Q: From the abbreviated name of the compound Gal (β1->4) Glc, we know that— C4 of galactose is…
A: From the abbreviated name of the compound Gal (β1->4) Glc, we know that— C4 of galactose…
Q: Write the complete electron configuration for the chlorine atom. Using NOBLE GAS notation write the…
A: Electronic configuration of element is define as the arrangement of electron in the orbitals. where…
Q: 8.2 mol NH3 are placed in a 10.5 L container at some temperature and allowed to come to equilibrium…
A: Initial moles of NH3 = 8.2 mole Volume of container = 10.5 L Moles of NH3 at equilibrium= 2.2 mole…
Q: Melting Point Determinaion Table A.1: Melting Points Compound Salicylic Acid Aspirin Experimental…
A: Melting point is defined as the temperature at which a solid starts melting and becomes liquid.…
Q: 7
A:
Q: Question 10 of 10 H₂C Provide the correct IUPAC name for the compound shown here. CH₂ CH₂ H₂C 1 H₂C…
A: The question is based on the concept of IUPAC naming. We write IUPAC name of given organic compound…
Q: A 19.75 g (mmetal) sample of metal is heated to 98.6°C (Tmetal initial) in a hot water bath until…
A:
Q: What is the percent by volume of ethanol (C₂H6O) in the final solution when 135mL of ethanol is…
A: Given, volume of ethanol = 135 mL volume of solution = 345 mL To find : percent by volume of…
Q: A reaction mixture (mixture 1) at 298 K has a N2O4(g) partial pressure of 0.1 bar and a NO2(g)…
A:
Q: 0.115 M KOH solution was used to titrate a 32.90 mL sample of a weak acid solution, HC3H5O2, of…
A:
Q: Provide the major organic product for each of the reactions below. If a specific stereochemistry…
A: The reaction of alkene with B2H6 then H2O2 , NaOH is known as hydroboration - oxidation reaction.…
Q: The half equivalence point in the titration of 0.100 M HCO2H (K a = 1.8 × 10–4) with 0.250 M NaOH…
A:
Q: Identify the expected major product of the following electrocyclic reaction. de 111 hv ? B IV
A: The given reaction is shown below The given options are We have to select the major product of the…
Q: The production capacity for acrylonitrile (C3H3N) in the United States is over 2 billion pounds per…
A: A limiting reagent is the species that is used up first in the chemical reaction. It decides the…
Q: Which of the following processes would absorb the most amount of heat energy (for 1 g of H20)? a)…
A: Fusion: It is process of conversion of solid into liquid state Condensation: It is process of…
Q: Solid zinc metal reacts with NO to make Zn(OH)4² and gaseous ammonia (NH3) in a solution with pH 8.2
A: Since you have asked multiple questions, we will solve the first one for you. If you want a specific…
Q: ning. TOUT SLUTC Will be based on the TUTTINCT OF Cunelmans minds the five on nel malenes. mere is…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 1) For each of the reactions below, draw the whether the relative stereochemistry of syn or unknown.…
A: We know , HBr helps in hydrobromination of double bond and mCPBA helps in epoxidation across double…
Q: A 0.10 M NaC2H3O2 (sodium acetate) solution in water has a pH of 8.4. Sodium acetate dissolved in…
A: Answer: CH3COONa is the salt of weak acid CH3COOH and strong base NaOH and we know that we acid gets…
Q: Indicate if you should add/increase or remove/decrease the following stresses when applied to the…
A: If more of the reactant are added then According to Le Chateliar principle , the effect will be to…
Q: 8.1 10 ultur 8 7.9 7.8 7.7 7.6 7.5 7.4 6 (ppm) 9 1.00 1.00 0.50 8 7 6 5 6 (ppm) 3 1.50 2 1 0
A: The question is based on the concept of organic spectroscopy. We need to identify the compound.
Q: Modify the structure to represent the organic product, if any, expected from each of the oxidation…
A:
Q: The following substrate is benzyl chloride that can undergo substitution reactions. Reaction of this…
A: SN1 reaction takes place in two steps. The first step is the formation of stable carbocation and the…
Q: a. CI CI C–C–CH,CH3 1 I HH b. CH3CH₂CH₂CHC1₂ -NH₂ KOC(CH3)3 DMSO Br C. CH3-C-CH₂CH3 1 Br d. Br Br…
A: The given reactions describe the conversion of dihallide to alkyne in the presence of base.
Q: state which of the following molecules show a pure rational microwave absorption spectrum and…
A: The molecules that show a pure rotational microwave absorption spectrum are those that have a…
Q: Concentration of NaOH (M): Molar mass of acetic acid: (g/mol) Titrate 5.00 mL of vinegar for each…
A: 1. Volume in liter multiplied by molarity (M) gives the number of moles of NaOH present in the…
Q: 1. Why does the tip of your buret need to be free of air bubbles?
