Determine the molar concentration of a Ca(OH)2 solution whose pOH is 3.47 at 25.0 °C. (the answer should be entered with 3 significant figures; do not enter units; give answer in scientific notation--valid notation examples include 1.23e-8 and 1.23e8 and -1.23e-4 and 1.23e0)
Determine the molar concentration of a Ca(OH)2 solution whose pOH is 3.47 at 25.0 °C. (the answer should be entered with 3 significant figures; do not enter units; give answer in scientific notation--valid notation examples include 1.23e-8 and 1.23e8 and -1.23e-4 and 1.23e0)
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter16: Acid-base Equilibria
Section: Chapter Questions
Problem 16.126QP: A solution is prepared from 0.150 mol of formic acid and enough water to make 0.425 L of solution. a...
Related questions
Question
15
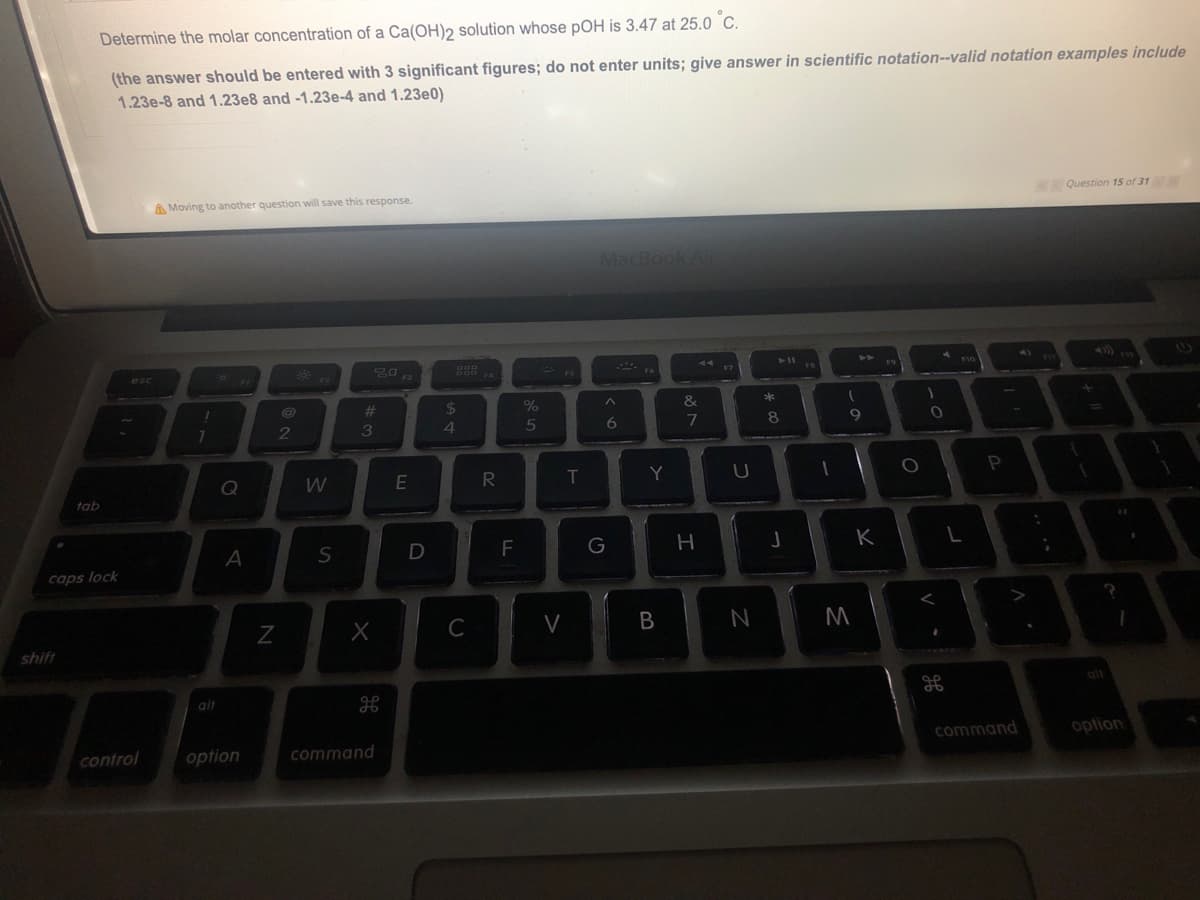
Transcribed Image Text:Determine the molar concentration of a Ca(OH)2 solution whose pOH is 3.47 at 25.0 C.
(the answer should be entered with 3 significant figures; do not enter units; give answer in scientific notation--valid notation examples include
1.23e-8 and 1.23e8 and -1.23e-4 and 1.23e0)
A Moving to another question will save this response.
Question 15 of 31
MacBook Air
20
44
esc
%23
24
&
4.
5
6
7
Q
E
T
Y
tab
D
F
G
K
caps lock
C
V
M
shift
alt
command
option
control
option
command
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co