Q: An aqueous solution contains 0.417 M nitrous acid. Calculate the pH of the solution after the…
A: pH of buffer solution can be calculated using Henderson hasselbalch equation.
Q: Starting with potassium phthalimide as your source of nitrogen and using any other reagents of your…
A: Reagents are substances used in chemical reactions to cause a specific chemical change or to detect…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. ui BrMg 1. Dry THF…
A:
Q: All of the following contain a sp-hybridized functional group except a carboxylic acid. a nitrile.…
A: Hybridization is a process of combining two atomic orbitals for the generation of new degenerate…
Q: (CH₂)₂CHCH₂OC-OCH₂CH₂ H₂SO4 H₂O
A: In the given reaction (c) and (d) an ester reacts with H2SO4 followed by H2O i.e. acid hydrolysis of…
Q: Use the following information to answer the next question. The following reaction equation…
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. (CH3CH2)2CuLi 2. H3O*…
A: R2CuLi specifically undergoes 1,4-addition to α,β-unsaturated ketone due to soft nature of alkyl…
Q: Which compound below has lowest heat of hydrogenation the A: 1, 5-hex diene B: 3,4- hexadiene (E)-1,…
A: we have to determine the compound with lowest heat of hydrogenation
Q: 19.24 Cyclohexene can be converted to 1-cyclopentenecarbaldehyde by the following series of…
A:
Q: Which processes release heat? deposition of 35.0 g CO₂ condensing 90.0 mL of steam vaporizing 65.0…
A: When solid changes to liquid or gas, it will require energy, and hence heat is absorbed. When a…
Q: CaC2 (s) + 2 H₂0 (1) → Ca(OH)2(aq) 2 Ca(OH)2(aq) + C₂H₂(g) AH = ??? Ca(s) + 2 C(graphite) → CaC2(s)…
A:
Q: a) Identify H and predict the products. ON CHO 4대ㅇ HCN Y li Al H3 #30+ ↑ b) Cive the mechanism of…
A:
Q: The solubility product for silver chloride is 1.6 x 10−10. What is the molar solubility of silver…
A: This question can be solved using concepts of solubility. Solubility product of a sparingly salt…
Q: 1. PPh3 2. BuLi 3. O
A: The reaction involves Phosphorus Y-lide.
Q: Part A Define the second law of thermodynamics. For any spontaneous process, the entropy of the…
A: According to second law of thermodynamics, For a spontaneous process, the entropy of the universe…
Q: How would you synthesize the following compounds from benzene using reagents from the table? a C d…
A: The organic reactions takes place by adding various reagents, to get the desired final product.
Q: the References to access important values if needed for this question. An aqueous solution contains…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . It…
Q: What is the IUPAC name for the compound shown? tr
A: -CHO group is known as aldehyde group. The general formula of any aldehyde is R-CHO (R =H/carbon…
Q: what product is formed in this reaction? 10-07FM What product is formod (Ch₂ Ch 3) 3 N (1/11 (1
A: Although tertiary amines are tending to be less nucleophilic than secondary amines, they are still…
Q: The ionization constant for water (Kw) is 5.9 × 10–14 at 50 °C. What is the [H3O+], [OH–], pH, and…
A:
Q: How would you define conceptual problem solving ? Can you give an example?
A: We need to explain:- How would you define conceptual problem solving ? Can you give an example?
Q: Draw the reactants that would react to form this cyclohexene derivative in a Diels-Alder reaction…
A: In Diels-Alder reaction, diene reacts with dienophile to form an adduct in which the stereochemistry…
Q: Which bond is the more polar bond in each of the following pairs? Select the single best answer.…
A:
Q: What are the reactants and products when butane combusts with excess oxygen? Express your answer as…
A: When any hydrocarbon reacts with excess oxygen, then it is said to be combustion reaction. In…
Q: سلة 3. -C—CH+H-O-CHỊCH, H*
A:
Q: Choose the most likely regiochemical outcome for the reactants and conditions shown below: 1.…
A:
Q: Indicate whether each of the following compounds will gave an acidic, basic or neutral solution when…
A:
Q: Why is the isotope 36CI radioactive while 35Cl and ³7 Cl are not? Elements with an Thus, Cl-35 and…
A: We know that, isotopes which consists proton - neutron numbers even-even, even-odd, odd-even are…
Q: Select the intermediate formed from the rearrangement shown below. ob ✪
A: The C-C bond between C2 and C11 breaks, new bond between C7 and C11 is formed and charge is shifted…
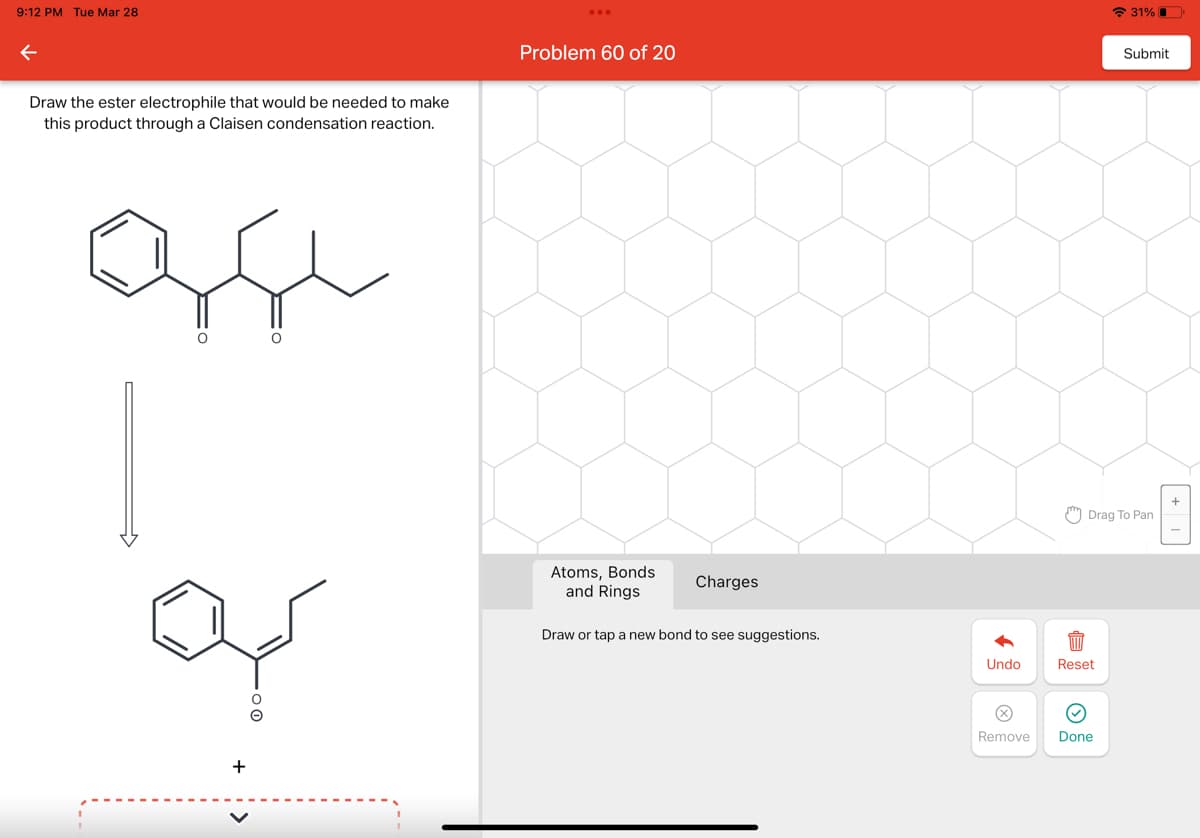
Q: Give the starting material and mechanism arrows needed to produce the given product by coordination.…
A: organic reaction mechanism.
Q: Calculating the LSFE of [Fe(H2O)6]Cl2, knowing that Δo = 10,400 cm–1 and P = 17,600 cm–1 Hints:…
A:
Q: The standard cell potential E°(cell) is +0.77 V for the redox reaction 2 Fe³+ (aq) + H₂(g) → 2 H(aq)…
A: The standard cell potential E°(cell) is +0.77 V for the redox reaction 2 Fe³+(aq) + H2(g) → 2 H+(aq)…
Q: Rank the following carbonyls according to their electrophilicity (1 = most) OEt O O xo NEt₂ O CF3 O
A:
Q: 1) The Name of the following Compound is NH₂ exc "Br
A: Nomenclature of substituted benzene: 1) naming is done by placing the substituent as a prefix to the…
Q: Search 9:08 PM Tue Mar 28 ← Draw the starting materials needed to complete this Robinson annulation…
A:
Q: A buffer solution contains 0.213 M NH3 and 0.265 M NH4Cl. What is the pH of the buffer solution…
A: 26.9 mL of 0.125 M NaOH was added to a buffer solution containing 0.213 M NH3 and 0.265 M NH4Cl.…
Q: Macmillan Learning Modify the structure to represent the organic product, if any, expected from each…
A: Generally, if we oxide a alcohol, at first it oxides in aldehyde and further oxidised and formed…
Q: What is the pH of 0.175 M methylammonium bromide, CH3NH3Br? (Kb of CH3NH2 = 4.4 × 10−4.)
