Decide how the objects in the following table would be listed, if they were listed in order of increasing kinetic energy. That is, select "1" next to the obje the lowest kinetic energy, select "2" next to the object with the second lowest kinetic energy, and so on. object kinetic energy A 1300. kg car moving at 89. km/h. (choose one) A 1300. kg car moving at 58. km/h. (choose one) A 1300. kg car moving at 31. km/h. (choose one) A 1000. kg car moving at 31. km/h. (choose one)
Decide how the objects in the following table would be listed, if they were listed in order of increasing kinetic energy. That is, select "1" next to the obje the lowest kinetic energy, select "2" next to the object with the second lowest kinetic energy, and so on. object kinetic energy A 1300. kg car moving at 89. km/h. (choose one) A 1300. kg car moving at 58. km/h. (choose one) A 1300. kg car moving at 31. km/h. (choose one) A 1000. kg car moving at 31. km/h. (choose one)
Chemistry for Today: General, Organic, and Biochemistry
9th Edition
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Chapter8: Reaction Rates And Equilibrium
Section: Chapter Questions
Problem 8.82E: Which of the following is the best example of potential energy changing to kinetic energy? a.Pushing...
Related questions
Question
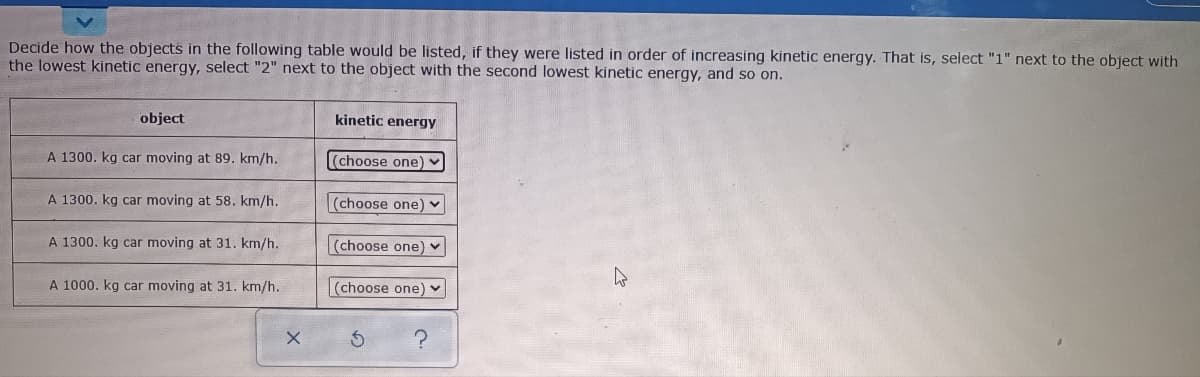
Transcribed Image Text:Decide how the objects in the following table would be listed, if they were listed in order of increasing kinetic energy. That is, select "1" next to the object with
the lowest kinetic energy, select "2" next to the object with the second lowest kinetic energy, and so on.
object
kinetic energy
A 1300. kg car moving at 89. km/h.
(choose one) -
A 1300. kg car moving at 58. km/h.
(choose one)
A 1300. kg car moving at 31. km/h.
(choose one) ♥
A 1000. kg car moving at 31. km/h.
(choose one)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning