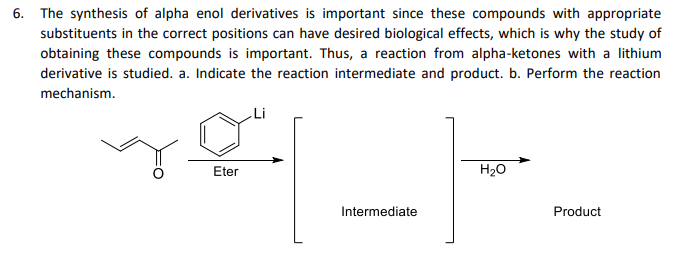
derivative is studied. a. Indicate the reaction intermediate and product. b. Perform the reaction mechanism. „Li Eter H20 Intermediate Product
Q: If K= 3.5 & K= 1.58 x 10 10, Suppose 100 ml of 0.010M aqueous amine is extracted with 200ml of benze...
A:
Q: When 0.25 g of the organic compound naphthalene are burned in a bomb calorimeter, the temperature of...
A: Given: Mass of naphthalene burned = 0.25 g. Initial temperature of calorimeter = 24.00 oC Final temp...
Q: A.) What volume of 8.4N Al(OH)3 will be used to prepare 350mL of 2.1M solution? B.) How many mL of w...
A: Given, Initial Concentration = 8.4 N Final volume, V2 = 350 ml Final Concentration, M2 = 2.1 M
Q: H3C CH3 H3C OCH3 CH3
A: This is a synthesis type transformation reaction , passing through several steps . Necessary reagen...
Q: n the calorimetry experiment was used to measure the heat of reaction for an endothermic process, wh...
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Write the letter of the pair of compounds that illustrates the given isomerism. CH, CH3 CH CH, он D ...
A: Here we have to find the pair compounds from the given compounds which are cis-trans geometrical iso...
Q: topic: matter do you think that each sample occupies space? write the reason(s) for each sample wa...
A: Yes, it does because whatever we see when you open ur eyes is matter and what you feel when you clo...
Q: The peroxydisulfate ion (S2O8-2) reacts with the iodide ion in aqueous solution via the reaction: S2...
A: (a) Answer -
Q: Food rots about 30 times more rapidly at 25 °C than when it is stored at 6 °C. Determine the overall...
A:
Q: when you mix 20ml sugar and 50ml water in a 250ml beaker, what is the volume of the sugar and water ...
A: Volume of mixture can be calculated from individuals volumes.
Q: 18The equivalence point is at the center of the steep part of the titration curve only if the reacti...
A: 4. The titration curve for a general acid-base titration can be represented as:
Q: Aluminum reacts with oxygen according to the equation: 4Al + 302 → 2A:IOs. How many moles of oxygen ...
A: Given reaction 4Al +3O2 -------> 2Al2O3 According to stochiometry of this reaction 4 moles of al...
Q: ular equations. (Use the lowest possible coefficients. Be sure to specify states such as (aq) or (s)...
A: As per our guidelines we can only solve first three sub-parts. Please resubmit the other questions a...
Q: Write the letter of the pair of compounds that illustrates the given isomerism. CH3 H,C. CHs CH3 H2N...
A: The E/Z isomerism is defined as the molecule which has substituent on the same side or opposite side...
Q: hink about sugar and water as make up of tiny particles. explain what happens when you mix it. use i...
A: Given : We have to explain what happens when sugar dissolves in water .
Q: What is the final temperature (in degrees Celsius) when 1940 J of energy are put into a 250 millilit...
A: We have to predict the final temperature.
Q: How many moles of solute particles are present in 1.43 mL of 0.447 M K2SO4? When you have your ans...
A: Number of solute particles can be calculated from the volume and concentration.
Q: f. 2,3-dimethyl-4-propyloctane g. 1,3,5-trimethylcyclohexane h. cis-1,2-dipropylcyclopentane i. 2-br...
A:
Q: Worksheet-IUPAC nomenclature Organic Nomenciature Practice Name Structural Formula butane
A: IUPAC naming of the given compounds. As per our guideline we have to answer first three questions ...
