Q: b. Draw the FULL electron-pushing mechanism for the reaction depicted including ALL intermediates…
A:
Q: Which is the least stable carbocation?
A: In order to determine the stability of carbocation we have to check following factors sequencly 1)…
Q: Add curved arrows to draw the first part of the mechanism: tautomerization of a ketone to an enol.…
A: Keto-enol is a type of structural isomerism which involves a keto form (containing C=O) with alpha…
Q: H3C CH3 H3C CH2 H А. В.
A: Here both nucleophiles have one and two acetyl groups respectively and the lone pair (negative…
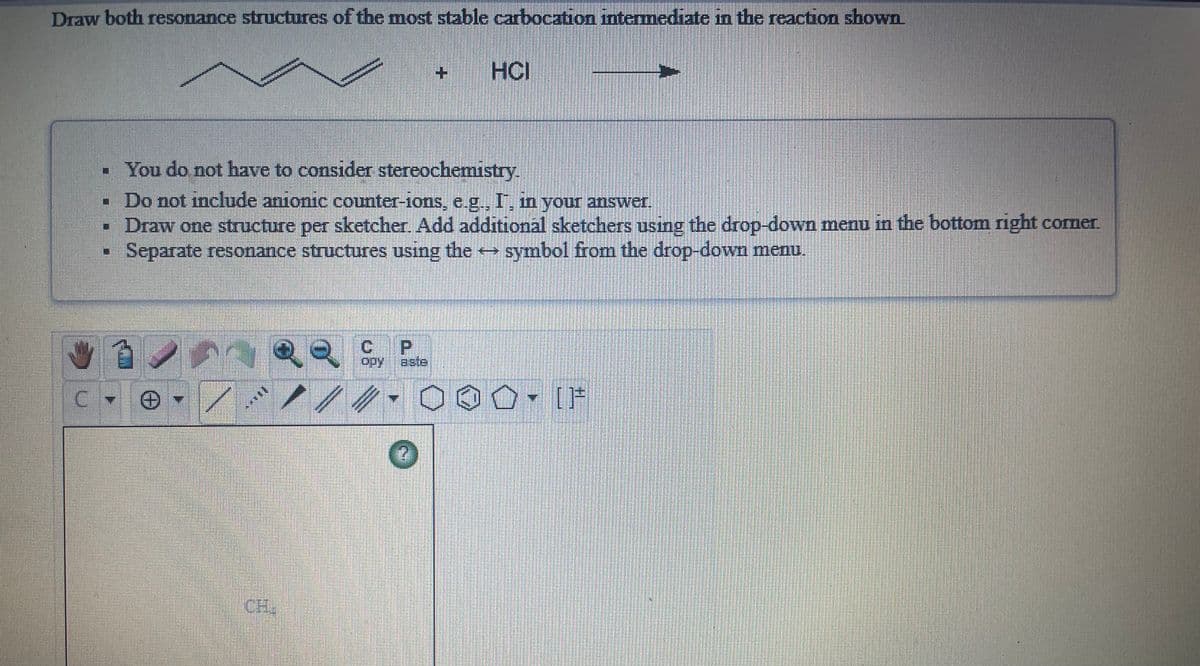
Q: Draw both resonance structures of the most stable carbocation intermediate in the reaction shown.
A: The attack of H+ on the given molecule is shown. A secondary allylic carbocation is formed.
Q: Based on Markovnikov’s rule, draw only the Major product for the following asymmetrical addition…
A: Given data : Major product
Q: Draw the FULL electron-pushing mechanism for the reaction, including ALL intermediates (with formal…
A: The ester hydrolysis mechanism for the given reaction is given below.
Q: Draw the most stable resonance form for the intermediate in the following electrophilic substitution…
A: The mechanism is as :-
Q: Draw the most stable resonance form for the intermediate in the following electrophilic substitution…
A: The reaction of chlorobenzene with acetyl chloride in presence of AlCl3 is a Friedel – Crafts…
Q: Rank the attached carbocations in order of increasing stability?
A: The order of stability of carbocations is: Tertiary carbocation > secondary carbocation >…
Q: Draw the two resonance structures of the carbocation intermediate formed in this reaction. Do not…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: Major product in the reaction
A: Common catalysts used are insoluble metals such as palladium in the form Pd-C, platinum in the form…
Q: Zaitsev's Rule refers to: O how resonance contributors stabilize cations/anions/radicals O which…
A:
Q: most stable to least stable nucleophiles
A: Nucleophiles are the chemical species which are nucleus loving and donates an electron pair to form…
Q: Draw all the products of the following reaction:
A: Please find below all the possible products of the above reaction.
Q: dissociation NaBr Select to Draw Carbocation Intermediate Carbocation Rearrangem ent Select to Draw…
A:
Q: Draw the carbocation rearrangement product of the secondary carbocation shown.
A: Rearrangement of the carbocation.
Q: Draw enol tautomer(s) for each compound. Ignore stereoisomers.
A: a) Please fine below the enol tautomer for compound 1
Q: What is more stable acetate (CH3COO-) or ethoxide (CH3CH2O-)? In three words or less explain your…
A: Stability of anion: Primary carbanion is more stable than secondary and tertiary carbanions due to…
Q: Draw the stereoisomers of the major monobromination products obtained from the following reaction.
A: It is given that 1,3-dimethyl cyclohexane is undergoing monobromination reaction in the presence of…
Q: Sometimes carbocation rearrangements can change the size of a ring. Draw a stepwise, detailed…
A: The -OH group of alcohol abstracts a proton (from acid) as shown below:
Q: Draw the curved arrow that explains how the product is formed. H H H H H + H
A: ∘Electron density from higher e- density region to lower e- density region.Given,
Q: Draw the two resonance structures of the carbocation intermediate formed in this reaction. Do not…
A:
Q: Rank the following radicals in order of decreasing stability. most stable least stable (CHдС-снсн,…
A: Free radicals are electron deficeint. the stability of tertiary radicals is more than that of…
Q: Use Hammond's postulate to determine which alkene in each pair would be expected to form a…
A:
Q: Rank the following carbocations in order of decreasing stability. Most stable
A: Carbocations are electron-deficient species that are formed by the removal of a hydrogen atom from…
Q: Draw the two resonance structures of the carbocation intermediate formed in this reaction. Do not…
A: Alkene or pi bond compounds on reaction with H+ to give the corresponding carbonation (an…
Q: Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. +…
A: Given : structure of reactant and reagent. This is a electrophilic addition reaction
Q: Draw all possible products in the following reaction and circle the most stable product. Br КОН ELOH
A: In this question, we will draw all products for this reaction and the circle the most stable…
Q: If the H colored red is eliminated as H*, then two possible diastereomers can form. Draw the curved…
A:
Q: For alkylhalides, elimination reaction is much favored than substitution reaction when A.…
A: Alkyl halides undergoes substitution reaction namely SN1 , SN2 .They also undergo elimination…
Q: The Markovnikov product resulting from an addition reaction to an unsymmetrical alkene is formed…
A: -> In markovnikov's addition reaction reaction proceeds through formation of the most stable…
Q: Please draw the possible resonance structures for the following carbocations. Please demonstrate…
A:
Q: 2. Draw the complete, detailed El mechanism for the following reaction (including including curved…
A: Tertiary alkyl halide gives E1 elimination to form an alkene. The alkyl halide eliminates hydrogen…
Q: Draw the product or products of the given reaction. Include charges where appropriate.
A:
Q: Rank the following radicals in order of increasing stability.
A: Since the stability of free radicals depends on two things 1) resonance => more is the resonance…
Q: Draw the carbocation rearrangements for each of the following carbocations. ye
A:
Q: ng carbocations
A:
Q: Draw both resonance structures of the most stable carbocation intermediate in the reaction shown.
