Q: [Review Topics] [References] Use the References to access important values if needed for this…
A: The relation between molality and mole is- Molality= mole / kg of solvent
Q: 11) Calculate the volume, in liters, occupied by each of the following: a. 2.00 moles of H₂ at 300.…
A:
Q: Octanitrocubane is a high energy explosive material composed of the elements C, N, and O. Combustion…
A:
Q: .2500 moles of dissolved silver nitrate is mixed with .2500 moles of dissolved sodium carbonate to…
A:
Q: CI LOCH3 'Br AICI 3 H₂SO4 hint: perform a single EAS reaction
A: Both the reactions are examples of Aromatic Electrophilic Substitutions. In aromatic electrophilic…
Q: 1. How many grams of NH4OH must be dissolved in enough water to make 350 mL solution such that the…
A: 1. A certain amount of NH4OH is dissolved in enough water to make 350 mL of solution in which…
Q: An unknown compound contains only carbon, hydrogen, and oxygen. Combustion of a 16.45 mg sample…
A: mass of sample = 16.45 mg = 0.01645 g mass of CO2 = 38.93 mg = 0.03893 g mass of H2O= 15.93 mg =…
Q: Give good solution ASAP a gas mixture contains 58.6% nitrogen and 41.4% krypton by mass.…
A:
Q: An atom of chlorine (CI) has a mass number of 37. Given only this information, which of the…
A: Given that atomic mass of chlorine = 37 Atomic number of chlorine = 17 Atomic number of neutral…
Q: bollit so of boon lost 2505 6918 Hydrogen peroxide (H₂O2 34.02 g/mol) is sold as an aqueous solution…
A:
Q: gas. How many moles of gas should be removed to be decreased the volume of container to 40L?-
A: Given intial volume (V1 ) = 85 L Number of moles (n1) = 3.4 mole Final volume (V2) = 40L…
Q: Calculate the number of O atoms in 0.0136 g of CaSO4 2H₂O. x 10 O atoms (Enter your answer in…
A: Answer: Given data: Mass of CaSO4.2H2O m=0.0136g
Q: Write a balanced equation for the following: 1. Mg | Mg2+ || Set | Sc 2. Sn Sn²+ || Pb²+ | Pb
A:
Q: KOH(s) + CO₂(g) → K₂CO₃(s) + H₂O(l) Calculate the mass in grams of KOH that will be required to…
A:
Q: 2Cu+ (aq) = Cu²+ (aq) + Cu (s) Calculate the standard free enthalpy.
A:
Q: H
A:
Q: What is the pH of 0.01 mol.L-¹ KOH solution ? Select one: a. 0 b. 2 c. 7 d. 12 e. 14
A: Given,0.01 mol.L-1 KOH solution .Required, The pH of the above solution .
Q: [References] A solution was made by mixing glucose (C6H12O6) and water (H₂O). Given that the mole…
A:
Q: X STS Al (Hg), Meot desulfonation
A:
Q: 8. 12 P OPER NEN 20 icomms svig of GOD negen 57-71 noggen 89-10 ZORDRIN t) Assume Dr. Bork gives you…
A: Given in following question in question 8 molecule of sucrose calculate how many grams of sucrose.…
Q: Sodium hydrogen carbonate (NaHCO3), also known as sodium bicarbonate or "baking soda", can be used…
A:
Q: The volume of the gas @ 17.5 psi decreases from 1.8L to 750 mL. What is the new pressure of the gas…
A: According to the Boyle's law, we know that pressure of a gas is inversely proportional to its…
Q: Give the proper IUPAC name for the molecule shown as a Fischer projection. Make sure to include the…
A:
Q: Calculate the solubility of Nickel Hydroxide, Ni(OH), in g/L 25C. Assume a large excess of the…
