Draw a mechanism for the following reaction: H,C HC Br CH, H-Br CH H,C H,C Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron- flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H* EXP. CONT.
Draw a mechanism for the following reaction: H,C HC Br CH, H-Br CH H,C H,C Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron- flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H* EXP. CONT.
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter14: Elimination
Section: Chapter Questions
Problem 20E
Related questions
Question
Transcribed Image Text:14 of 15
Review Constants Periodic Table
Part A
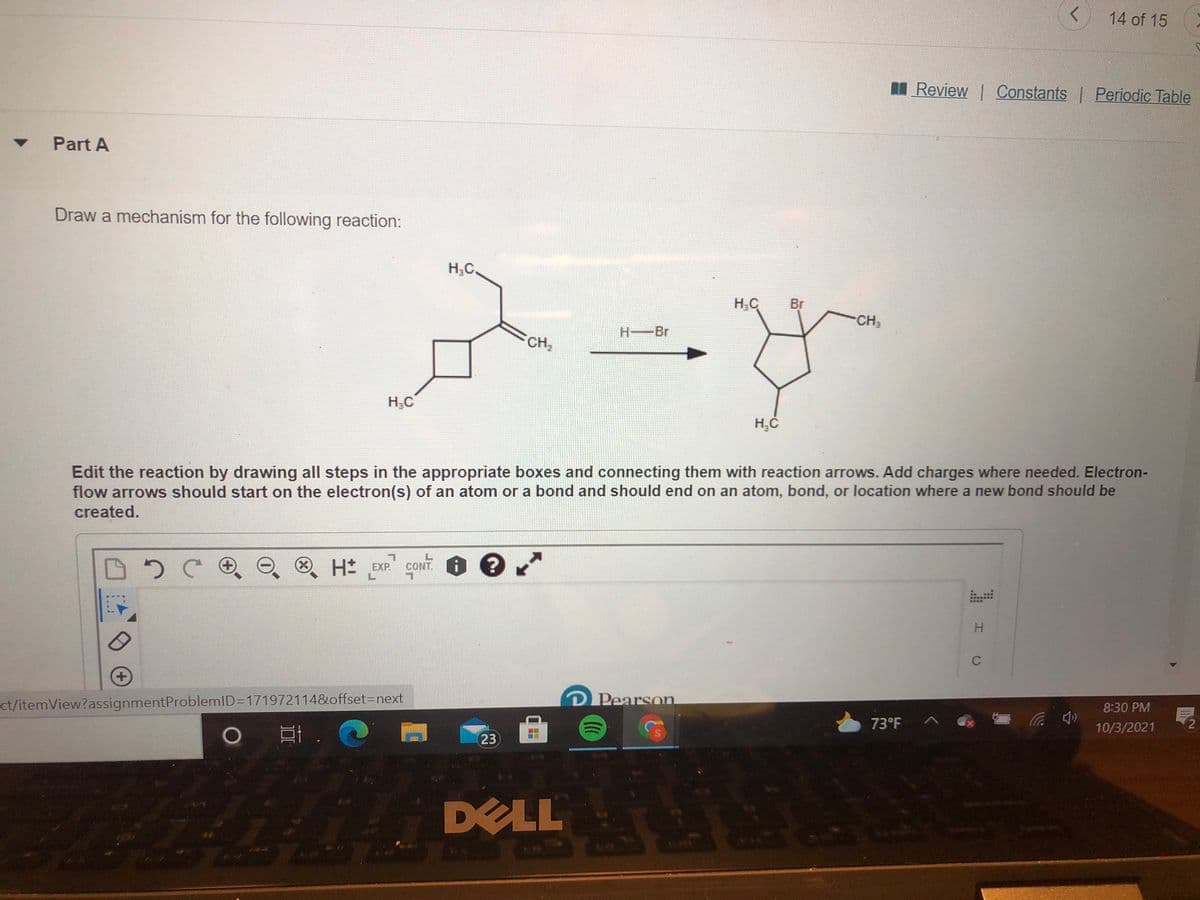
Draw a mechanism for the following reaction:
H;C.
H,C
Br
CH3
H-Br
CH2
H,C
Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron-
flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be
created.
CONT. O e
(X)
O H EXP.
C
ct/itemView?assignmentProblemID=1719721148&offset3Dnext
Pearson
8:30 PM
73°F
口i. @
10/3/2021
23
DELL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