A: The cation that acts as a intermediate in the reaction is known as carbocation. In carbocation,…
Q: Draw the major intermediate and product of the reaction between (2E)-4-methylhex-2-ene and Br, in…
A:
Q: Please draw the mechanism for the following reaction. The first step in the reaction mechanism is…
A:
Q: Which carbocation is more stable? Briefly explain why. A B Ph, + Ph Ph Ph
A: Since, We know that, More is the resonating structure , more will be the stable carbocation formed.
Q: The carbocation intermediate formed in the first step is resonance-stabilized. Draw the other…
A: Resonance means delocalisation of pie electrons. Resonance gives stability to the intermediates like…
Q: Draw
A: Missing reagent should be nucleophile which attack on carbonyl carbon.
Q: Draw a curly arrow mechanism to clearly describe why the following reaction gives a trans dibromo…
A: Reaction of alkenes with bromine results in the formation of vicinal dibromides i.e. 1,2-dibromides.…
Q: Determine the product for the following Claisen rearrangement. heat OH
A:
Q: Draw the major product of the following reaction a). Remember to include the stereochemistry of the…
A: Given reactions are acetalisation reactions.
Q: Rank the following radicals in order of increasing stability.
A: The stability of free radical is governed by various factors such as: Allylic / benzylic radical 3°…
Q: Which is the least stable carbocation? A. В. С. D. Select one: D
A: Correct answer is (B) Carbocation B is least stable because it is not stabilized by resonance and…
Q: Which is more stable?
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw the structure(s) of the major organic product(s) obtained after workup of the following reaction. You do not have to consider stereochemistry. If no reaction occurs, draw the organic starting material. Draw structure(s) of product(s) after workup to neutralize acid. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.please point out the ones that are the resonance structure of the most stable carbocation, please.What is the main organic product (B) of the reaction scheme in Figure 11?
- Draw out the mechanism to explain this reaction - pls include formal charges, lone pairs, arrows and a transition state - use dashes and wedges - lastly what is the stereochemistry of this rxn? why doesn’t it apply to this situation?Rewrite the entire reaction in Full Structure Format with the Ending Products [The second image is an example of Full Structure Format ] THANKSSSSSSSSSSSSPlease help me with the organic chemistry problem below: Consider the reaction below: (Check the attached image) (it is between Furan and maleic anhydride, a DIels-Alder reaction) a) Will this reaction for an endo product (with a melting point of 80-81 degrees) or the exo product (with a melitng point of 114 degrees)? b) Carefully explain why the product must have been formed the way it did (exo or endo). c) Provide a mechanism for this reaction.
- Draw the structure(s) of the major organic product(s) obtained after workup of the following reaction. Use the wedge/hash bond tools to indicate stereochemistry where it exists. If no reaction occurs, draw the organic starting material. Draw structure(s) of product(s) after aqueous workup. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.The question is: "Draw the curved arrow mechanism for the reaction between (2S,3S)-3-methylhexan-2-ol and PCl3. Note the specific instructions for each box. Include nonzero formal charges and lone pairs of electrons on all appropriate atoms" I attached screenshots of the picture of the atoms below. What type of reaction (SN1, E1, SN2, or E2) would the reaction be, and how would these molecules interact? The question asks for multiple steps so I am guessing it is either SN1 or E1, but what is the reaction mechanism?Carbocations rearrange with some ease. An example of this is shown below. Indicate, with the corresponding arrows, how this process occurs. The carbocation 2 is more stable than 1, why is this? Explain it using (drawing) structures.
- Draw the most stable carbocation that can be formed by the compound in Figure 5.1 example of reaction for each of the types of organic reactions: substitution, elimination, addition, and rearrangement. Show:a. the overall reaction (reactants --> products)b. the reaction mechanism (indicate intermediate product)c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?)d. overall description of the reaction eg., radical substitution or SRHighlight the electrophilic carbon in red, and highlight the leaving group in blue. Highlight the atom in the nucleophile that will attack the electrophilic center in green. Only atoms need to be highlighted and not the lone pairs or formal charges