Q: v Saved Question 5 (Mandatory) (1 point) What is the Lewis diagram for oxygen? a) b): O : c) • 0 : d...
A: Lewis symbol is written as symbol of element and putting dots equal to number of valence electrons.
Q: Search the NIST library for the UV spectrum of biphenyl. What is the expected lambda (A) max?
A: Hydrocarbon is the term furnished to the molecule that has only carbon and hydrogen atoms as its con...
Q: suck water from a cup up to the 30ml of a syringe without needles. cover tightly the tip of the syri...
A: Given: Suck water from a cup up to the 30ml of a syringe without needles. cover tightly the tip of t...
Q: Lodu.edu/ultra/courses/_393452 1/cl/outline v Question Completion Status: A student wants to determi...
A: Ethyl acetate and water are colourless while copper (l) bromide is green in colour.
Q: Quiz: Name the ionic compound CI -1 K +1 (SO.)-2 Ca +2 Al+3 (NH,)+1
A:
Q: 4. The heat capacity of solid lead oxide is given by the equation: Cp(T) = 44.35 + 1.47 × 10-3 × T %...
A: This is can be calculated by Kirchhoff equation.
Q: If each dose of amoxicillin is 2.5ml ,how many doses of amoxicillin are contain in 50ml of amoxicill...
A:
Q: Prepare 250mL of a 0.75M solution of phosphoric acid from 75.0% phosphoric acid that is pure. The gr...
A: we are required to prepare 250 ml of 0.75 M of phosphoric acid from 75% phosphoric acid by using the...
Q: Using your experimental data for the lower molal potassium chloride solution, what is the k, of wate...
A: In a solution, certain physical properties of the solution have deviation from the physical properti...
Q: The element germanium is in Group 14, in Period 4. It is classed as a semimetal or metalloid, as is ...
A: A question based on Group 14 elements that is to be accomplished.
Q: explain what will happen to the liquid on a watch glass if it is transferred into a small container ...
A: Given : We have to tell what will happen if liquid kept on watch glass is placed in freezer for a fe...
Q: Write ionic equations and net ionic equations in the following reactions: Na2S(aq) + ZnCl2(aq) → ...
A: In an ionic equation, the chemical reaction is written as the ions present in the reactants and in t...
Q: Rank the compounds in each group according to their reactivity toward electrophilic substituti 1 = m...
A: Rate of Electrophilic substitution reaction increases if benzene ring is attached to the electron wi...
Q: 1. Calculate by Van der Waals' equation the pressure at which 2 moles of ammonia will occupy a volum...
A: Van der Waal's equation is, P = [RT/(Vm-b)] - [a / Vm2] Where, P = Pressure in atm R = Universal Gas...
Q: select the matching answers that apply to stereoisomers, constitutional isomers, and conformational ...
A: The compounds with the same molecular formula but the bonds connectivity is different, known as cons...
Q: topic: matter does each sample have a measurable mass? how do you measure the mass of each sample? ...
A:
Q: 0.2 molal aqueous solution of an electrolyte A,B is 90% ionised. The boiling point of the solution a...
A: Molality of solution = 0.2 molal Kb for water = 0.512KKg/mol
Q: when you pull the pluger of the syringe without needle, until it reaches 30mL, press your thumb on t...
A: Given: you pull the plunger of the syringe without a needle, until it reaches 30mL, press your thumb...
Q: Which of the following substances has the lowest absolute molar entropy at 25°C? Group of answer c...
A: The substances given are HCl(l), HCl (g), HCl (s), HBr (g) and HI (g).
Q: Methane and water react to form carbon monoxide and hydrogen, like this: CH,(2)+H,O(2) -→ CO(2)+3H,(...
A: The reaction shifts towards the side which has fewer moles or pressure.
Q: но H,0 он CH3 CBD
A: Alkene reacts with alcohol in presence of an acid catalyst by an electrophilic addition reaction mec...
Q: The proposed mechanism for a reaction is 1. A(g) + B(g) ⇄ X(g) Fast 2. X(g) + C(g) ⇄ Y(g) Fast 3....
