Q: HO-CH₂ H 1. OH 2. CH3(CH2)22 H₂N 3. H O H H CH₂OH H NH₂ O(CH₂)17CH3 H O C-N-C C H -0 4. 0 5. сно HIC…
A: Biomolecules can be defined as the compounds that are present in Living beings.
Q: Standardization of 1N Potassium Hydroxide Accurately weigh about 5 g of potassium biphthalate,…
A: Given that - Mass of potassium biphthalate = 5.1037 g Volume of Solution used = 75 mL Volume of…
Q: Naturally-occurring amino acids have the L-configuration. All amino acids are chiral except for…
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for…
Q: Given the reaction: H3AsO3 + I2 + NaHCO3 → H3AsO4 + NaI + CO2 + H2O The oxidation state of…
A: In this question, we will determine the oxidation state of As, reduction species, reducing agent and…
Q: 2. The reaction of between hydrazine (N₂H₁) and dinitrogen tetroxide (N₂0₂) in their liquid form is…
A:
Q: [Report answer to correct number of significant figures] Convert 25.9 lb/in to g/cm [1kg = 2.205 lb;…
A: Here we have to convert 25.9 lb/in into g/cm . This can be done by unit dimension analysis.
Q: How many stereocenters are present in the given structure (mark by star). Also determine the value…
A: If four different ligands are attached to a carbon, then it is called cenreocentre. The number of…
Q: 1. Solid tin reacts with diatomic chlorine to produce tin (IV) chloride. If 95 grams of tin reacts…
A: Given : 1. Mass of tin = 95 gram 2. Moles of CO2 produced = 1.8 moles 3. Moles of magnesium =…
Q: which among the three: inductive, hyperconjugation or steric effect?
A: Answer: Nucleophilic substitution is the characteristic reaction of alkyl halide that takes place…
Q: Identify the conjugate acids and bases in the substances mentioned in question 7(a) above.
A: Conjugate pairs are acids and bases that differ only by a proton (H+).
Q: 1. (a) Determine AH (kJ/mol) using Hess's law for the following reaction using the listed enthalpies…
A:
Q: Fluorometry is more selective and more sensitive than absorption spectrometry.Tell what is meant by…
A: Sensitivity: It is relatively easy to detect lower level of light from fluorescent emissions than…
Q: Show the retrosynthetic analysis of 1-Pentanoic acid and then further show the synthesis of the same…
A: The 1-pentanoic acid can be considered as derivative of substitituted acetic acid. Malonic ester…
Q: A. Starting from 1-butanol, synthesize the major compounds formed assigned from letters A to L.…
A: #A and B: NaH being a strong base, deprotonates ROH to form RONa. Then RONa reacts with R'-Br to…
Q: BOYLE'S LAW PROBLEM SOLVING 1.) An argon gas occupies 56.2 L at 760 torr. If the volume of gas is…
A:
Q: A. What is the cell notation of the set-up? B. What is the standard cell potential (Eºcell)? C. If…
A: #B: Since Au has a higher reduction potential, Au3+(aq) undergoes reduction to Au(s) nad Ni(s)…
Q: 5. Using a flow chart like that on page (6-2), outline a procedure to isolate each of the chemicals…
A: We have to draw a flow chart of procedures to separate the given compounds from a mixture .
Q: Cerebrovasular disease and hypertension is associated with نوع السؤال: خيار واحد High calcium intake…
A:
Q: The coefficient of hydroxide and the number of electrons transferred after balancing the reaction is…
A:
Q: I. Write the condensed structures corresponding to the following names: 1.…
A: See below
Q: 14.106 A voltaic cell is constructed in which the following reaction takes place: Cu (s) + 2Ag+ (aq)…
A:
Q: 5. A 295 g sample of unknown metal is at an initial temperature of 13.00 °C absorbs 75.0 kJ. The…
A: 5. Given: Mass of metal = 295 g Initial temperature = 13.00oC Final temperature = 295oC Heat…
Q: One dietary supplement capsule weighing 4.90 was ground into a fine powder. Two portions of the…
A: Standard addition involves adding different amount of analyte to a standard amount of sample and…
Q: ➡Al2O3 + Fe NaNO2 + O₂ NH3 1. FeO + Al 2. NaNO3 3. N₂ + H₂ 4. Al + Fe(NO3)2 5. SO2 + H₂O 6. Mg(OH)2…
A:
Q: b. O HC1, H₂O N₂OH H₂O
A:
Q: What is produced from triacylglycerol that forms a mixture of long chain carboxylate salts?
A: we obtain soaps. Base catalyzed hydrolysis of triacylglycerides led to glycerol and sodium salt of…
Q: Determine the oxidation number of the transition metal in the following metal complexes. -3 -2 -1 0…
A: Coordination complex consists of central metal ion surrounded by molecules known as ligands.
Q: Determine AH for the following reaction, N₂(g) + 3 H₂(g) → 2 NH3(g) given the thermochemical…
A:
Q: Five white, 500-mg uncoated ascorbic acid (AA) tablets with an average weight of 0.6100-g were…
A: To calculate the amount of ascorbic acid present in a uncoated ascorbic acid tablet having an…
Q: Attempt 4 After 63.0 min, 34.0% of a compound has decomposed. What is the half-life of this reaction…
A:
Q: 2.a) Calculate the actual Concentration of Stock Solution (1) in ppm Na+. b) Calculate the Volume of…
A: Since you have asked multiple questions, we will solve first question (question-2) for you. For…
Q: Copper has a density of 8.96 g/cc. If a cylinder of copper weighing 42.388 g is dropped into a…
A:
Q: How many grams of fluorine will react completely with 1.000 gram of sulfur to produce 1.00 mole of…
A: Given, Mass of S = 1.000 g Moles of sulfur tetraflouride = 1.00 mole Then, How many grams of…
Q: 2. Evaluate each of the following expressions. State the answer to the proper number of significant…
A: Rules for writing significant figures in addition subtraction multiplication and division; In case…
Q: Exactly 6.00 ml of acetic acid (density = 1.05 g/mL), MW=60.05) was added to a 1-liter volumetric…
A: 6.00 mL of acetic acid was added to a 1 L flask and filled with distilled water . A conductance…
Q: Amount of 12 absorbed by the unsaturated fatty acids in 1 g of the fat sample O lodine number O Acid…
A: Iodine number is defined as the amount of iodine consumed by 100 g of fat.
Q: The four dyes used for ph determination.
A: pH is defined as the negative logarithm of hydrogen ions present in solution.
Q: 2. Complete the following tdde with the needed [H3O+], [CH], and pH, and dassify the solution as…
A:
Q: Synthesize B from A keeping stereochemistry in mind: N 111 A у ome HC
A: In this question, we will synthesized B from starting material A and also keeping stereochemistry in…
Q: hat will bring about dissociation of a diatomic molec aving a dissociation energy of 390.4 kJ mol-¹.
A:
Q: Q1- A 0.58 mol sample of organic compound is burned in a calorimeter whose heal capacity equals…
A: As you post multiple question we are supposed to answer only one question. Kindly repost remaining…
Q: Many plastic bags are made of polyethylene. Despite only containing dispersion forces, the material…
A:
Q: s). Te supematant quid was iated with Calculate otal hardness (this includes the Mg2+) of the water…
A: Here we have to determine hardness of water.
Q: 8. An electric current is passed through a solution of Cobalt (II) nitrate using inert electrodes.…
A: Strongest oxidizing agent is the one which has the highest reduction potential while strongest…
Q: A 0.9134-g of KIO3 is dissolved in 500-mL distilled water. A 50.0-mL aliquot portion was run down…
A:
Q: Shown here is a splitting diagram. What geometry does this splitting diagram represent? dx2-y2 dxz…
A:
Q: Calculate the mass of sodium-24 that will remain in a 20.0 g sample after 75 hours. Sodium has a…
A: Chemical kinetics is branch of chemistry in which we deal with the speed at which rate of reaction…
Q: Milligram of base required to completely hydrolyze & neutralize 1 g of triglyceride/fat sample…
A:
Q: Consider the metal complexes shown below. Identify the geometry of each complex. trigonal planar…
A:
Q: 9. Explain briefly why ethers are used as solvents?
A: According to bartleby guidelines we're supposed to answer only first question among these questions…
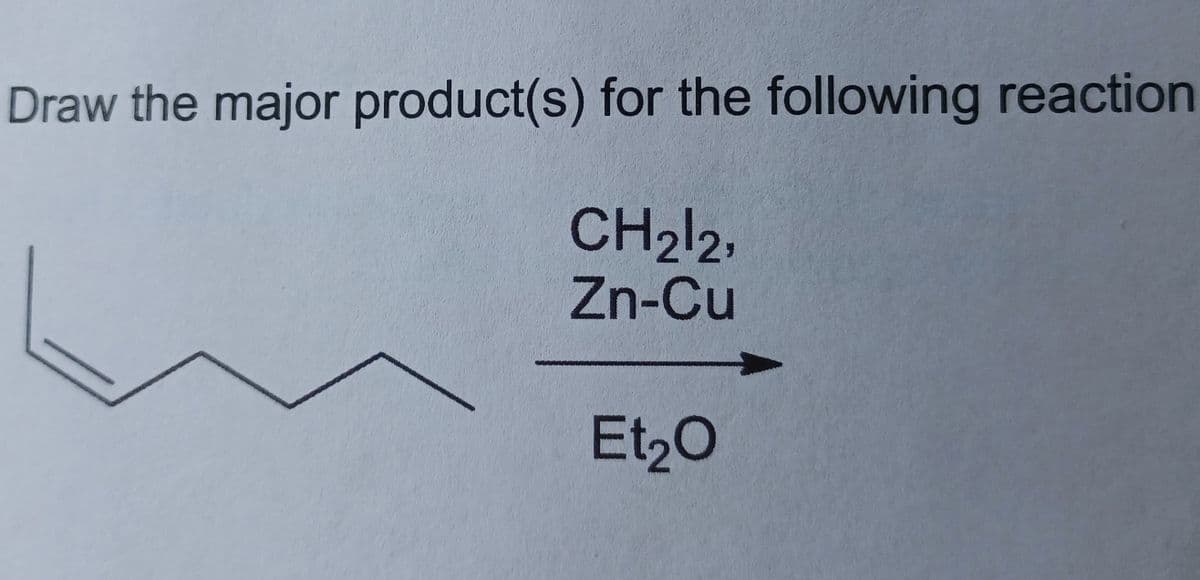
Step by step
Solved in 2 steps with 2 images

- 2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3What is the expected major product of the reaction? 1. A 2. B 3. C 4. No reactionWhich of the following reacts the fastest with NaOH, H2O? A. cis-2,3-dimethyloxirane B. ethylene oxide (oxirane) C. trans-2,3-dimethyloxirane D. 2,2,3,3-tetramethyloxirane