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 4. Show the synthesis of acetophenone from bromobenzene. (You must use "old" reactions)
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: When 18.0 mL of a 6.86 x 10^-4 M lead acetate solution is combined with 12.0 mL of a 2.28 X10^-4 M…
A:
Q: What element forms an ion with an electronic configuration of [Ne] and a -2 charge? element symbol:
A: We have to tell which element forms an ion with an electronic configuration of [Ne] and a -2 charge.…
Q: An ionic compound if formed between the cation X+3 and the anion Y-2 What is the formula of this…
A: The numerical value of the two atoms' ion charges is crossed across when using the criss-cross…
Q: For the following reaction, 3.90 grams of methane (CH4) are mixed with excess carbon tetrachloride.…
A:
Q: arch <Chapter 11 Homework Draw the Structure Question 17 27 6354 ▼ Part A What is the correct…
A: IUPAC Name 1,4-dimethyl benzene
Q: (c) What is the pKa of the acid? (only confused on part c, answers for a and b are given)
A: We have been given a weak acid which we are titrating with a strong base sodium hydroxide. we need…
Q: The data below were collected for the following reaction: 2NO2(g) + F2(g) → 2NO₂F(g) R. Initial Rate…
A:
Q: Phosphorus pentachloride decomposes according to the chemical equation PC1, (g) PC13(g) + Cl₂(g) Kc…
A:
Q: Use the following equilibrium. H2(g) + CO2(g) H2O(g) + CO(g) K = 16.0 The initial partial…
A: H2(g) + CO2(g) ⇌ H2O(g) + CO(g) Equilibrium constant (K) = 16.0 Initially Partial pressure of H2…
Q: 4000 20- 3500 2500 1000 3000 2000 1500 500 30- 690,14 40- 964.95 50- 733.21 %Transmittance 60-…
A: The question is based on the concept of IR SPECTROSCOPY.. We need to assign the characteristic bands…
Q: 7. Silver (Ag) reacts with cyanide (CN) and oxygen (O₂) to make silver (I) cyanide (Ag(CN)2);…
A: Since you have asked multiple questions, we will solve the first one for you. If you want a specific…
Q: Determine the number of atoms in 27.0 grams of bromine, Br. (The mass of one mole of bromine is…
A: This problem is based on mole concept.
Q: Synthesis ??? Ph
A: After the conversion of reactant into product, we can figure out that an alkene functional group is…
Q: The following reaction is occurring on the surface of a catalyst. 2A(g ) → B(g ) The equation below…
A: The following reaction is occurring on the surface of a catalyst. 2A(g ) → B(g ) The equation below…
Q: What would be the rate order for the following reaction: 2H₂ + O2 --> 2H₂O A B с D zero order first…
A: Order of a reaction is an experimental quantity. It can't be determined from the given chemical…
Q: In this experiment, you will perform this copper(I)-TEMPO oxidation on a small substrate scope…
A:
Q: Arrange these ions according to ionic radius. 0²- F™ Largest radius Smallest radius N³- Answer Bank…
A: Isoelectronic species have same number of electrons. Here, O2-, N3- , Na+, Mg2+ and F- all have 10…
Q: Arrange these elements according to electron affinity. Most energy released by gaining an electron…
A: We have been given three elements Na,Mg and Cl and we have been asked to arrange these elements…
Q: At a certain temperature, 0.4811 mol of N₂ and 1.601 mol of H₂ are placed in a 3.00 L container.…
A: Since,Equilibrium constant or Kc is the ratio of the equilibrium concentrations of product over…
Q: Question 18 of 20 If a system does 84.0 kJ of work on its surroundings and releases 105 kJ of heat,…
A:
Q: A compound such as CH3CH₂CH₂OCH₂CH3 is classified as what type of organic compound? A) alcohol B)…
A: A condensed formula is a type of chemical formula that represents the atoms and bonds in a molecule…
Q: When aluminum is placed in concentrated hydrochloric acid, hydrogen gas is produced. 2 Al(s) + 6…
A:
Give detailed Solution with explanation needed..please explain
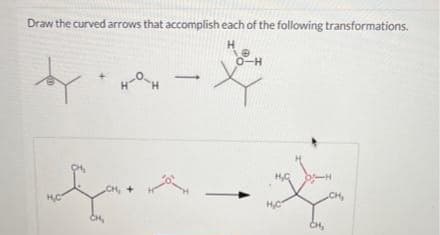
Step by step
Solved in 3 steps with 2 images

- Add curved arrows to the reactant side of the SN2SN2 reaction shown.For the reaction given below, draw a mechanism (curved arrows) and then predict which side of the reaction is favoured under equilibrium conditions.1. Which absorbs at higher frequencies: a C-H or C-D bond? Explain. 2. Predict which alkene will predominate at equilibrium. Explain.

