Q: Fifty grams of Xenon (AW = 131.293 g/mol) undergoes a reversible isothermal expansion from 1.00 atm,…
A:
Q: 1. Describe Joule's experiment and the conclusion he came up with as a result.
A: Joule's experiment is change in internal energy when a gas expand isothermally (gas expand into…
Q: What would be the best sequence of reactions/reagents to use for this transofmration (the one that…
A: Elimination reaction of alkyl halide: Alkyl halide gives an elimination reaction by reacting with a…
Q: Question 5 of 19 Maemillan Learni What is the concentration of a 52.45 mL solution of HBr that is…
A:
Q: Consider the reaction given bellow Al(OH), (s) Al³+ (aq) + 3OH- (aq) ⇒ 3 Adding acid to this mixture…
A:
Q: Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are…
A: We will first concentrate on the reaction, what product it may result. And then we can predict…
Q: 1. Initial temperature of solid specimen = 98 Initial temperature of vessel, stirrer and water = 29…
A: Given, Initial temperature of solid specimen , Ts=98 Initial temperature of vessel , stirrer and…
Q: The heat of combustion of liquid benzaldehyde (C6H5CHO) is -3393 kJ/mole, and that of liquid…
A: (a) The heat of combustion of liquid benzaldehyde is given to be -3393 kJ/mol while that of liquid…
Q: b) Write the products of your compound and the following reagents. If the reaction would not work…
A: R/S nomenclature done considering the CIP rule. Reactions are involves nucleophilic attack, imine…
Q: A 1.50 mol sample of He occupies a volume of 2.50 L at a pressure of 14.7 atm. What will be the…
A:
Q: OCHEMICAL REACTIONS Dilution A chemist must dilute 43.3 mL of 205. uM aqueous silver(II) oxide (AgO)…
A: We have to calculate the final volume of solution
Q: Identify the relationship between the following compounds. TH H Br O constitutional isomers O…
A: We have to determine the relationship between the two given compounds
Q: Macmillan Learning Write the balanced equation for the combustion of 1-naphthol, C,H,OH, to give CO₂…
A: Combustion of the any substance means burning in presence of air or more precisely in presence of…
Q: Hydrogen sulfide decomposes according to the following reaction: 2H2S(g) → 2H2(g) + S2(g) - For this…
A:
Q: Name the following compound MnSO3
A: Name of the compound is- Manganese(II) sulfite (or, manganese sulfite)
Q: b) Write the products of your compound and the following reagents. If the reaction would not work…
A:
Q: In the following net ionic equation, identify each reactant as either a Bronsted-Lowry acid or a…
A:
Q: Use the following data to determine the average of %K3[Al(C₂O4)3] in samples prepared by students.…
A: According to the question, we can write the balanced chemical equation between KMnO4 and…
Q: 1. Calculate q, w, AU, AH for each reversible process below. P O M N LM: reversible, isothermal…
A: For reversible, isothermal process (LM) Isothermal process: Temperature remains constant ∆U = ∆H=0…
Q: OO HUAWEI P30 LEICA TRIPLE CAMERA a N M
A: Titration: Titration is a quantitative technique usually performed to determine the unknown…
Q: which of the following compounds the central atom uses sp3 hybridized orbitals to form bonds? Group…
A: Steric number = number of loan pair + number of bond pair Steric number = 2 -> sp Steric…
Q: The name of KBrO4 is a) Potassium Bromide b) Potassium Bromite c) Potassium Bromate d) Potassium…
A:
Q: Macmillan Learning How many grams of Br are in 395 g CaBr₂? mass:
A:
Q: A voltaic cell is constructed from a standard Al³+|Al half cell (Eº, red standard Sn2+ | Sn half…
A:
Q: Exercise The volume of 1 mole of Helium (He) gas at different temperature values given in the…
A: On plotting graph of Volume of He vs Temperature :
Q: 3 Assign the alkene stereochemistry as E or Z
A: Steps to find the E / Z configuration in a compound : Assign priority to each substituent around…
Q: 9. Sulfuryl dichloride is formed when sulfur dioxide reacts with chlorine. The data refer to 298K.…
A: Rxn : SO2(g) + Cl2(g) → SO₂Cl2(g) ∆H°Rxn = { ∆H°f(Products)} -{ ∆H°f (Reactants)} =…
Q: What is the ionic formula for the following ? zirconium thiosulfite aluminum borate
A: In the ionic formula, different ions like cations and anions are mentioned along with their formal…
Q: Radium is a radioactive element whose nucleus emits an alpha particle (a helium nucleus) that has a…
A: Kinetic energy of alpha paricle = 7.6×1013 J
Q: Which of the following corresponds to the resonance structure that can be obtained from the given…
A: The resonating structures of the given compounds are shown below:-
Q: Starting from the following alkene, which products will be obtained upon the action by the following…
A: Answer:- This is answered by using the simple concept of chemical reactions of organic compounds…
Q: Explain why a crystal made of atoms with a weakly bound outer electron is a conductor. Illustrate…
A: Conductor - it is a object or meterial that allow to flow electricity in it.
