Determine the composition of each phase at 1300C, 1250C, and 1200C temperatures. Indicate wt%Ni of the first solid to form and the last liquid to solidify. Show it on phase diagram below.
Determine the composition of each phase at 1300C, 1250C, and 1200C temperatures. Indicate wt%Ni of the first solid to form and the last liquid to solidify. Show it on phase diagram below.
Welding: Principles and Applications (MindTap Course List)
8th Edition
ISBN:9781305494695
Author:Larry Jeffus
Publisher:Larry Jeffus
Chapter26: Welding Metallurgy
Section: Chapter Questions
Problem 12R: What is a eutectic composition?
Related questions
Question
(a) Determine the composition of each phase at 1300C, 1250C, and 1200C temperatures. Indicate wt%Ni of the first solid to form and the last liquid to solidify. Show it on phase diagram below.
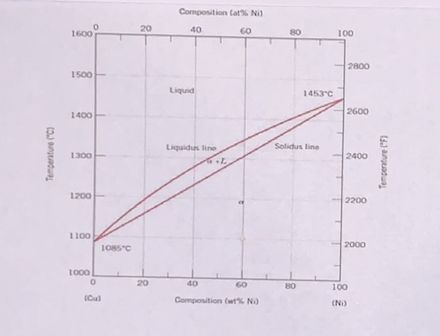
Transcribed Image Text:Composition Cat% Ni
20
40
60
80
1600
100
2800
1500
Liquid
1453 C
2600
1400
Liquidus line
Solidus line
1300
2400
1200
2200
1100
2000
1085"C
1000
20
40
60
80
100
(Cul
Composition (wt% NO
(Ni)
Temperature ("F)

Transcribed Image Text:(b) Draw the changes in microstructure of Cu-40%Ni alloy during equilibrium solidification at 1300°C,
1250°C and 1200°C . See diagram below.
(c) Calculate the amount of each phase at those temperatures.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Welding: Principles and Applications (MindTap Cou…
Mechanical Engineering
ISBN:
9781305494695
Author:
Larry Jeffus
Publisher:
Cengage Learning

Welding: Principles and Applications (MindTap Cou…
Mechanical Engineering
ISBN:
9781305494695
Author:
Larry Jeffus
Publisher:
Cengage Learning