don’t understand the first part of the question with the: “You need 2 moles of magnesium…” How do I do that? I got the molar mass but my teacher says you don’t use that in the equation. If you can see, I set it up correctly but how do I get the moles? I am confused how
don’t understand the first part of the question with the: “You need 2 moles of magnesium…” How do I do that? I got the molar mass but my teacher says you don’t use that in the equation. If you can see, I set it up correctly but how do I get the moles? I am confused how
Chapter13: Isolation Of Eugenol From Clov
Section: Chapter Questions
Problem 9Q
Related questions
Question
I don’t understand the first part of the question with the: “You need 2 moles of magnesium…” How do I do that? I got the molar mass but my teacher says you don’t use that in the equation. If you can see, I set it up correctly but how do I get the moles? I am confused how to solve this and Sig Figs.
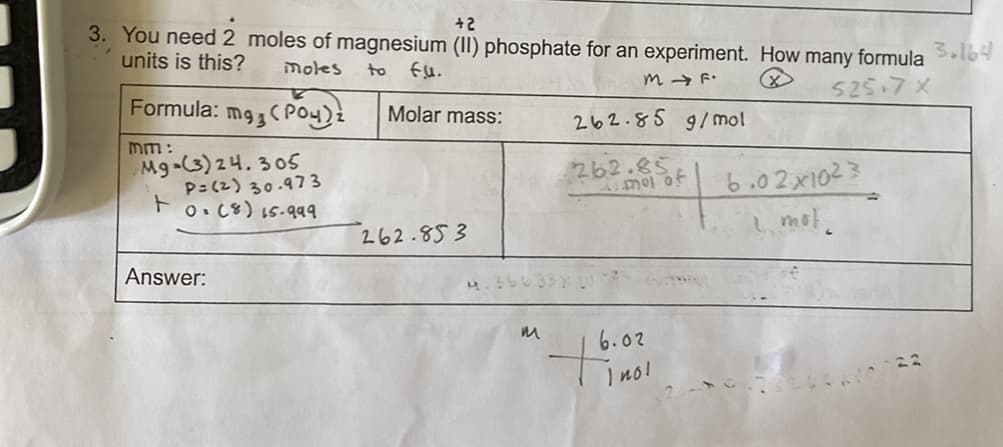
Transcribed Image Text:+2
3. You need 2 moles of magnesium (II) phosphate for an experiment. How many formula Sl64
units is this?
moles
to
m → F.
525.7x
Formula:
Molar mass:
262.85 9/mol
mm:
Mg-(3) 24.305
P=(2) 30.973
ト
262.85
6.02x1023
1, mot.
O. C8) 15.99
262.853
Answer:
6.02
ino!
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning