Q: Suppose a 500. mL flask is filled with 0.70 mol of H, and 1.9 mol of I,. The following reaction beco...
A: Number of moles of H2 = 0.7 mol Number of moles of I2 = 1.9 mol Volume of rea...
Q: 1. Name using IUPAC substitutive nomenclatare nles
A: Write IUPAC name by using rules of that,
Q: Nitrogen dioxide decomposes to nitric oxide and oxygen via the reaction: 2NO2 → 2NO + O2. In a parti...
A: Given Reaction:- 2NO2 → 2NO + O2
Q: Q2 (a)Explain why the following reaction is exothermic based on your knowledge of both bond enthalpy...
A: Enthalpy of reaction - Is the amount of heat evolved during the progression of a chemical reaction...
Q: 12. Select the correct statement for energy level n-9 a) b) The two s orbitals have the same energy ...
A:
Q: Which of the following equations is a mathematical definition of enthalpy? Ο ΔΗ- ΔΕ-ΡAV Ο ΔΗ-ΔΕ + ΡA...
A: enthalpy is the sum of the internal energy and the product of the pressure and volume of a thermodyn...
Q: Mixing water and arterial fluid in the reservoir of the machine is called secondary dilution. True...
A: mixture of arterial fluid with water and /or supplemental fluids is called primary dilution.
Q: What is the mass of taurine if the number of moles is 0.4249 mol? What is the number of moles in iro...
A:
Q: 2) 2-Mpentanol has the mass spectrum shown. Propose structures for fragment ions of m/z 88, 73, 70, ...
A: Mass spectrum is the technique used in analytical chemistry for the separation of ions based on the ...
Q: Find the energy per molecule of CO2 released in eV and compare this to the energy per CO2 molecule r...
A:
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Ni(s) + 02(g)→2N¡O(s) AH° ...
A: Given, 1) 2Ni(s) + O2(g) ➝ 2NiO(s) ∆H° = -479.4 kJ 2) 2Hg(l) + O2(g) ➝ 2HgO(s) ∆H° = -181.6 kJ 3...
Q: 6.21 Both of the following reactions involve nucleophilic substitution. The product of reaction (a) ...
A:
Q: The quantum numbers of the last electron of nickel could be: a) b) c) n= 3, € = 2, me =0, ms = 1/2 %...
A: To find the set of quantum number for last electron of nickel , we would first write the electronic ...
Q: Doubling a gas temperature from 100 K to 200 K causes a gas volume to double. Would you expect a sim...
A: Temperature in degree scale(oC) and kelvin scale(K) are related as following. Co+273.15=K
Q: 1. Below are reactions showing the acids dissolving in water. Draw a particle diagram' representing ...
A:
Q: а) Consider the reaction scheme below. -[•] PhLi HONH,, HCI A ELOH B D (i) Deduce the structures of ...
A: We have to give the structures of the compounds A, D, and the intermediates C. We also have to give ...
Q: 2NH3(g) + 202(g)-N20(g) + 3H20(1) Using standard thermodynamic data at 298K, calculate the free ener...
A:
Q: CH,CH,CH,CH,C
A: A tubular fixed bed reactor is a type of packed bed reactor. This is used for heterogeneous reaction...
Q: Wine goes bad soon after opening because the ethanol (CH,CH,OH) in it reacts with oxygen gas (0,) fr...
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: Name the highlighted chemical group in each molecule. Lewis structure name of highlighted group H H ...
A:
Q: What metabolic reaction is illustrated by the image below? * СООН COOH -OCOCH3 CH,COOH -он + Ester h...
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please re...
Q: For items 11 to 15 Predict the direction of the reaction that will be favored by the system below wh...
A:
Q: Use electronic factors to explain why methy carbanion is more stable than tertiary butyl carbanion
A: Answer is :
Q: Reaction (AGº) (kJ) a. 2S(s, rhombic) + 302(g) → 2S03(g) 2SO3(9) b. S(s, rhombic) + O2(g) → 2SO2(g) ...
A:
Q: There is a 15.85% by mass KCl (molecular mass is 74.5513 g mol) solution at 25°C. The density of the...
A: We know that , Osmotic pressure= (Molar concentration of solute*ideal gas constant*temperature)
Q: Aqueous sulfuric acid (H,SO,) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium su...
A: Sulfuric acid reacts with sodium hydroxide to form sodium sulfate. The equation for the balanced che...
Q: 4. Consider the three statements below. Which statement(s) is(are) true? 1. Hydration is a special c...
A: True or False !
Q: Which of the following statements is true about activation energy in chemical reactions? O By increa...
A:
Q: Enumerate the differences between a serological and a volumetric pipette and enumerate the uses, adv...
