Q: how to do step-by-step Lewis structure for FeBr3
A: A Lewis Structure is defined as a highly simplified illustration of the valence electrons in an…
Q: tion 3 of 30 a) Which molecules have polar bonds? H. 1. CH, CH, CH H,C CH, CH, Ch H.
A: Polar bond A polar bond is a certain class of a covalent bond. We can also say that it is the…
Q: The compound above is an example of a(n)
A:
Q: A 1.51 How are the molecules or ions in each pair related? Classify them as resonance structures,…
A: Isomers have the same number of elements in the compounds, but with different structures, Resonance…
Q: Follow the curved arrows to draw a second resonance structure for eachspecies.
A: Given species,
Q: What orbital(s) are the lone pairs of electrons on the O atom placed? - What orbital is the lone…
A: The given molecular structure is known as peptide where the carboxylic acid group of one amino acid…
Q: What do resonance structures represent, such as the one shown? Please select all the correct…
A:
Q: CH3S+ pg In this structure, what is formal charge on S? Please click on jpg file to see image 0. O…
A: The formal charge on the atom in a molecule can be calculated as follows Formal charge = Valence…
Q: Draw a regular Lewis structure for the following molecule and label every bond using the notation.…
A:
Q: Considering structures A–D, classify each pair of compounds as isomers, resonance structures, or…
A: Isomer:Isomers are the organic compounds that have same molecular formula but different molecular…
Q: Consider compounds A-D, which contain both a heteroatom and a double bond. N. H. A B a. For which…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: Use the octet rule to determine which is a correct structure. (Only one is con A B нн нн Н-с—ҫ—о-н…
A: To determine the correct structure examine each structure carefully..
Q: Lewis structure name of highlighted group H :0 С — н H :0 — Н H :0: H -C- N - H н н - -
A:
Q: How are the molecules or ions in each pair related? Classify them as resonance structures, isomers,…
A: a.
Q: : NH
A: When one Lewis structure of a single molecule cannot fully explain the bonding that occurs between…
Q: a) Draw the Lewis structure for NH2CH3. S b) What is the shape or geometry at the nitrogen? c) What…
A: Solving only first three subparts in accordance with the guidelines. You can repost remaining…
Q: e Lewis structure for
A:
Q: In this structure, what is formal charge on S? O-S=O.jpg Please click on jpg file to see image O +1…
A:
Q: How are the molecules or ions in each pair related? Classify them as resonance structures, isomers,…
A:
Q: Draw the Lewis structure of the missing reactant. Make sure to include lone pairs and non-zero…
A:
Q: Which structure represents a correct resonance structure for the following structure? +) +) :o:
A:
Q: -H- C-C
A: Resonance comes into play when it is possible to draw more than one Lewis structure of a given…
Q: Which of the given resonance structures (A, B,or C) contributes most to the resonance hybrid? Which…
A: Most contributing structure as be seen on the basis of stability and stability can be seen as:…
Q: Use formal charge to choose the best Lewis structure for CH3SOCH3. H :ö H H :ö H B) H-C=S-Ċ-H A)…
A: To determine the best Lewis structure using formal charges, following rules must be followed: In a…
Q: Click to see additional instructions The following questions are about the best Lewis structure of…
A: See the lewis structure of O3 +1 Single bond, double bond, and lone pairs of electrons.
Q: draw all possible lewis/resonance sturcures for CBr2O and circle/explain the best one
A: All the contributing structures for the CBr2O are given as follows.
Q: Although we usually don't show lone pairs on skeletal structures, sometimes it's necessary, for…
A:
Q: H H H H H H H H of a) Name the molecule diagramed above.
A: The name of the diagram is
Q: Lewis structure name of highlighted group Н — С: | Н — С — Н - Н —N— Н .. н н Н — С — С - Н —С — Н H…
A:
Q: Draw the Lewis structure of SF,. Include all the lone pairs. 2° Select Draw Rings More F
A: Lewis structures can be defined as the structures that show the bonding between atoms of a molecule…
Q: CH3
A:
Q: Which molecules have polar bonds? А. A CI B =. E. ö- В. CI Е F H н F. О- Which are polar molecules?…
A: In the formation of the chemical bond with the same or different elements, all elements tend to…
Q: Lewis structure name of highlighted group H H .. H C -О — Н - н н
A:
Q: Draw the lowest energy Lewis structure of CN^-. What is the formal charge on the carbon Adam
A: Lowest energy Lewis structure have more number of covalent bonds. Since valency of N atom is 3 so…
Q: 1.51, How are the molecules or ions in each pair related? Classify them as resonance structures,…
A: a. Both has same molecular formula C6H12O. But the first one contains six membered ring while second…
Q: Formula Model Molecular geometry BeCl, ball & stick labels PO,(OH); ball & stick labels NO;CI ball &…
A:
Q: How are the molecules or ions in each pair related? Classify them asresonance structures, isomers,…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.
Q: What is the number of σ and π bond inN ≡ C – CH = CH – C ≡ N?
A: N ≡ C – CH = CH – C ≡ N The number of sigma and pi bonds are given.
Q: For each full Lewis structure below, draw a bond-line structure. H. ннн b) : Br-C-с-с-н н нно-н н…
A: Change the given lewis structure into bond line structure-
Q: Draw the Lewis structure for acetic acid (C,H,0,). Acetic acid molecules have one hydroxy group and…
A: Lewis Structure: It is a very simplified representation of the valence shell electrons in a…
Q: 23. Write another resonance structure for ethyl acetate. Include formal charges. CH, ÖCH,CH,
A:
Q: Draw all possible resonance structures for the species below: H а. b. с.
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.
Q: Attached the curved arrows to draw a second resonance structure for each ion.
A: Given allyl carbocation and enolate ions are :
Q: Draw all possible resonance structures for NPO in your work. Then, assign formal charge to each atom…
A: The compound given is NPO.
Q: Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in. H…
A:
Q: How many o and ä bonds are in this molecule? H EC-H number of o bonds: number of 7 bonds:
A:
Q: Compare the two values below: A number of lone pairs on central atom in NH, B number of lone pairs…
A: Correct answer is (a) A is larger than B. Number of lone pairs on central atom in NH3 = 1 A = 1…
Q: ough we usually don't show lone pairs on skeletal structures, sometimes it's necessary, for example,…
A: Lewis structure is a method of of representing bonding pattern in a molecule. it represent how…
Q: how are the molecules or ions in each pair related2 Classify them as resonance structures, isomers,…
A: Answer for a. Both the structures are functional isomers Answer for b. Both the structures are…
Q: a. Se, b. 20. Show the direction of the dipole in each bond. Label the atoms with &+ and 8-. a. H-F…
A: Here the atom which is more elctronegative will carry fractional negative charge and less…
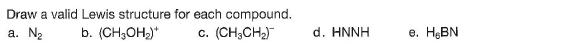
Step by step
Solved in 3 steps with 3 images

- How can you convert CsCl into NaCl like structure?Good hand written explanation Asap A molecule of chloroform, CHCl3, has the same shape as a molecule of methane, CH4. However, methane’s boiling point is -164 degreeC and chloroform’s boiling point is 62 degree C. Explain the difference in boiling points, using 3D Lewis Structures to aid your simple explanation.Draw a second resonance structure and the hybrid for each species, and then rank the two resonance structures and the hybrid in order of increasing stability.
- Draw the lowest energy Lewis structure of CN^-. What is the formal charge on the carbon Adam?Make a model of propane (C3H8), and draw this model using dashed lines and wedges to represent bonds going back and coming forwardHow are the molecules or ions in each pair related? Classify them as resonance structures, isomers, or neither.

