Q: 2. The concentration of NH4CI solution (aqueous solution) is 33.5% by mass. If the mass of solution ...
A:
Q: 4. What is the concentration of PM10 in air in ppmv if its concentration is 0.15 mg/m3? Assume atmos...
A: Given the concentration of PM10 (molecular weight = 12 g/mol) in air = 0.15 mg/m3. Atmospheric press...
Q: What is the retention factor (Rf) of the most polar and least polar spot present in the standard ink...
A: We have to give the retention factor (Rf) of the most polar and least polar spots present in the sta...
Q: a. Alcohol b. Sea water c. Water d. Mercury e. Dead Sea f. Acetone Among those choices, which one ha...
A: Intramolecular forces are the forces that hold atoms together within a molecule. Intermolecular forc...
Q: Which of the following islare TRUE about molar conductivity? I. The molar conductivity provides info...
A: 4th option is correct that is I, II ,III Or I.The molar conductivity pr...
Q: (c) Outline the mechanism for the bromination of 3-methylcyclobutene.
A:
Q: KOGRESS CHECK NO, 3 Rank the following sets of substituents in order of priority according to the Ca...
A:
Q: 4. Starting from the universal gas constant (R = 8.314 1.30 moles of an ideal gas at 27.0°C and a pr...
A:
Q: 1. Using the equation E + S ⇆ ES → E + P, supply reaction involved in the experiment... 2. How did ...
A: A question based on catalysis that is to be accomplished.
Q: e AH FAY
A:
Q: Starting with the following compounds, outline a practical synthesis of 1-butanol: 1. 2-chlorobutane...
A: Given, 1. 2-chlorobutane -----> 1-butanol 2. 1-butyne -------> 1-butanol
Q: Calculate the pressure exerted by CO2 in a cola bottle if its molar solubility in the solution is 1....
A: Henry law: This law states that the concentration of a gas dissolved in a liquid depends on the part...
Q: Question 2 The AGʻrxn for the reaction: A (g) + B (g) 2C is +192 kJ/mol at 25 °C. What is the equili...
A: Equilibrium constant expresses the relationship between the concentrations of reactants and products...
Q: IN B C. LI %#3 Assume that it is possible to heat water with perfect insulation (all the heat from c...
A: Assume that it is possible to heat water with perfect insulation (all the heat from combustion of et...
Q: MgCl2 + Fe(NO3)3 Balanced equation: Net ionic:
A: The balanced equation and net ionic equation for the above equation is given below
Q: Кы(еxperiment)
A:
Q: Compare the following: Acid 1: hydrofluoric acid , HF Acid 2: hydrosulfuric acid , H2S Acid 3: sulfu...
A: Acid is a substance which donate H+ ion and form well stabilised conjugate base
Q: How many aromatic amino acids are present in vancomycin? Identify and color it.
A: Vancomycin has a total of 7 amino acids. Aromatic acids have a benzene ring.
Q: Determine the [H3O+] of the eight buffer solutions made by mixing 1.10 M acetic acid and 0.900 M sod...
A:
Q: molar masses
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any spe...
Q: For the hydrolysis reaction of a nitrile with H30+, H30+, which of the following is produced interme...
A: the hydrolysis of nitriles under acidic conditions to make carboxylic acids or their salts while a...
Q: A 0.543-g sample of solid magnesium is burned in a constant-volume bomb calorimeter that has a heat ...
A:
Q: Draw the major monobromination product when the alkane shown is subjected to radical bromination at ...
A: The reaction is a free radical bromination of alkane in presence of light.
Q: Which one has the highest and strongest intermolecular forces and why? a. Alcohol b. Sea water c. Wa...
A: A question based on intermolecular forces that is to be accomplished.
Q: 14. Including stereochemistry, the correct IUPAC name for the following compound is: Name:
A: (5S, 4S)-5-bromo-4-chloro-2-flouro-3-methylhex-2-ene
Q: What is the pH of a 0.069 M weak acid solution. K, for the weak acid is 5.4x10-6.
A:
Q: 2.48 The molar heat capacity of for temperatures in the range 300 to erate pressures can be approxim...
A: Number of moles = 2.00mol Gas constant, R = 1.987cal/molK
Q: A 5.96 g sample of a compound that is not an electrolyte is dissolved in 212.0 g of benzene. The fre...
A: Freezing point depression = i × m × Kf
Q: Preparation of HCI solution a. Measure 20.7 ml of concentrated Hcl (37% wt HCI) using the pipet and ...
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out o...
Q: According to Kolrausch's law of independent ionic migration, the molar conductivity of an electrolyt...
A:
Q: Indicate which of these terms can be used to describe the first step of the following reaction. More...
A: Regioselectivity refers breaking or forming of bonds in particular position favourable by attacking...
Q: How can the use of non-stick cookware help reduce the amount of fat in our diet? What risks are asso...
A: We use nonstick cookwares to reduce the amount of fat in our diet.
Q: Describe all of the intermolecular forces Carbon dioxide would experience. Justify your answer using...
A:
Q: What volume of 0.150M HNO3 solution is needed to exactly neutralize 35.0mL of 0.150M NaOH solution? ...
A: Nitric acid reacts with sodium hydroxide to form sodium nitrate and water. The equation for the bala...
Q: -concentration is reg
A:
Q: Determine the amount in grams of NaCl needed to make the following solution 1.5 % NaCl in a total v...
A:
Q: Some amount of a diprotic acid is dissolved in 100 mL of H20. 70.0 mL of 0.100 M NAOH is added to re...
A:
Q: 4. Starting from the universal gas constant (R 8.314 ), determine the volume (in m³) occupied by 1.3...
A: Given : 4. Moles = 1.30 moles Temperature = 27.0 °C = 300 K Pressure = 2.03 x 105 Pa 5. Volume...
Q: Compute the empherical formula of the following: a. 19.16 Sn and 80.9% I b. 25.9% Fe and 74.1% Br
A: With the help of percentage of composition of elements and their atomic masses we can determine the ...
Q: A sample of methane gas at a pressure of 0.947 atm and a temperature of 29.0 °C, occupies a volume o...
A: According to Boyle's law, at constant temperature with increase of pressure of ideal gas volume decr...
Q: Write out the molecular, ionic, and net ionic equations for the neutralization reaction between sulf...
A: KOH(aq) + H2SO4(aq)
Q: The acid ionization constant for Ga(H20)63+(aq) is 2.5x10-3. Calculate the pH of a 0.1120 M solution...
A:
Q: The acid ionization constant for Pb(H20)62+(aq) is 6.3×10-7. Calculate the pH of a 0.0307 M solution...
A:
Q: Experiment name : Heat of Combustion of benzoic acid and Naphthalene in Bomb Calorimetry Q1:Purpose ...
A:
Q: temperature of the .f
A:
Q: W̲h̲y̲ ̲a̲r̲e̲ ̲s̲o̲m̲e̲ ̲m̲a̲t̲e̲r̲i̲a̲l̲s̲ ̲c̲o̲l̲o̲r̲e̲d̲ ̲a̲n̲d̲ ̲o̲t̲h̲e̲r̲s̲ ̲a̲r̲e̲ ̲n̲o̲t̲?̲...
A: Materials appear different colours because they absorb some colours of definite wavelength and refle...
Q: Mercury and bromine will react with each other to produce mercury (II) bromide: a. What mass of HgBr...
A: The mass of a compound can be calculated by the number of moles of the respective compound from a ba...
Q: For each system listed in the first column of the table below, decide (if possible) whether the chan...
A: Entropy increases when liquid or gaseous molecules get more space to move.
Q: 4. Predict the shift in the following equilibrium system resulting from each of the following change...
A: #4: Since the energy is on the product side, energy is released during the reaction and the reaction...
Q: What is the ppm of 15 mg of NaCl crystals dissolved in 850 mL of distilled water?
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

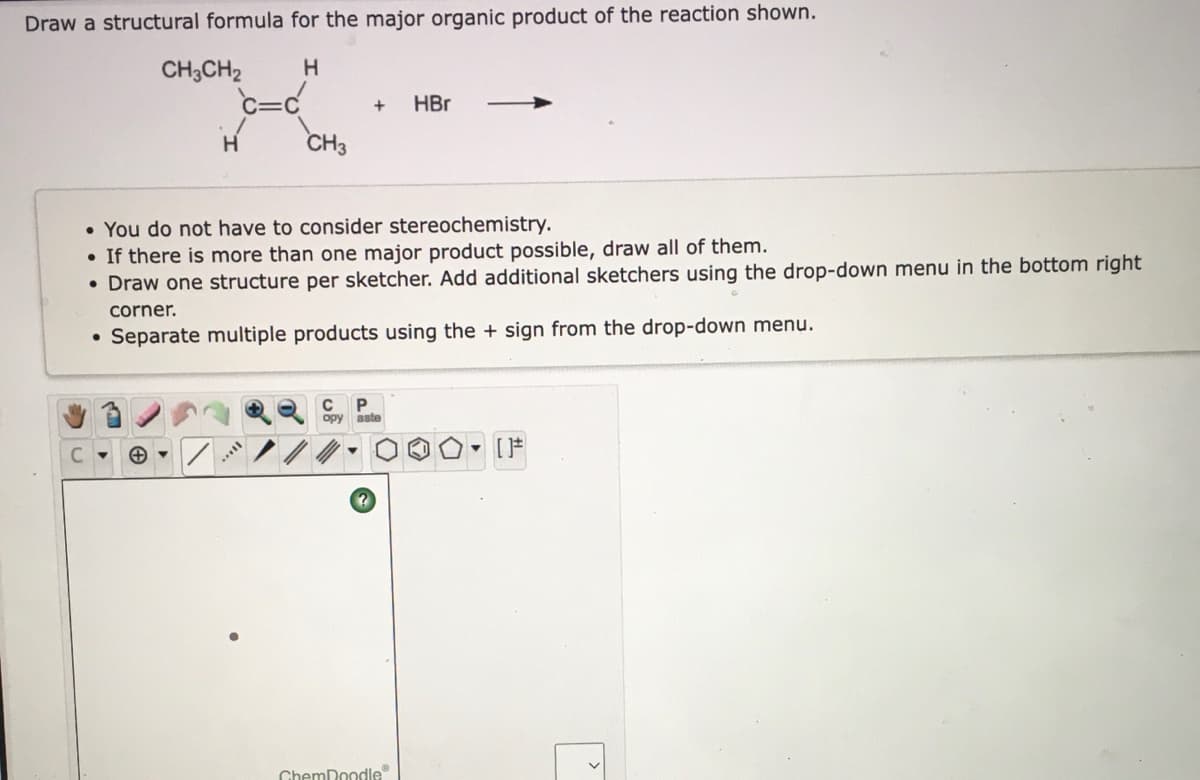
- Draw the structure of the major organic product(s) of the reaction. You do not have to consider stereochemistry. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.Draw the structure(s) of the major organic product(s) obtained after workup of the following reaction. You do not have to consider stereochemistry. If no reaction occurs, draw the organic starting material. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.Draw the structure(s) of the major organic product(s) obtained after workup of the following reaction. You do not have to consider stereochemistry. If no reaction occurs, draw the organic starting material. Draw structure(s) of product(s) after workup to neutralize acid. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
- This question has multiple parts. Work all the parts to get the most points. a Draw all of the monochlorination products that you might obtain from the free-radical chlorination of 2,2,4-trimethylpentane. You do not have to consider stereochemistry. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.What is the main organic product (B) of the reaction scheme in Figure 11?Draw the organic product(s) of the following reaction. You do not have to consider stereochemistry. Include cationic counter-ions, e.g., Na+ in your answer, but draw them in their own sketcher. If no reaction occurs, draw the organic starting material. Separate multiple products using the + sign from the drop-down menu.
- Orgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???1) What is the major product for the reaction of 3-bromo1-butene and HBr? Give the name of the product. 2) For this product, how many chiral carbon(s) are there in the structure?3) How many stereoisomers could we write for this product? 4) Write the Fischer Projections of all stereoisomers. (attach a file or other ways you could show your work)Enters the preferred IUPAC name of the connection in Figure 29.
- First image directions are "Fill in the blanks with the major organic product(s). Make sure to indicate stereochemistry in your drawings where appropriate." Second image directions are "Fill in the blanks with the major organic product or starting material(s)."F ONLY- ( STRUCTURAL ANSWER ONLY)Draw the structure(s) of the major organic product(s) obtained after workup of the following reaction. Use the wedge/hash bond tools to indicate stereochemistry where it exists. If no reaction occurs, draw the organic starting material. Draw structure(s) of product(s) after aqueous workup. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.

