Q: Question: Calculate the molar solubility of magnesium hydroxide (Mg(OH)₂) in a solution that is…
A:
Q: [Keview Topics] [References] Draw a structural formula for the hemiketal formed when one molecule of…
A: In the formation of hemiketal, one molecule of alcohol reacts with ketone in the presence of acid.…
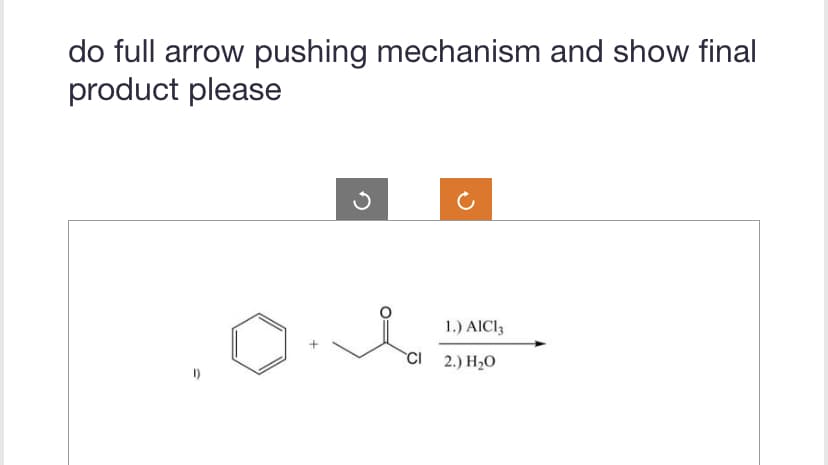
Q: show all reactants and intermediates to obtain final product
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: Fill in the potential double replacement products for each reaction shown below. Then for any that…
A: A replacement reaction is the reaction where one group or functional group is replaced by the other…
Q: Use the two-point version of the Clausius-Clapeyron equation. The vapor pressure of liquid iron is…
A:
Q: The accumulation of ketone bodies in the blood results in which of the following? A. a decrease in…
A:
Q: Use the table below to answer the question that follows. [H+] (mol/L) 1x 100 1x 10/1 1x 10/² 1x 10-³…
A: From the table, molar concentration of tomato juice [ H+ ] = 1 × 10-4 M
Q: Rank the following 0.10 m aqueous solutions in order from highest freezing point (listed first) to…
A:
Q: Which of the following describes the main difference between electrolytic and galvanic cells? Eºcell…
A: We have to find out the main difference between electrolytic cell and galvanic cell.
Q: Determine the pH of a solution with [OH-] = 4.63 x 10-10 M. Your answer should contain 3 decimal…
A:
Q: Predict the major products of the following reaction. Be sure to show the stereochemistry of the…
A:
Q: What is pH of a 500mL solution with 0.165M diethyamine and 0.125 M diethammonium chloride? with…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: A student combined 45.3 mL of a 0.549 M calcium nitrate Ca(NO3)2 solution with 73.70 mL of a 1.321 M…
A:
Q: Calculate the emf of the following concentration cell: Mg(s) | Mg2+(0.25 M) || Mg2+(0.79 M) | Mg(s)
A: The concentration cell contains the same material on both half cell with different concentration of…
Q: Strong base is dissolved in 665 mL of 0.600 M weak acid (K₁ = 4.09 × 10-5) to make a buffer with a…
A:
Q: Humans are heterotrophs that consume food for energy. These foods comprise of sugars that are broken…
A: Since you have asked a question with multiple subparts, we will solve the first three subparts for…
Q: Consider the reaction 2NO(g) + O₂(g) 2NO₂(g) Using the standard thermodynamic data in the tables…
A: Here I am using the standard ∆G° values from the book.Using the standard values of ∆G°f for NO, O2…
Q: When triphenylphosphine is added to nickel(II) bromide dissolved in carbon disulfide at -78°C, a red…
A: We have given that When triphenylphosphine is added to nickel(II) bromide dissolved in carbon…
Q: The equilibrium constant, Kp, for the following reaction is 2.01 at 500 K. 7 PCI3(g) + Cl₂(g)…
A: Initial temperature (T1) = 500 KFinal temperature (T2) = 589 KK1 = 2.01K2 = ?ΔHo = -87.9 kJ=-87900 J
Q: 6. Calculate AHF of propane, C3H8, given the following information: 2C3H8(g) + 10 O2(g) →6CO2(g) +…
A: Answer:- This question is answered by using the simple concept of Hess law of constant heat…
Q: i. NaOMe ii. BnBr H i. NaOH iii. CH3Br
A:
Q: (COCI)2 DMF OH Hexane (CH;CH,),NH CI Hexane
A: Oxalyl chloride is also used for conversion of carboxylic acid to acid chloride.
Q: 1. Consider the two compounds below. xx a. What is their structural relationship? and X
A: Enantiomers are mirror images of each other and lack of plane of symmetry.
Q: The equilibrium constant, Kp, for the following reaction is 2.01 at 500 K. PCl3(g) + Cl₂(g) PCI,(g)…
A: We have T1 = 500 KT2 = 589 Kk1 = 2.01k2 = ?ΔH = -87.9 kJ
Q: Draw the explicit structures of the coordination compounds given below. a) Hexaaquatitanium(III)…
A:
Q: ) Does antifreeze ingredient ethylene glycol (HOCH2CH2OH; MM = 62.07 g/mol) have a lower or higher…
A: Intermolecular forces are the attractive or repulsive forces that exist between molecules or atoms…
Q: The pH and electrical conductivity of four different mixtures were measured: Mixtures Conductivity…
A: As per rule we will solve first three sub-parts of the question. kindly repost other parts as a…
Q: The pH of a basic solution is 10.15. What is pOH?
