Dinitrogen trioxide, N2O3, is largely dissociated into NO and NO2 in the gas phase where there exists the equilibrium, N2O3 NO + NO2 In an effort to determine the structure of N2O3, a mixture of NO and "NO2 was prepared containing isotopically labeled N in the NO2. After a period of time the mixture was analyzed and found to contain substantial amounts of both *NO and *NO2. Explain how this is consistent with the structure of N2O3 being ONONO. Because this is an equilibrium process, there is a change that either the forward or the reverse reactions will occur. As NO reacts with NO2 we form This compound can decompose and give either NO2 and and "NO2. or After sufficient time, v will be present in the mixture. both "NO and NO Attempts: 0 of 3 used Submit Answer Save for Later only "NO only NO
Dinitrogen trioxide, N2O3, is largely dissociated into NO and NO2 in the gas phase where there exists the equilibrium, N2O3 NO + NO2 In an effort to determine the structure of N2O3, a mixture of NO and "NO2 was prepared containing isotopically labeled N in the NO2. After a period of time the mixture was analyzed and found to contain substantial amounts of both *NO and *NO2. Explain how this is consistent with the structure of N2O3 being ONONO. Because this is an equilibrium process, there is a change that either the forward or the reverse reactions will occur. As NO reacts with NO2 we form This compound can decompose and give either NO2 and and "NO2. or After sufficient time, v will be present in the mixture. both "NO and NO Attempts: 0 of 3 used Submit Answer Save for Later only "NO only NO
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter9: Nuclear Chemistry
Section: Chapter Questions
Problem 9.88P
Related questions
Question
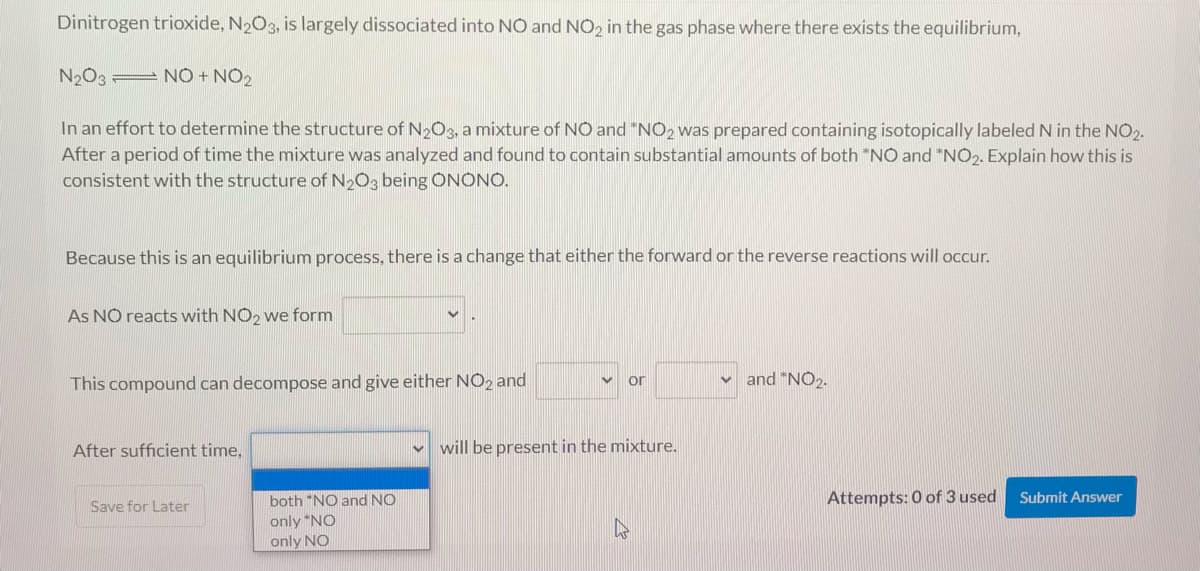
Transcribed Image Text:Dinitrogen trioxide, N2O3, is largely dissociated into NO and NO2 in the gas phase where there exists the equilibrium,
N2O3 = NO + NO2
In an effort to determine the structure of N2O3, a mixture of NO and "NO2 was prepared containing isotopically labeled N in the NO2.
After a period of time the mixture was analyzed and found to contain substantial amounts of both *NO and *NO2. Explain how this is
consistent with the structure of N2O3 being ÔNONO.
Because this is an equilibrium process, there is a change that either the forward or the reverse reactions will occur.
As NO reacts with NO2 we form
This compound can decompose and give either NO2 and
and "NO2.
or
After sufficient time,
v will be present in the mixture.
both "NO and NO
Attempts: 0 of 3 used
Submit Answer
Save for Later
only *NO
only NO
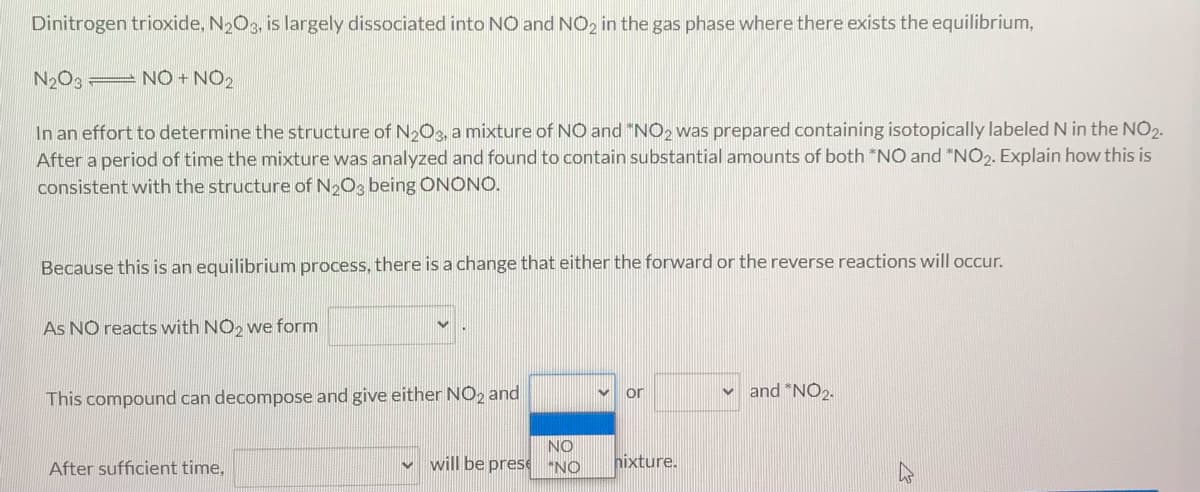
Transcribed Image Text:Dinitrogen trioxide, N2O3, is largely dissociated into NO and NO2 in the gas phase where there exists the equilibrium,
N2O3 NO + NO2
In an effort to determine the structure of N2O3, a mixture of NO and "NO2 was prepared containing isotopically labeled N in the NO2.
After a period of time the mixture was analyzed and found to contain substantial amounts of both *NO and "NO2. Explain how this is
consistent with the structure of N2O3 being ONONO.
Because this is an equilibrium process, there is a change that either the forward or the reverse reactions will occur.
As NO reacts with NO2 we form
or
and "NO2.
This compound can decompose and give either NO2 and
NO
will be prese "NO
hixture.
After sufficient time,
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning