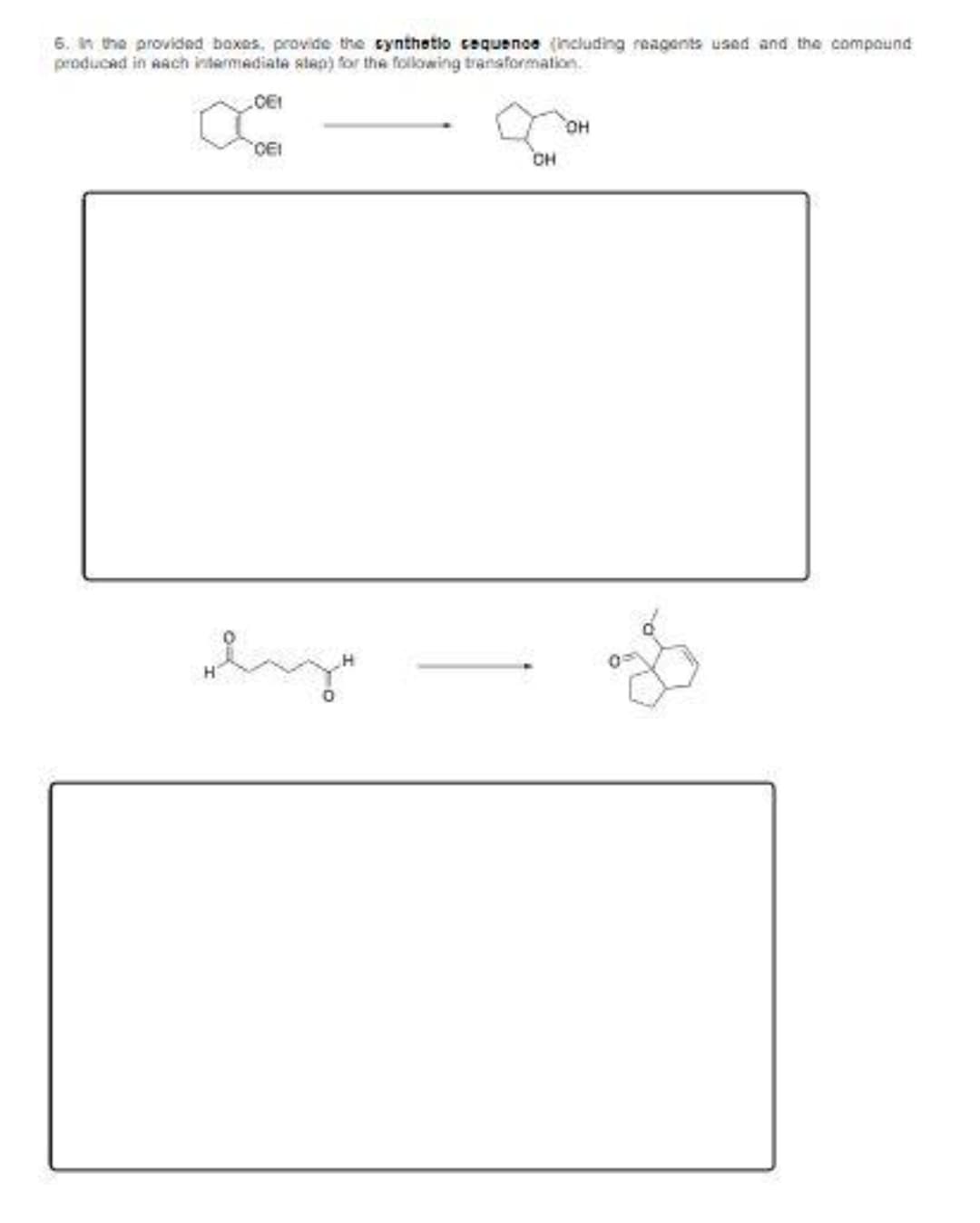
Step by step
Solved in 8 steps with 8 images

- What type of sigmatropic rearrangement is illustrated in each reaction?For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Choose one from each parenthesis. Based on the reactions below, the probable structure of alkene 1 is (BAT / MAT / SAT / FAT) while the process using Reagent1 is (reaction via acidic KMnO4 / reaction via cold, dilute, neutral KMnO4 / Ozonolysis) and that of Reagent2 is (reaction via acidic KMnO4 / reaction via cold, dilute, neutral KMnO4 / Ozonolysis).
- Give the reagents and conditions to provide the following transformations, usethe 1. 2. etc. notations as appropriateFill in the reagents a–c that are missing from the following scheme. Specify if any reaction conditions required in each step:An inexperienced graduate student treated dec-5-ene with borane in THF, placed the flask in a refrigerator, and left for aparty. When he returned from the party, he discovered that the refrigerator was broken and that it had gotten quite warminside. Although all the THF had evaporated from the flask, he treated the residue with basic hydrogen peroxide. Tohis surprise, he recovered a fair yield of decan-1-ol. Use a mechanism to show how this reaction might have occurred.(Hint: The addition of BH3 is reversible.)
- Give the structure of the product(s) for each step of the following reaction sequence.Give the major organic product(s) for each step of the following reaction2. In not more than three (3) sentences, explain why terminal alkynes are acidic.3. What impurities are removed when acetylene gas is made to pass through an acidifiedsolution of CuSO4?4. Explain the difference in the rate of free radical bromination reactions of toluene and cyclohexane.
- Which of the synthetic procedures shown in Image 31 would carry out the following transformation? A. b B. c C. a D. d1. Complete the following reaction schemes by adding the appropriate reactant(s), reagent(s), conditions, and/or major product(s). Make sure to indicate the proper regiochemistry and stereochemistry when necessary:A key step in the synthesis of naproxen, an NSAID more commonly known by its brand name, Aleve (Section 3.9), is a coupling reaction of 2-bromo-6-methoxynaphthalene to form 2-methoxy-6-vinylnaphthalene. Show three different coupling reactions, and the required reagents, that could be used tocarry out this step.