A: The solubility expression for Ni(OH)2 is Ni(OH)2 (s) <-------> Ni+2 (aq) + 2 OH– (aq) At…
Q: Pheromones are chemical substances secreted by members of a species to stimulate a response in other…
A:
Q: A 50.0 mL solution of Ca(OH)₂ with an unknown concentration was titrated with 0.340 M HNO₃. To reach…
A: Calcium hydroxide reacts with nitric acid to form calcium nitrate. This reaction is an example of a…
Q: Describe the difference in the reactivity tests when the iron complex is treated with SCN by itself…
A: When Fe3+ ion react with SCN- in presence of acid there is formation of blood red colored complex.…
Q: ΔS° for the reaction shown is equal to -610.5 (J/mol-K). Use the information given in the table to…
A: Given, 3 A(s) + 2 B(g) → 1 C(s) + 2 D(g) ∆S°rxn = -610.5 J/mol.K Standard entropy, S° for he…
Q: What is the specific rotation of D after the dilution? specific rotation: What is the specific…
A: To find out the specific rotation of D after the dilution. The specific rotation of an optical…
Q: What is the correct orbital diagram sequence of 3d6? Up, Up, Up, Up, Up, Down Down, Down, Up, Up,…
A:
Q: What is the charge on the R-group of a glutamic acid molecule at a pH of 8.5? Please use the…
A: Henderson-Hasselbach equation: pH = pKa + log [salt][acid] protonated form is acid and deprotonated…
Q: 1. Given the following solvents and their dielectric constants: Hexane Benzene Ethanol Water b.…
A: The given solvents and their dielectric constants are Hexane - 1.88 Benzene - 4.6 Ethanol - 25 Water…
Q: Asses.1: The initial-rate data in the table below were obtained for the represented reaction. A+B 2C…
A:
Q: The rate constant for the reaction of persulfate - iodide reaction is 12.5 *102 M¹s¹. At certain…
A:
Q: The 1H NMR spectrum of 1-bromo-2-methylpropane would be expected to have a.) singlet (9H) , doublet…
A:
Q: g) 2U is produced by B-emission h) 56Mn is produced by a-emission i) B forms by positron emission j)…
A: Nuclear reaction: The given reaction is a nuclear reaction. In the nuclear reaction, one nuclide is…
Q: Calculate the surface area of charcool in m²/gram charcool covered by acetic acid when 100.0 ml of…
A:
Q: Among the following H atom transitions, which would emit a photon of light with the longest…
A: We know, the electron will move from a lower stationary energy state to a higher stationary energy…
Q: Consider the below oxidation rection: C12H22011 0₂ C0₂2 + H₂O a) Calculate the grams of oxygen…
A: The explanation is given below-
Q: 11.) You may look up R, Rydberg's equation, Planks constant, Planks equation, and third to the first…
A:
Q: Given that there 199967.8036 ton of CO2 can fit in a (hypothetical) mine, how would this value…
A: Given the total CO2 produced by the hypothetical mine = 199967.8036 ton
Q: Calculate the number of H ions in 7.76 g of BeH₂. x 10 Hions Enter your answer in scientific…
A: Here we have to determine the number of H1- ions present in 7.76 g of BeH2 .
Q: Calculate the pH of the following solutions: 0.1 M HCI 0.1 M NaOH 1 nM HCI 0.1 M Acetic acid, Ka=…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: d. Circle the functional groups listed below that are in PF-00835231. Carboxylic Acid Aldehyde Ester…
