Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.9QAP
Related questions
Question
Determine the structure for the molecule that most likely produced these spectra.
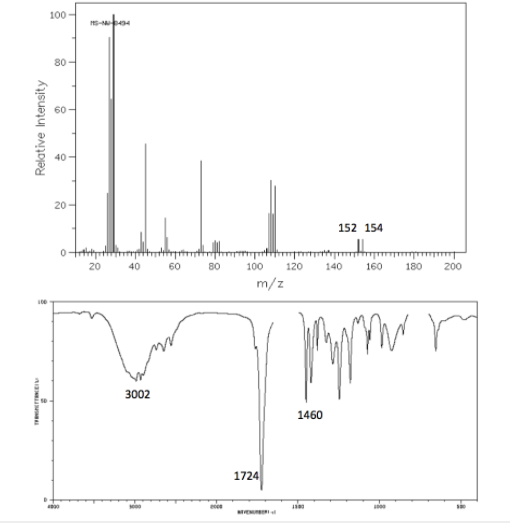
Transcribed Image Text:100 -
80
60
40-
20-
152 154
20
40
60
80
100
120
140
160
180
200
m/z
3002
1460
1724
ce
Relative Intensity
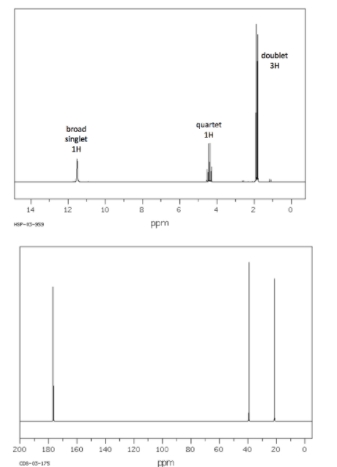
Transcribed Image Text:doublet
3H
quartet
broad
1H
singlet
1H
14
12
10
H
ppm
200
L80
160
140
120
100
60
40
20
C--175
pm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you


Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning