Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter4: Polar Bonds, Polar Reactions
Section: Chapter Questions
Problem 11CTQ
Related questions
Question
Please click on the image if the problem isn’t showing up on default. Thank you :)
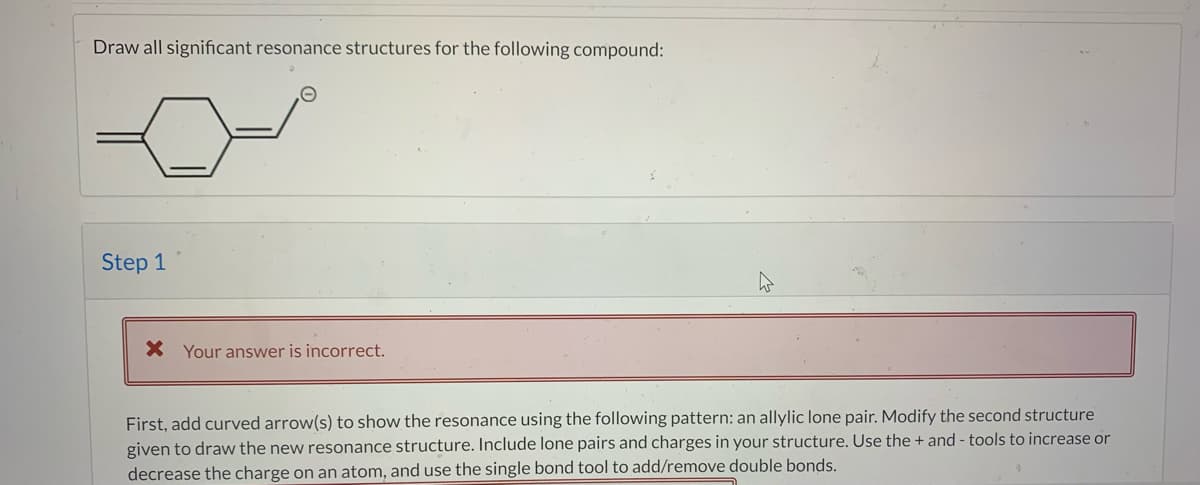
Transcribed Image Text:Draw all significant resonance structures for the following compound:
Step 1
X Your answer is incorrect.
First, add curved arrow(s) to show the resonance using the following pattern: an allylic lone pair. Modify the second structure
given to draw the new resonance structure. Include lone pairs and charges in your structure. Use the + and - tools to increase or
decrease the charge on an atom, and use the single bond tool to add/remove double bonds.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