A: The relationship between pH and pOH of a solution is: pH + pOH = 14 ... (1)
Q: A geochemist in the field takes a 12.0 mL sample of water from a rock pool lined with crystals of a…
A:
Q: How many chemically inequivalent carbons are there in this molecule? Select an answer and submit.…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved full questions…
Q: Which of the following is the strongest base? T Toplantılar 3,33 Puan A B D CL CLOA NO3 F
A: A conjugate base is a species formed by the removal of a proton from an acid. The strength of the…
Q: IZ NÄY LiAlHy (Ht workup)
A: LiAlH4 is a strong reducing agent that reduces amide (-CONH2 ) to amine (-CH2NH2)
Q: Calculate the modulus of elasticity of a metal-matrix composite under isostrain conditions. Assume…
A: Ec=VmEm + Vf Ef Where,Vm =Volume fraction of matrixVf =Volume fraction of fibresEm =Elastic module…
Q: References Use the References to access important values if needed for this question. A chunk of…
A: Mass of iron = 18.1 g Initial Temperature of iron = 97.77°C Mass of water = 80.4 g Initial…
Q: A sample of solid chromium is heated with an electrical coil. If 74.6 Joules of energy are added to…
A: Amount of energy q =74.6 JMass of sample (m) = 10.6 gInitial temperature T1 = ?Final temperature T2…
Q: Copper has an isotope with an atomic mass of 62.93 and an abundance of 69.17%. A second isotope has…
A:
Q: Can someone please help me with this? The question is: Draw a reasonable mechanism for this…
A:
Q: What is the calculated value of the cell potential at 298 K for an electrochemical cell with the…
A: The given redox reaction is 2Ag+(aq) + Mn(s) →2 Ag(s) + Mn2+(aq) The concentrations of the species…
Q: Consider the reaction below. 2 HF(g) = H₂(g) + F₂(g) (K. = 1.00 x 10-²) Given that 1.00 mol of…
A: Given - Mole of HF(g) =1.00 mol Mole of H2(g) =0.312 mol Mole of F2(g) =0.750 mol Volume of Flask=…
Q: The following results were obtained for the below reaction in experiments designed for the study of…
A: Rate law of reaction is : Rate (r) = k[A]x [B]y Where x = order of reaction with respect to A y =…
Q: Create a flowchart to demonstrate how to separate a butyric acid and hexane mixture.
A: A separatory funnel is used to separate immiscible liquids. When two immiscible liquids are placed…
Q: What is the pH of a solution that is 0.753 M in ethylammonium chloride (CH3CH₂NH3CI)? The K, of…
A: Given , Kb(ethylammonia ) = 5.6×10-4
Q: Which of the products below would result from the indicated transamination reaction: A. I cooe +…
A: This reaction is dependent on coenzyme pyridoxyl phosphate that is PLP.
Q: 12- The force of attraction between a divalent cation and a divalent anion is 1.67 × 10-8 N. If the…
A: Force of attraction , F= 1.67×10-8 N divalent cation radii, r2+ =0.080 nm divalent anion radii,…
Q: In a chlorous acid (HClO2) buffer, chlorous acid will neutralize added __________. water hydroxide…
A: definition of a buffer is a solution with reserve acidity or alkalinity that resists pH change when…
Q: Determine whether this alkene has a configuration of E or Z. CI CO₂H O neither E nor Z OZ OE Which…
A: We have find out the answer.
Q: Design a synthesis for 3-(3'-chlorophenyl)-2-ethyl-2-propenal starting with butanol and benzyl…
A: The target molecule can be formed by major three steps. 1. Synthesis of 3-chlorobenzaldehyde 2.…
Q: Which of the following complex does not have any unpaired electrons? [FeC16]4- [Fe(OH2)6]²+…
A: 1) In first complex, the ligand Cl have -1 charge and overall charge on complex is -4. Thus, central…
Q: How to make 1% strach indicator in 250 ml water
A: Volume of water = volume of solution = 250 ml We have to make 1% of starch solution i.e starch…
Q: Sulfuric acid (H₂SO₂) reacts with sodium bicarbonate (NaHCO³) to produce sodium sulfate, carbon…
A: Given, Volume of sulfuric acid = 2.48 L Molarity of sulfuric acid = 3.00 M Mass of sodium…
Shbject: che
Step by step
Solved in 3 steps with 1 images

- Calculate volume and weight of sludge generated in thickener by chemical coagulation.Data given as:Wastewater flow: 10000 m3/dInlet concentration: 1100 mg/LFeCl3 added: 500 mg/LCaO added: 400 mg/LOverall particle removal efficiency: 80%Sludge’s specific gravity: 1.05Sludge’s moisture content: 95%Assume STANDARD chemical reactions and their product’s behavior as:FeCl3 + H2O → Fe(OH)3CaO + H2O → Ca(OH)21. Samples that should not be stored should be prepared/analyzed as soon as possible, especially for measurement…a. hardnessb. Nitritec. Conductivityd. Phenol 2. The following are general sample treatments in pesticide analysis, except…a. Extractionb. Cleaningc. Concentrationd. FilteringPlease provide Typed solution

