Done 7.1 Practice Question 5 View Policies Show Attempt History education.wiley.com Current Attempt in Progress Your Answer Correct Answer (Used) AA C C Which of the following is a good approximation for the change in the rate of a reaction when the temperature is increased by 10 °C? The reaction rate is only half as fast. The reaction rate approximately doubles. The reaction rate will stay the same. The reaction rate will increase by a factor of 4.
Done 7.1 Practice Question 5 View Policies Show Attempt History education.wiley.com Current Attempt in Progress Your Answer Correct Answer (Used) AA C C Which of the following is a good approximation for the change in the rate of a reaction when the temperature is increased by 10 °C? The reaction rate is only half as fast. The reaction rate approximately doubles. The reaction rate will stay the same. The reaction rate will increase by a factor of 4.
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.36PAE: The reaction NO(g) + O,(g) — NO,(g) + 0(g) plays a role in the formation of nitrogen dioxide in...
Related questions
Question
100%
Please explain me why the rate approximately doubles in the following question.
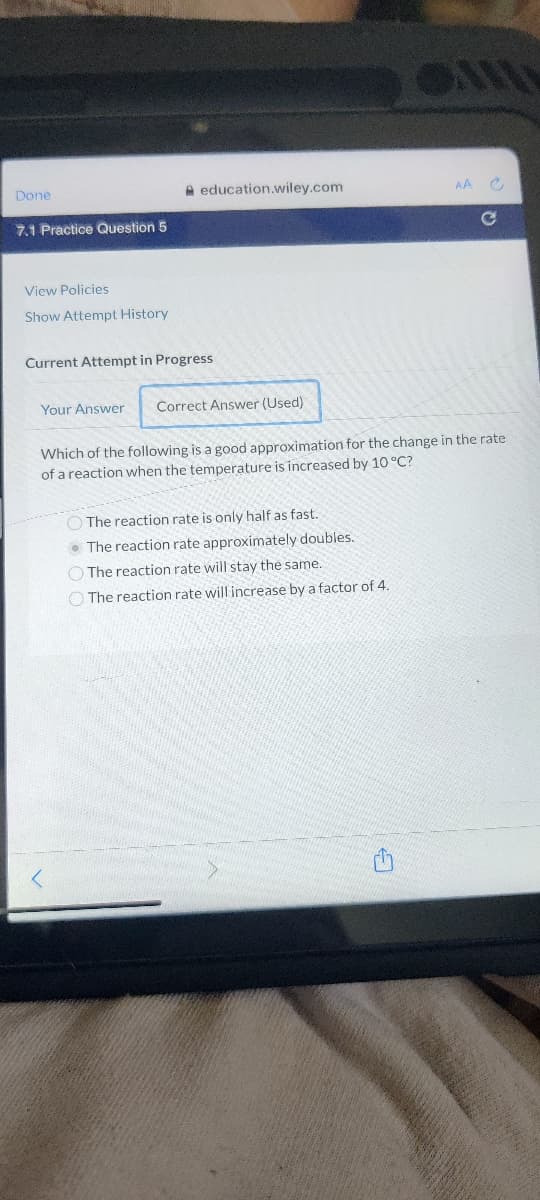
Transcribed Image Text:Done
7.1 Practice Question 5
View Policies
Show Attempt History
education.wiley.com
Current Attempt in Progress
Your Answer Correct Answer (Used)
AA C
C
Which of the following is a good approximation for the change in the rate
of a reaction when the temperature is increased by 10 °C?
The reaction rate is only half as fast.
The reaction rate approximately doubles.
The reaction rate will stay the same.
The reaction rate will increase by a factor of 4.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