A: We have to predict the rate law for the reaction..
Q: MİCRONS 5.5 2.5 100 26 27 28 2,9 3 3.5 4,5 11 12 13 90 70 50 40 30 20 1773 cm 10 3800 300 3400 3200 ...
A:
Q: This compound was synthesized from benzene by the scheme outlined. CH3 Br ČH3 1. CH3CI, AICI3 2. CH3...
A: The first step of this synthesis is called Friedel craft alkylation . It is an electrophilic aromati...
Q: 5. Particles of matter moved depending on the spaces between them. What word is used to describe the...
A: A matter is composed of atoms. These atoms will be arranged in a specific manner in different states...
Q: Calculate the pH of 1.0×104M solution of HNO3. (Round to whole number).
A: Given : We have to calculate the pH of solution.
Q: 3..Suggest how to make syndiotactic/isotactic polystyrene on the one hand and on the other hand how ...
A: Isotactic polymers have the side groups attached on one side of the backbone chain whereas the syndi...
Q: How many grams of KNO3 (101.11g/mol) are in 10.8 ml of a 1.25 M solution?
A:
Q: Match the reservoir with the correct turnover rate of carbon: + land biosphere A. centuries + ocean ...
A: Turnover rate: fraction of material that leaves a reservoir in a specifed time interval. Mathematic...
Q: one lone electron
A:
Q: entropy
A:
How can I do this reaction?
Step by step
Solved in 2 steps with 1 images

- a,b-Unsaturated carbonyl compounds are susceptible to either 1,2 addition or 1,4 (Michael)addition. Give examples of nucleophiles that would favor each case and explain yourreasoning8.29 Give the mechanistic symbols ( Sn1, Sn2, E1, E2) that are most consistent with each of the following statements: (d) The substitution product obtained by sovolysis of tert-butyl bromide in ethanol arises by this mechanism (e) In ethanol that contains sodium ethoxide, tert-butyl bromide reacts mainly by this mechanism (f) These reaction mechanisms represent concerted processesPlease give the appropriate reagents FOR 3 and 4 to complete the following synthesis. ******** Keep mind, H2/PD does NOT go into 3 and KMNO4 does NOT
- Which of the following statements about terminal alkynes is FALSE?I I. A geminal dihalide is produced by the hydrohalogenation reaction.II. The proton in the terminal carbon is acidic but just slightly.III.They create an aldehyde when they react with H2O, H2SO4, and HgSO4.IV. A silver acetylide is formed after treatment with alcoholic AgNO3.Part 2 - Syntheses (64 points in total) Propose a plausible synthesis for each of the following transformations. Write all steps with required reagents and intermediates. As a product, one of the enantiomers is shown only.The enolate derived from diethyl malonate reacts with a variety of electrophiles (not just alkyl halides) to form new carbon–carbon bonds. With this in mind, draw the products formed when Na+ −CH(CO2Et)2 reacts with each electrophile, followed by treatment with H2O.
- Fill in the missing reagents below.Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpRank the following in increasing order of reactivity towards nucleophilic acyl substitutiona. CH3COCl, CH3COOCH3, CH3CONH2b. CH3COOCH3, CH3COOCH2CCl3, CH3COOCH(CF)3
- The enolate derived from diethyl malonate reacts with a variety ofelectrophiles (not just alkyl halides) to form new carbon–carbon bonds.With this in mind, draw the products formed when Na+ −CH(CO2Et)2reacts with each electrophile, followed by treatment with H2O.Give the major organic product of each reaction of γ‑valerolactone with each of the given six reagents under the conditions indicated. Do not draw any byproducts formed. Reagent 1. NaOH, H2O, heat, then H+, H2O 2. (CH3)2CHCH2CH2OH (excess), H+ 3. (CH3CH2)2NH and heat 4. CH3MgI (excess), ether, then H+/H2O 5. LiAlH4, ether, then H+/H2O 6. DIBAL, toluene, low temperature, then H+/H2OChemistry Find the most efficient synthetic route for the followinf transformations.