A: Functional group is atom or group of atom when attached with ring change its chemical and physical…
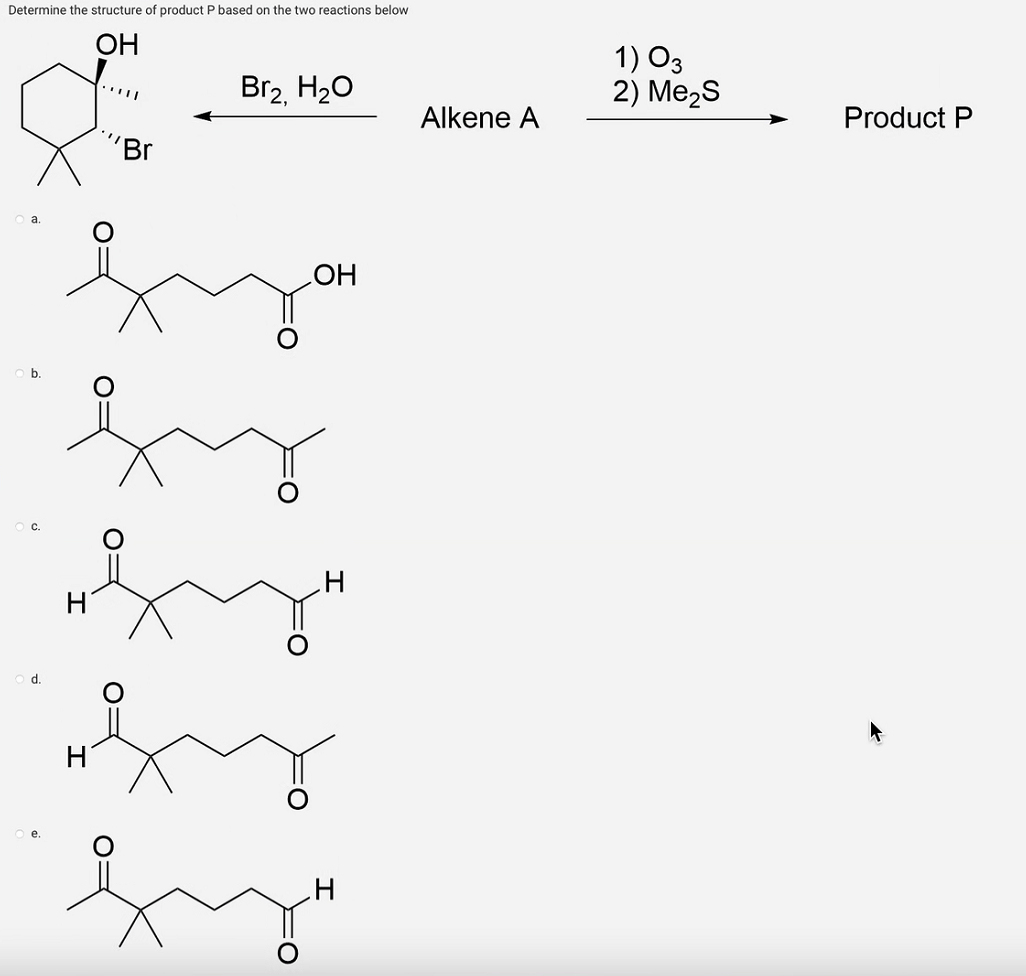
Q: Draw the structure of an alkane or cycloalkane that has more than three but fewer than ten carbon…
A: Organic compounds are made up of carbon and hydrogen. These organic compounds are called…
Q: "Reduction of Cr(VI) to Cr(III) is environmentally favorable as the latter species is not toxic to…
A: We will find order one by one and then rate constant
Q: Which of the following terms or phrases describe the CATHODE RAY TUBE experiment? discovered the…
A: Cathode ray tube is a vacuum tube which contains electron guns which ejected electrons in the tube .
Q: The initial conductivity of 0.1 M ethylacetate and 0.1M NaOH is 5.0*10-3 ohm 1, at the end of…
A: Saponification of ethylacetate proceeds by second order reaction.CH3COOC2H5 + OH- →CH3COO- + C2H5OH
Q: Macmillan Learning O A solution containing a mixture of metal cations was treated as outlined. 1.…
A: This is part of qualitative inorganic analysis, in which we followed the procedure as follows; 1.…
Q: Consider the mass of Sulfur and Fluorine in compound A (16.6 g S, 9.80 g F) and compound B (23.2 g…
A: According to atomic theory the atoms do no react in arbitrary numbers but in multiples of fixed…
47
Step by step
Solved in 3 steps with 3 images

- What is the organic chemistry mechanisms to get to this final product? one reagant is NaOCH2CH32. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 31. What type of reaction is occuring in step 3? (halogenation, hydrohalogenation, reduction, keto–enol tautomerism, dehydrohalogenation, acid-catalyzed hydration, base-catalyzed hydration) 2. Which reagent is necessary for step 3? (Br2, HBr, H2/Pt, NaNH2, H20/H2SO4/HgSO4)
- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Arrange the alkyl halides in order of increasing reactivity in an SN2 reaction with KI in acetone (least first). I, IV, III, II II, III, I, IV IV, I, III, II III, II, IV, IWhat is a possible formula for a carbocation with m/z=97
- M 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :1.Label kinetic product 2.Label the thermodynamic product 3.Predict the four products from reaction of alkene with one equivalent HBr1) What is the major product generally formed when a tertiary alkyl bromide (R3Br) reacts with sodium methoxide in methanol? 2) How is this major product formed?
- 1. What is reaction 1? a Halogenation via SR b Halogenation via SE c Halogenation via AE d Hydrohalogenation via AE 2. Which pair of reagent/catalyst is needed for reaction 1? a HBr / DCM b Br2 / CCl4 c NBS / H2O2 d Br2 / uv 3. What is reaction 2? a FC Acylation b FC Alkylation c Halogenation via SE d Bromination2) What is the main product for each of the following reactions? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (h) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ?Rank the species below in order of increasing nucleophilicity in protic solvent. I. H2O II. CH3S— III. CH3COO— IV. t-BuO— I, II, IV, III I, III, II, IV I, III, IV, II I, II, III, IV

