Phase Diagrams: You are given the lead tin (Pb-Sn) phase diagram. Below the eutectic isotherm is identified as the two phase region a + B. a) At 30wt % Sn, in the circles below draw the microscopic observations evolving at 3000C, 2250C, and 1000C for this hypoeutectic alloy. b) At a temperature just above the isotherm at 1830C, for the 30 wt% Sn Alloy, calculate the fractions of each phase present at equilibrium and the corresponding composition of each phase.
Phase Diagrams: You are given the lead tin (Pb-Sn) phase diagram. Below the eutectic isotherm is identified as the two phase region a + B. a) At 30wt % Sn, in the circles below draw the microscopic observations evolving at 3000C, 2250C, and 1000C for this hypoeutectic alloy. b) At a temperature just above the isotherm at 1830C, for the 30 wt% Sn Alloy, calculate the fractions of each phase present at equilibrium and the corresponding composition of each phase.
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Kreith, Frank; Manglik, Raj M.
Chapter5: Analysis Of Convection Heat Transfer
Section: Chapter Questions
Problem 5.2P: 5.2 Evaluate the Prandtl number from the following data: , .
Related questions
Question
100%
Phase Diagrams: You are given the lead tin (Pb-Sn) phase diagram. Below the eutectic isotherm is identified as the two phase region a + B.
a) At 30wt % Sn, in the circles below draw the microscopic observations evolving at 3000C, 2250C, and 1000C for this hypoeutectic alloy. b) At a temperature just above the isotherm at 1830C, for the 30 wt% Sn Alloy, calculate the fractions of each phase present at equilibrium and the corresponding composition of each phase.
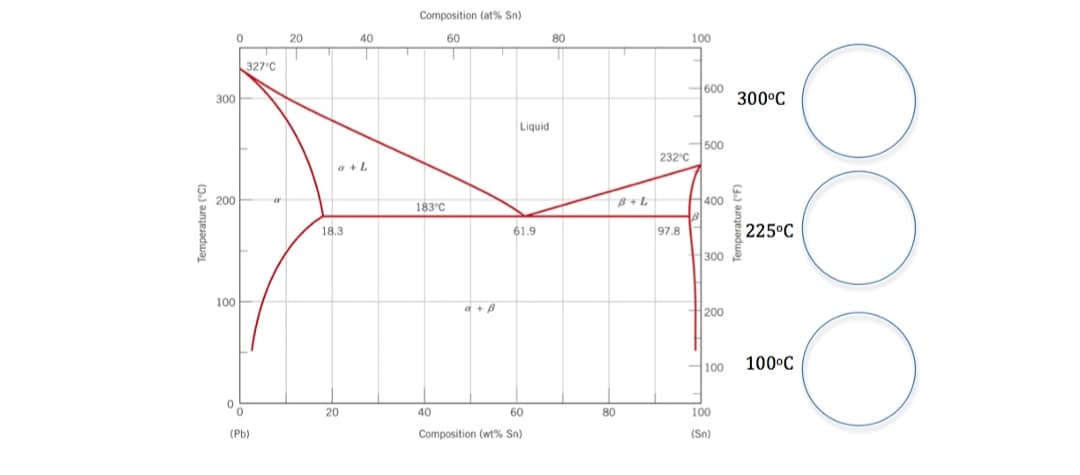
Transcribed Image Text:Composition (at% Sn)
20
40
60
80
100
327 C
600
300°C
300
Liquid
500
232 C
a +L
200
B+L
400
183°C
18.3
61.9
97.8
225°C
300
100
a +B
H200
H100
100°C
20
40
60
80
100
(Pb)
Composition (wt% Sn)
(Sn)
Temperature ("C)
Temperature ("F)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning