Direct Cross-Dehydrogenative-Coupling (CDC) between Benzyl Ethers and Simple Ketones: DDQ O-RI 100°C DDQ(0.25mmol) 100°C, N2. 2.5h 1a 13b 0.2mmol 0.6mmol isolated yield 50% 16c 16d
Direct Cross-Dehydrogenative-Coupling (CDC) between Benzyl Ethers and Simple Ketones: DDQ O-RI 100°C DDQ(0.25mmol) 100°C, N2. 2.5h 1a 13b 0.2mmol 0.6mmol isolated yield 50% 16c 16d
Chapter27: Biomolecules: Lipids
Section27.SE: Something Extra
Problem 47AP: Cembrene, C20H32, is a diterpenoid hydrocarbon isolated from pine resin. Cembrene has a UV...
Related questions
Question
( plz give mechanism of all i will upvote )
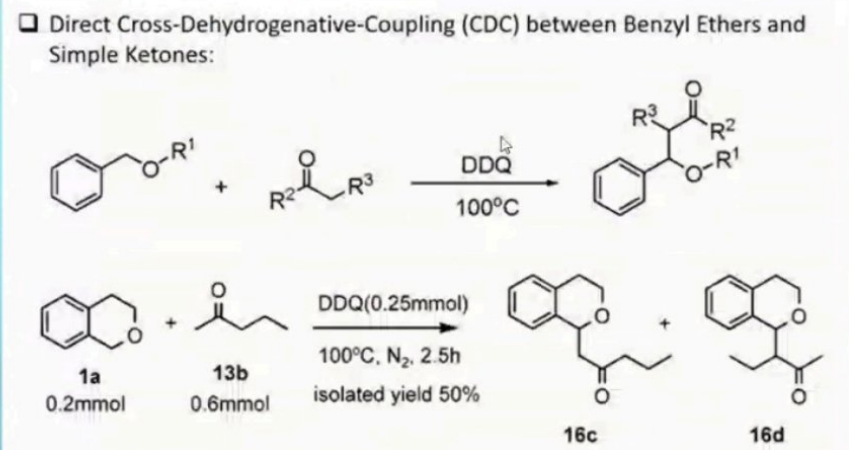
Transcribed Image Text:Direct Cross-Dehydrogenative-Coupling (CDC) between Benzyl Ethers and
Simple Ketones:
DDQ
O-RI
100°C
DDQ(0.25mmol)
100°C, N2. 2.5h
1a
13b
0.2mmol
0.6mmol
isolated yield 50%
16c
16d
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