Q: A 56.0 g sample of brass is put into a calorimeter (see sketch at right) that contains 100.0 g of…
A:
Q: Arrange the following phenols in terms of increasing reactivity with NaOH. Drag the correct boxes to…
A: Given 2-iodophenol 3-methoxyphenol 4-cyanophenol
Q: Explain the different steps of making wine and what are the chemical reactions involved in wine.…
A: Wine is made from grapes.
Q: emonstrated in General Chemistry Laboratory (I), Chem. 105. The following table ontains the results…
A: Given is the data of the experiment performed by various groups and we have to find the correct…
Q: Consider the equilibrium reaction: CaCO3(s) + H₂O(g) Ca(OH)2(s) + CO2(g) Which of the following…
A: In this reaction solid CaCO3 react with gaseous water to form solid Ca(OH)2 and carbon dioxide gas.…
Q: <Homework 9C-Attempt 1 Exercise 8.101 Part A Explain why atomic radius decreases as you move to the…
A: Atomic radius is the distance from centre of the nucleus to the outermost shell or valence…
Q: Classify each of the molecules in the table.
A: we have to classify the given molecules
Q: What is AG rxn for the following reaction? 3NO2(g) + H2O(1) 2HNO3(0) + NO(g) Substance AG°f (kJ/mol)…
A:
Q: In a gas mixture, the partial pressures are argon 440 mmHg, neon 70 mmHg, and nitrogen 130 mmHg.…
A:
Q: Problem 19.91 Liquid A standard air conditioner involves a refrigerant that is typically now a…
A: Since you asked multiple images questions so as per Q&A guidelines of portal I solved first…
Q: 21. At STP, how many moles are in these volumes of gases? a. 67.2 L SO₂ b. 0.880 L He c. 1.00 × 10³…
A: We have to calculate the number of moles of gas at STP
Q: Refer to the structures shown below. Which of the following is/are true? (Marks will be deducted for…
A: -> In hexoses there is 6 carbon chain . ->Enantiomers are compounds in which each chiral…
Q: ASSUMING EACH OF THE FF. PAIRS OF MOLECULES HAS THE SAME CARBON CHAIN LENGTH, WHICH MEMBER OF EACH…
A: Boiling Point of compound depends upon the strength of intermolecular forces of attraction. The…
Q: II: Assign common name to each of the following amines: 1. CH3- CH2 – NH - CH2 – CH3 2. CH3-CH2-CH2…
A: For the common nomenclature of amines the carbon chain is treated as alkyl group bonded to N atom.…
Q: Two moles of an ideal gas at 30°C and 1.00 MPa pressure is expanded adiabatically to a final…
A:
Q: What is the the root-mean-square speed of H₂ molecules at 31.3 °C? i eTextbook and Media m/s
A:
Q: Write a net ionic equation to show why the solubility of Pb(OH)₂(s) increases in the presence of a…
A: In aqueous solution PbOH2 s ionizes in the following way - PbOH2 s ↔…
Q: In the following net ionic equation, identify each reactant as either a Bronsted-Lowry acid or a…
A:
Step by step
Solved in 2 steps with 1 images

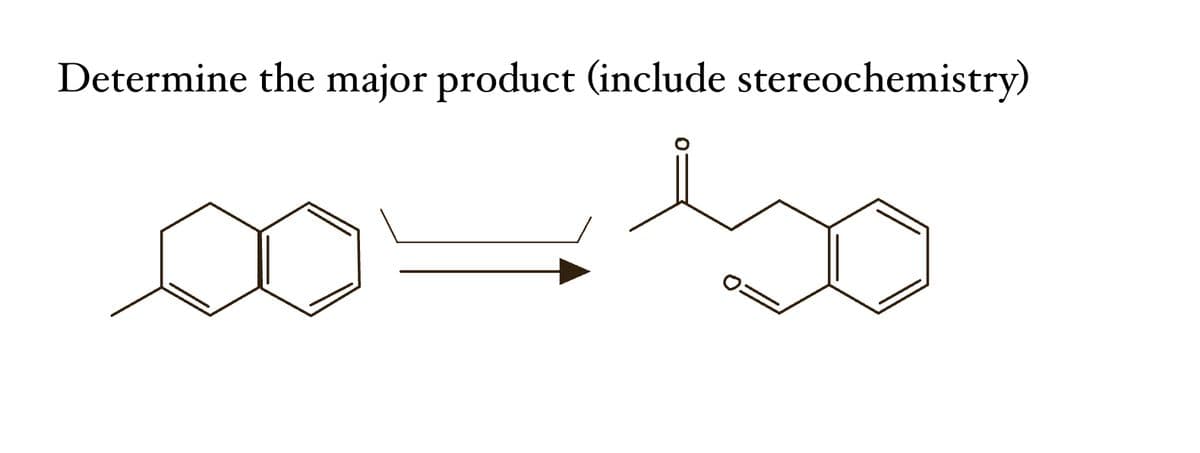
- Draw the product of the attached reaction, including stereochemistry.Determine the major organic products for the reactions in addition to whether they are racemic or not?Draw the structure of the product, substrate or condition in the following reactions (should clearly indicate the stereochemistry).
- Predict the major products of both organic reactions. Be sure to use wedge and dash bonds to show the stereochemistry of the products when it's important, for example to distinguish between two different major products.Draw the structure of product, substrate or condition in the followingreactions (should clearly show the stereochemistry).In an early transition state the TS structure resembles?