A:
Q: Synthesize: 27) २८) ott
A: The given product from the benzene can be synthesized through Friedel-Crafts acylation and followed…
Q: Part A For the reaction below, what can you conclude about AG rxn for the reaction? H₂(g) + Br₂(g) 2…
A: The question is based on the concept of chemical thermodynamics. we need to predict standard Gibbs…
Q: In the reaction HSO4 (aq) + OH(aq) SO42 (aq) + H₂O(), the conjugate acid-base pairs are A. B. C. D.…
A: According to the Bronsted - Lowery concept of acid - base, " the Bronsted acids are those species…
Q: Identify and label the Brønsted-Lowry acid, its conjugate base, the Brønsted-Lowry base, and its…
A:
Q: OH 1.03 2. Z₁ or Me₂S .? OH
A:
Q: 2. Predict the product(s) for each of the following reactions. N (CH,CH,),NH, H* NaCNBH3 H₂O+ H3O+…
A:
Q: For the reaction 2H₂O(l) → 2H₂(g) + O2(g) 461 kJ and AH° = 572 kJ at 338 K AG° and 1 atm. = This…
A: Answer: Decomposition of water into hydrogen and oxygen gas is taking place and sign of Gibbs free…
Q: For the reaction 3Fe₂O3(s) + H₂(g)- →2Fe3O4(s) + H₂O(g) AH° = -6.00 kJ and AS° = 88.7 J/K At…
A: Spontaneity of a reaction depends on change in Gibbs free energy. If ΔGo < 0 , the reaction is…
Q: Unexpectedly, 7-bromocyclohepta-1,3,5-triene will readily dissolve in water. Draw the aromatic…
A: There are several conditions that must be met for a compound to be considered aromatic: The…
Q: 9 rea Use the References to access important values if needed for this question. A buffer solution…
A: Given Concentration of NaH2PO4 = 0.474 M Concentration of K2HPO4 = 0.290 M Volume = 125 ml = 125 x…
Q: 14.40 The equilibrium-constant expression for a reac- tion is K = [NH3]4[0₂]5 [NO] [H₂O]6 the What…
A: The equilibrium expression for the reaction is: Kc=NH34O25NO4H2O6 ......(1) Equilibrium constant is…
Q: [Review Topics] [References] Scoring: Your score will be based on the number of correct matches…
A:
Step by step
Solved in 3 steps with 3 images

- In the experiment where an Electrophillic aromatic substitution sulfonation reaction is conducted on Indigo to produce the food dye FD&C Blue #2(indigo blue or indigotine). show a mechanism showing a single sulfonation on a molecule of indigo.For problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent. (d) Sodium cyanide in dimethyl sulfoxide (e) Sodium azide in aqueous ethanol (f) Sodium hydrogen sulfide in ethanolThe base-promoted rearrangement of an a-haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. Jo Han Sow La NaOH, THF O Nat H3O+ OH NaOH, THF The mechanism involves the following 5 steps: -ō cyclopropanone intermediate 1. Abstraction of a proton to form enolate anion 1; 2. Formation of a cyclopropanone intermediate 2 with expulsion of chloride ion; 3. Addition of hydroxide ion to form tetrahedral intermediate 3; 4. Collapse of the tetrahedral intermediate and breakage of the three-membered ring to form carbanion intermediate 4; 5. Proton transfer to form the rearranged carboxylic acid. For the following reaction, draw the reaction out on paper, and then draw the structure of cyclopropanone intermediate 2 in the window. 1. NaOH, THF 2. H3O* OH
- The following reaction has a ΔSsystem < 0 O2(g) → 2O(g) T or F can you explain why this is false?For problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent. (g) Sodium methanethiolate (NaSCH3) in ethanolDraw reaction mechanism for week 2 A wittig reaction experiment: Week 1 -》 preparation of phosphonium salt Materials : triphenylphosphine (5.3g),methyl Bromoacetate 3.36g (2.1ml) , ethanol 30ml . The yield obtained was 8.93g .The percentage yield was 106.4% Week 2 -》 Formation of the ylide and wittig reaction Materials :Phosphonium salt (5g), napthyl-2- carboxaldehyde( 2.65g), 20 ml of water and 5 nl of Nahco3. Yield obtained was 0.51g . The percentage yield was 14.2%. Week 3: Solvent free wittig reaction Materials: Benzyltriphenylphosphonium chloride (0.5g ), 4 - Bromoabenzaldehyde (0.24g), Potassium phosphate (tribasi c) 0.275g .The yield obtained was 1.21g. The percentage yield was 35.6%.
- Scheme 1: Reagents: (a) acetic anhydride, sulfamic acid, 60°C,30min,91%; (b) Et3SiH,I2,0-5°C,30min,82%. 1) Draw the FULL, ARROW-PUSHING MECHANISM 2) Provide a verbalization of the mechanism for your reaction mechanism 3) What is happening at each step? What atom is nucleophilic? Which atom is electrophilic? Which bonds are made? Which bonds are broken? 4) Imagine you are trying to describe the reaction mechanism and I can't see your display - how would you describe the mechanism in a way that I know, that you know, what you are talking about.dedcue the major organic product from the following reations. be ablw to draw hydration halogenation hydrohalogenation, halohydrin formation, oxymercuratiom demercuration qnd hydroboration oxidation.Predict the reaction of methyl 3-oxocyclohexane-1-carboxylate with NaBH4/ h20 show the mechanism
- When the rate constants for the hydrolysis of several morpholine enamines of para-substituted propiophenones are determined at pH 4.7, the r value is positive; however, when the rates of hydrolysis are determined at pH 10.4, the r value is negative. a. What is the rate-determining step of the hydrolysis reaction when it is carried out in a basic solution? b. What is the rate-determining step of the hydrolysis reaction when it is carried out in an acidic solution?Predict the products of reaction of pent-1-yne with the following reagents. warm, concd. KMnO4, NaOHI. Propose a mechanism reaction for the reaction below. 2. Identify the major and the minor products.