A: The main difference between the volumetric and serological pipette is that The volumetric pipette o...
Q: Tom measures a balloon to have an initial volume of 306.4 ml. When he causes the temperature of the ...
A: Using Charles's law we get the answer
Q: Calculate the mass of forsterite (Mg, SiO,) that contains a million (1.000 x 10°) magnesium atoms. B...
A:
Q: Q/Write the possible compounds from the reaction of one and a half mole of benzaldehyde with one mol...
A: Benzaldehyde reacts with acetone and undergoes condensation reaction. Here we have to find out the c...
Q: WHAT IS THE SIGNIFICANCE OF THE RATION OF STRENGTHS OF ACIDS AND BASES? what is the purpose of dryi...
A: To know about the extent to which any acid or base ionized in water when dissolved is the important ...
Q: A student heats 61.27 grams of silver to 97.71 °C and then drops it into a cup containing 82.16 gram...
A:
Q: What is the pH of a solution containing 0.642 mol L-1 of a weak acid with pKA = 3.01 and 0.865 mol L...
A:
Q: Rank the samples of gas described in the table below in order of increasing average speed of the ato...
A:
Q: Examine the molecules below. Which site is most basic? Which H is most acidic? Highlight this site. ...
A: Given:- we know most basic means which can donate its lone pair easily. Most acidic means which ...
Q: n atomic orbital
A:
Q: Please identify the asymmetric carborn in the following molecules and mark them with an X. There may...
A: Asymmetric carbon centre is the carbon center with sp3 hybridised and having four different valencie...
Q: Using the specific heat capacities of CO2 and air determine which sample (air or CO2) shows the grea...
A: Specific heat capacity is the amount of heat absorbed by 1 g of substance to raise its temperature b...
Q: 4. For the following aldohexoses which of them will give an optically inactive compound when reacted...
A: HNO3 is strong oxidizing agent option IV is correct
Q: Question 5 What is the pH of 0.01 M acetic acid solution (Ka = 1.8x10-5) O 3.22 O 3.55 O 3.37 O 4.24
A: Concentration of acetic acid = 0.01 M Ka = 1.8 × 10-5
Q: Use the systematic treatment to get the concentrations of all the species in a solution of: a.) 0.35...
A:
Q: Describe/explain the following : 1.) van der Waals forces A. dipole-dipole interaction B. dipole-...
A: Internolecular forces can be defined as the forces of interactions existing between molecules, atoms...
Q: What is the calibration correction of a 10-mL pipet that delivers 9.9300 g of H2O at 19.0°C which is...
A: A numerical problem based on dimensional analysis that is to be accomplished.
Q: In writing the equilibrium constant, the concentration of the product should be placed in the expone...
A:
Q: For the reaction of 2A - 3B, a plot of In[A] vs. time is linear with a slope of -0.20. What is the r...
A:
Q: An 85.00 g sample of an unknown compound contains 24.26 g molybdenum, 24.33 g sulfur, and the rest i...
A:
Q: -[ ] PhLi HONH,, HCI A C ELOH B D
A: The solution of the question is given below:
Q: For items 11 to 15 Predict the direction of the reaction that will be favored by the system below wh...
A: Answer is as follows
Step by step
Solved in 2 steps with 2 images

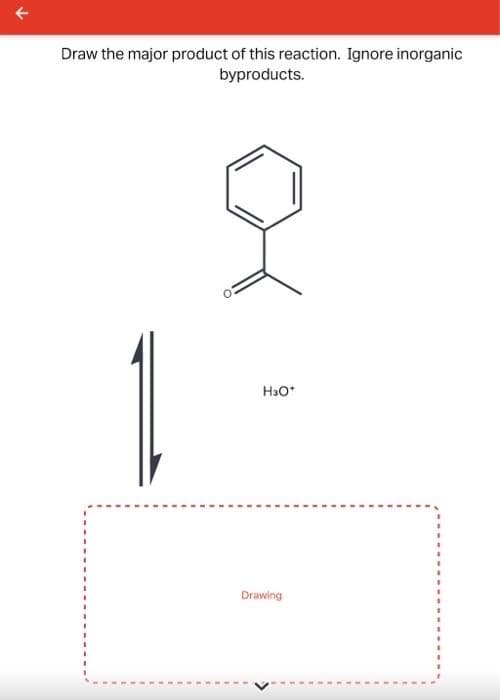
- Draw the major product of this reaction. Ignore inorganic byproducts. Assume thatthe water side product is continuously removed to drive the reaction towards products. Reagent= (CH2OH)2, TsOHDraw the major product of this reaction. Ignore inorganic byproducts.What is the major product of the reaction. Show all the arrow pushing mechanism to get to the major product.

